
-
 Tokyo, Seoul shares gain, war sends oil higher
Tokyo, Seoul shares gain, war sends oil higher
-
Artemis mission headed for first lunar flyby since 1972

-
 South Korea president says regrets 'reckless' drones sent to North
South Korea president says regrets 'reckless' drones sent to North
-
Coughlin captures third LPGA title at Aramco Championship

-
 What to know about the Artemis 2 mission's Moon flyby
What to know about the Artemis 2 mission's Moon flyby
-
Mystique of the green jacket endures as Masters looms

-
 In El Salvador's mass trials, 'the innocent pay for the guilty'
In El Salvador's mass trials, 'the innocent pay for the guilty'
-
Trump makes stark threat to Iran after US airman rescued

-
 Artemis astronauts ready for Moon flyby on fifth day of historic mission
Artemis astronauts ready for Moon flyby on fifth day of historic mission
-
Israel renews Lebanon strikes, forces Syria border crossing closed

-
 Eagle-eyed Spaun snatches Texas Open victory
Eagle-eyed Spaun snatches Texas Open victory
-
Brown, Tatum propel Celtics in win over Raptors

-
 Paul battles past Burruchaga to win ATP Houston title
Paul battles past Burruchaga to win ATP Houston title
-
Major sponsors drop Kanye West London gigs as PM voices concern

-
 Inter close in on Serie A title by thumping Roma
Inter close in on Serie A title by thumping Roma
-
Trump makes foul-mouthed threat to Iran after US airman rescued

-
 Monaco sink Marseille for seventh Ligue 1 win in a row
Monaco sink Marseille for seventh Ligue 1 win in a row
-
Inter thump Roma to extend Serie A lead to nine points

-
 Lebanon's Christians mark Easter in solidarity with war-hit south
Lebanon's Christians mark Easter in solidarity with war-hit south
-
Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years

-
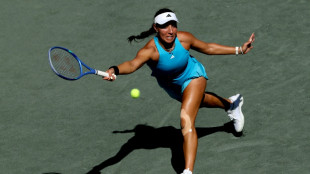 Pegula romps to WTA Charleston Open victory
Pegula romps to WTA Charleston Open victory
-
David six-hitting spree powers Bengaluru to IPL win

-
 Union draw leaves St Pauli stranded in Bundesliga drop zone
Union draw leaves St Pauli stranded in Bundesliga drop zone
-
UK police arrest protesters near base used by US

-
 Trump issues foul-mouthed threat to Iran after US airman rescued
Trump issues foul-mouthed threat to Iran after US airman rescued
-
Alcaraz plans to play full clay-court season, get 'socks dirty'

-
 'Super Mario Galaxy' blasts off in N. America box office debut
'Super Mario Galaxy' blasts off in N. America box office debut
-
Artemis astronauts begin fifth day on historic Moon mission

-
 Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup
Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup
-
Trump draws criticism with fiery Easter message on Iran

-
 OPEC+ hikes oil production quotas, issues warning
OPEC+ hikes oil production quotas, issues warning
-
British PM slams London event for booking Kanye West, sponsor quits

-
 Pogacar wins joint-record third Tour of Flanders
Pogacar wins joint-record third Tour of Flanders
-
Trump threatens 'hell' for Iran over Strait of Hormuz

-
 Shami, Pant help Lucknow beat Hyderabad in nervy IPL clash
Shami, Pant help Lucknow beat Hyderabad in nervy IPL clash
-
What we know about the race to rescue downed US airman in Iran

-
 US commandos went deep into Iran to rescue downed airman: media
US commandos went deep into Iran to rescue downed airman: media
-
Liberated McIlroy eyes more Masters magic after career Slam

-
 Van Dijk apologises for Liverpool thumping by Man City
Van Dijk apologises for Liverpool thumping by Man City
-
British PM slams London festival for booking Kanye West

-
 'Choose peace': Pope marks first Easter under cloud of Mideast war
'Choose peace': Pope marks first Easter under cloud of Mideast war
-
British royals attend Easter service without Andrew

-
 US media says commandos probed deep into Iran to rescue downed airman
US media says commandos probed deep into Iran to rescue downed airman
-
Revellers parade giant penises to dash stigma in Japan's fertility festival

-
 Artemis astronauts glimpse Moon's 'Grand Canyon' ahead of historic lunar flyby
Artemis astronauts glimpse Moon's 'Grand Canyon' ahead of historic lunar flyby
-
Middle East war hits Britain's fish and chip shops

-
 Artemis astronauts to study the Moon's surface using mainly their eyes
Artemis astronauts to study the Moon's surface using mainly their eyes
-
Second US airman downed over Iran 'SAFE and SOUND': Trump

-
 Indonesia lays to rest peacekeepers killed in Lebanon
Indonesia lays to rest peacekeepers killed in Lebanon
-
Pharmaceutical logistics in demand as war rattles supply chains


Feds Move To Axe Key DEA Marijuana Rules Clearing the Way for MMJ's FDA Compliant HD and MS Trials
Under President Trump's Executive Order 14192 ("Unleashing Prosperity Through Deregulation"), DOJ's notice eliminates several DEA proposals long cited as "in process".
Under President Trump's Executive Order 14192 ("Unleashing Prosperity Through Deregulation"), DOJ's notice eliminates several DEA proposals long cited as "in process".
WASHINGTON, DC / ACCESS Newswire / September 11, 2025 / In a sweeping deregulatory action, the U.S. Department of Justice today withdrew 16 pending rulemakings and 38 projected rules , including multiple Drug Enforcement Administration (DEA) initiatives touching medical marijuana research and manufacturing. For MMJ International Holdings, MMJ BioPharma Cultivation, and MMJ BioPharma Labs , the message is unmistakable: there are no more DEA procedural fig leaves to delay legitimate, FDA compliant science.

Under President Trump's Executive Order 14192 ("Unleashing Prosperity Through Deregulation") , DOJ's notice eliminates several DEA proposals long cited as "in process" most notably:
Medical Marijuana and Cannabidiol Research Expansion: RIN 1117-AB83
Amendments to DEA Hearing Regulations:RIN 1117-AB50
Analytical Labs and Hemp: RIN 1117-AB77:
Revision of Application for Manufacturing Quota: RIN 1117-AB86
"This DOJ notice proves what we've said for years: the DEA kept MMJ and other companies in limbo by pointing to phantom rulemakings. Those rules will be withdrawn. There are no more excuses," said Duane Boise , President & CEO of MMJ BioPharma. "Administrator Terry Cole can approve our registration today under the Controlled Substances Act and let science move forward. The longer he waits, the more patients suffer."
Seven Years, Two Orphan Drug Designations, and a DEA Maze
MMJ applied for DEA registration in 2018 to produce pharmaceutical-grade cannabis for FDA-authorized clinical trials targeting Huntington's disease and Multiple Sclerosis. The company secured FDA Orphan Drug Designations , built a DEA-spec facility , and passed inspections - yet remains stalled by administrative gamesmanship MMJ and allies describe as "unconstitutional, shifting, and retaliatory."
The withdrawal of RIN 1117-AB50 (DEA Hearing Regulations) is particularly salient following Supreme Court rulings (e.g., Axon and Jarkesy ) and DOJ's own admissions regarding ALJ insulation. With AB50 now off the agenda, the DEA cannot credibly claim imminent reform of its in-house tribunals - leaving the status quo both un-reformed and constitutionally suspect.
What DOJ's Withdrawal Means Now
1) Potentially No more "we're waiting on a rule."
DEA will lose the shield of pending rule-making. Applications like MMJ's must be decided under the current Controlled Substances Act and existing regulations - promptly and on the record.
2) Pathways narrow-so decisions matter more.
With RIN 1117-AB83 (the Medical Marijuana and CBD Research Expansion) withdrawn, the agency is not implementing the streamlined timelines, electronic processes, or supply safeguards many researchers hoped for. That places greater urgency on direct administrative decisions by DEA leadership and potential congressional or FDA oversight .
3) The pattern problem is bigger than MMJ.
MMJ is aware of other companies that suffered unfair treatment, misrepresentations, and apparent intentional obstruction - including registrants that never grew, or operations left idle and bankrupt while compliant science was sidelined. Today's withdrawal highlights years of mission drift and mismanagement .
"If cannabis is 'too dangerous' to research here, why is the VA seeking to import it from overseas?" Boise added. "American patients and veterans deserve U.S. made, FDA regulated medicine - not bureaucratic delay."
What Administrator Terry Cole Must Do
Approve MMJ's Registration Now.
MMJ has met DEA and FDA standards. No new rule is required to act.Publish Clear, Consistent Guidance Under Existing Law.
End moving goalposts - particularly around "bona fide supply agreements" and quota justifications.Clean Up the Hearing Process.
In light of constitutional rulings and the abandonment of AB50, provide a transparent pathway that respects Article III oversight where appropriate.Establish Timely, Public Decision Milestones.
Even without AB83, Administrator Cole can impose internal deadlines that honor congressional intent to expedite research and ensure adequate supply .
Patients Can't Wait
Huntington's disease is a progressive, fatal neurodegenerative disorder with no cure and limited symptomatic relief.
Multiple Sclerosis causes irreversible nerve damage and long-term disability.
FDA-compliant cannabis medicines - such as MMJ's soft-gel formulations - could address spasticity, pain, and neuroinflammation under rigorous clinical controls.
"This isn't about dispensaries or Big Weed - it's about real, prescription grade medicine," Boise said. "Congress passed the Research Expansion Act in 2022 for a reason. It's time for DEA to respect the law's intent and stop blocking clinical progress."
About MMJ BioPharma
MMJ BioPharma is developing FDA-regulated cannabinoid therapeutics for Huntington's disease (MMJ-002) and Multiple Sclerosis (MMJ-001) , each with FDA Orphan Drug Designation . MMJ's pharmaceutical soft-gel capsules are manufactured to cGMP standards. The company is pursuing a fully compliant U.S. pathway and maintains international partnerships for botanical supply and EU distribution .
MMJ is represented by attorney Megan Sheehan .
CONTACT:
Madison Hisey
[email protected]
203-231-8583
SOURCE: MMJ International Holdings
View the original press release on ACCESS Newswire
T.Ward--AMWN

