
-
 Case closed for Morocco despite Senegal Afcon outrage
Case closed for Morocco despite Senegal Afcon outrage
-
22 migrants die off Greece after six days at sea: survivors

-
 Henderson backs England's White after Wembley boos
Henderson backs England's White after Wembley boos
-
Zelensky visits UAE, Qatar for air security talks with Gulf

-
 Hollingsworth upsets Hunter Bell as Gout Gout fails to fire in Melbourne
Hollingsworth upsets Hunter Bell as Gout Gout fails to fire in Melbourne
-
Iran footballers pay tribute to victims of school strike

-
 Questions over Israel's interceptor stockpiles as Mideast war drags on
Questions over Israel's interceptor stockpiles as Mideast war drags on
-
Sweet heist? Nestle says 12 tonnes of KitKat stolen

-
 Pope denounces widening gap between the rich and poor on Monaco visit
Pope denounces widening gap between the rich and poor on Monaco visit
-
Yemen's Houthi enter war with missile targeting Israel

-
 USS Gerald Ford arrives in Croatia for maintenance
USS Gerald Ford arrives in Croatia for maintenance
-
Antonelli leads Mercedes 1-2 as Verstappen suffers qualifying shock

-
 Verstappen calls his Red Bull 'undriveable' after more woes
Verstappen calls his Red Bull 'undriveable' after more woes
-
Antonelli takes pole for Japanese Grand Prix in Mercedes 1-2

-
 Millions angry with Trump expected to fill American streets
Millions angry with Trump expected to fill American streets
-
Attacks across Middle East as Iran war enters second month

-
 Late surge lifts Thunder, Celtics rally to down Hawks
Late surge lifts Thunder, Celtics rally to down Hawks
-
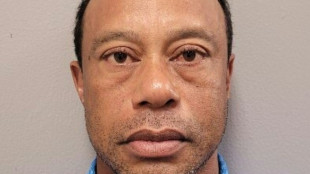
Tiger Woods arrested, charged with DUI after Florida crash
-
 Antonelli leads Mercedes one-two in final Japan practice
Antonelli leads Mercedes one-two in final Japan practice
-
Unease for Iranian-Canadians after shooting at ayatollah critic's gym

-
 Sequins, slogans, conspiracies: Inside the right-wing culture at CPAC
Sequins, slogans, conspiracies: Inside the right-wing culture at CPAC
-
NBA fines T-Wolves center Reid $50,000 for ripping refs

-
 Sinner ousts Zverev to book Miami Open final with Lehecka
Sinner ousts Zverev to book Miami Open final with Lehecka
-
McKellar hails 'special memory' after Waratahs stun Brumbies

-
 Best Crypto Roth IRA Company in the US Announced (2026 Update)
Best Crypto Roth IRA Company in the US Announced (2026 Update)
-
Tuchel takes positives from scrappy England draw against Uruguay

-
 Japanese star Sakamoto signs off with fourth world skating gold
Japanese star Sakamoto signs off with fourth world skating gold
-
Tuchel disappointed after England fans boo White

-
 US envoy hopeful on Iran talks as strikes target nuclear facilities
US envoy hopeful on Iran talks as strikes target nuclear facilities
-
Controversial African champions Morocco salvage Ecuador draw on Ouahbi debut

-
 Dutch end Norway's unbeaten run as Haaland rests
Dutch end Norway's unbeaten run as Haaland rests
-
'Strait of Trump': US president says Iran must open key waterway

-
 Wirtz steals show as Germany win thriller in Switzerland
Wirtz steals show as Germany win thriller in Switzerland
-
White jeered on England return as Uruguay snatch friendly draw

-
 Tiger Woods arrested, charged with DUI after Florida crash: police
Tiger Woods arrested, charged with DUI after Florida crash: police
-
Oyarzabal double fires Spain to win over Serbia

-
 More to IOC gender testing than appeasing Trump: ex-IOC executive
More to IOC gender testing than appeasing Trump: ex-IOC executive
-
Japan's Sakamoto ends career with fourth world skating title

-
 'Whatever it takes' - Sabalenka faces Gauff for second straight Miami Open crown
'Whatever it takes' - Sabalenka faces Gauff for second straight Miami Open crown
-
US hopes for Iran meetings 'this week': envoy Witkoff

-
 Uncertainty over war-induced oil crisis dominates key energy summit
Uncertainty over war-induced oil crisis dominates key energy summit
-
Czech Lehecka beats France's Fils to reach Miami Open final

-
 No pressure? Pochettino urges US co-hosts to 'play free' at World Cup
No pressure? Pochettino urges US co-hosts to 'play free' at World Cup
-
Duckett eager to show hunger for England success after Ashes flop

-
 'We are ready': astronauts arrive at launch site for Moon mission
'We are ready': astronauts arrive at launch site for Moon mission
-
Fishy trades before major news spark insider trading allegations

-
 Tiger Woods involved in Florida car crash: reports
Tiger Woods involved in Florida car crash: reports
-
WTO reform talks coming to the crunch

-
 Renaissance master Raphael honored at New York's Met museum
Renaissance master Raphael honored at New York's Met museum
-
At 'Davos of energy', AI looks to gas to power its rapid expansion


iTolerance, Inc. Appoints Sandra Cohen Kalter to Strategic Advisory Group
Legal & regulatory executive with extensive expertise in FDA law and regulatory leadership for one of the world's leading medical device companies
Legal & regulatory executive with extensive expertise in FDA law and regulatory leadership for one of the world's leading medical device companies
MIAMI, FL / ACCESS Newswire / September 22, 2025 / iTolerance, Inc. ("iTolerance" or the "Company"), an early-stage privately-held biotechnology company focused on the development of innovative regenerative medicines, today announced the appointment of Sandra Cohen Kalter to its Strategic Advisory Group.
"This appointment represents another step toward bolstering our ability to navigate complex regulatory pathways, potentially accelerate clinical translation and unlock the full potential of our pipeline. Our Strategic Advisory Group was formed to provide strategic guidance from individuals with a diverse background of expertise to support our initiatives, and Sandra is a wonderful addition that provides valuable insight and expertise. Over the course of her career, Sandra has navigated a number of regulatory pathways, inspections and integrations which have provided her with a depth and breadth of expertise that we believe will be instrumental as we move our development plans forward," commented Anthony Japour, Chief Executive Officer of iTolerance.
Mrs. Kalter added, "I am pleased to serve in this capacity for iTolerance and leverage my knowledge and experience from my career to help advance this important technology forward. iTolerance's platform technology, which has the potential to eliminate the need for life-long immunosuppression, continues to demonstrate promise, and I look forward to bolstering the Company's efforts to provide patients with a treatment option where there remains significant unmet need."

Sandra Cohen Kalter is a seasoned senior legal executive most recently serving as Vice President & Chief Regulatory Counsel at Medtronic plc ("Medtronic"), overseeing global strategies in regulatory compliance, quality, clinical affairs and medical safety for a world-renowned medical-device leader. She has led transformative enterprise-wide initiatives, including a patient safety and quality program that delivered major reductions in FDA findings and field corrective actions.
Additional career highlights include:
Built and managed an in-house FDA legal team that navigated 17 regulatory inspections in a single year, ensuring product continuity
Guided due diligence and integration efforts for more than 27 acquisitions, led regulatory responses to FDA violations and high-profile media scrutiny, and helped steer approval strategies for groundbreaking devices like the Evolut TAVR system and Micra leadless pacemaker
Previously served as FDA/Health Care Counsel at King & Spalding LLP, where she advised on complex regulatory filings and FDA panel proceedings
Received several awards including: Service to FDLI Award, Food and Drug Law Institute, 2023; Rapaport Lifetime Achievement Award, Juvenile Diabetes Research Foundation, 2019; Medtronic Wallin Leadership Award, June 2010; Marketing Campaign Team Award, Spinal & Biologics "Just the Facts," 2010; Medtronic Star of Excellence Award, 2009
Member of AdvaMed (Advanced Medical Technology Association) Legal Committee and Business Case for Quality Working Group
Co-Chair of AdvaMed Advertising and Promotion Working Group , Off-Label Working Group, and Business Case for Quality Working Group
Board of Directors of Food and Drug Law Institute (FDLI)
Board of Directors of Juvenile Diabetes Research Foundation (JDRF) and Corporate Recruitment Chair of JDFR One Walk
Member of Food and Drug Law Institute Medical Devices Advisory Committee
Mrs. Kalter attended George Washington University, The National Law Center and received a Juris Doctor Degree and a Bachelor of Science in Journalism from Northwestern University.
About iTolerance, Inc.
iTolerance is a regenerative medicine company developing technologies to enable tissue, organoid or cell therapy without requiring life-long immunosuppression. Leveraging its proprietary biotechnology-derived Streptavidin-FasL fusion protein/biotin-PEG microgel (SA-FasL microgel) platform technology, iTOL-100, iTolerance is advancing a pipeline of programs using both allogenic cadaveric and stem cell-derived pancreatic islets to potentially cure Type 1 diabetes. Utilizing iTOL-100 to induce local immune tolerance, the Company is developing its lead indication as a potential cure for Type 1 Diabetes without the need for life-long immunosuppression. Additionally, the Company is developing iTOL-201 for treating liver failure by utilizing hepatocytes and iTOL-401 as a nanoparticle formulation for large organ transplants without the need for life-long immunosuppression. For more information, please visit itolerance.com.
Forward-Looking Statements
This press release contains "forward-looking statements" within the meaning of the "safe-harbor" provisions of the Private Securities Litigation Reform Act of 1995. When used herein, words such as "anticipate", "being", "will", "plan", "may", "continue", and similar expressions are intended to identify forward-looking statements. In addition, any statements or information that refer to expectations, beliefs, plans, projections, objectives, performance or other characterizations of future events or circumstances, including any underlying assumptions, are forward-looking.
All forward-looking statements are based upon the Company's current expectations and various assumptions. The Company believes there is a reasonable basis for its expectations and beliefs, but they are inherently uncertain. The Company may not realize its expectations, and its beliefs may not prove correct. Actual results could differ materially from those described or implied by such forward-looking statements as a result of various important factors, including, without limitation, anticipated levels of revenues, future national or regional economic and competitive conditions, and difficulties in developing the Company's platform technology. Consequently, forward-looking statements should be regarded solely as the Company's current plans, estimates and beliefs. Investors should not place undue reliance on forward-looking statements. The Company cannot guarantee future results, events, levels of activity, performance or achievements. The Company does not undertake and specifically declines any obligation to update, republish, or revise any forward-looking statements to reflect new information, future events or circumstances or to reflect the occurrences of unanticipated events, except as may be required by law.
Investor Contact
Jenene Thomas
Chief Executive Officer
JTC Team, LLC
T: 908.824.0775
[email protected]
Media Contact
Susan Roberts
T:202.779.0929
[email protected]
SOURCE: iTolerance, Inc.
View the original press release on ACCESS Newswire
Ch.Kahalev--AMWN
