
-
 Zelensky visits UAE, Qatar for air security talks with Gulf
Zelensky visits UAE, Qatar for air security talks with Gulf
-
Hollingsworth upsets Hunter Bell as Gout Gout fails to fire in Melbourne

-
 Iran footballers pay tribute to victims of school strike
Iran footballers pay tribute to victims of school strike
-
Questions over Israel's interceptor stockpiles as Mideast war drags on

-
 Sweet heist? Nestle says 12 tonnes of KitKat stolen
Sweet heist? Nestle says 12 tonnes of KitKat stolen
-
Pope denounces widening gap between the rich and poor on Monaco visit

-
 Yemen's Houthi enter war with missile targeting Israel
Yemen's Houthi enter war with missile targeting Israel
-
USS Gerald Ford arrives in Croatia for maintenance

-
 Antonelli leads Mercedes 1-2 as Verstappen suffers qualifying shock
Antonelli leads Mercedes 1-2 as Verstappen suffers qualifying shock
-
Verstappen calls his Red Bull 'undriveable' after more woes

-
 Antonelli takes pole for Japanese Grand Prix in Mercedes 1-2
Antonelli takes pole for Japanese Grand Prix in Mercedes 1-2
-
Millions angry with Trump expected to fill American streets

-
 Attacks across Middle East as Iran war enters second month
Attacks across Middle East as Iran war enters second month
-
Late surge lifts Thunder, Celtics rally to down Hawks

-
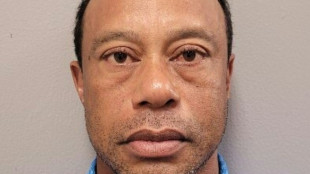 Tiger Woods arrested, charged with DUI after Florida crash
Tiger Woods arrested, charged with DUI after Florida crash
-
Antonelli leads Mercedes one-two in final Japan practice

-
 Unease for Iranian-Canadians after shooting at ayatollah critic's gym
Unease for Iranian-Canadians after shooting at ayatollah critic's gym
-
Sequins, slogans, conspiracies: Inside the right-wing culture at CPAC

-
 NBA fines T-Wolves center Reid $50,000 for ripping refs
NBA fines T-Wolves center Reid $50,000 for ripping refs
-
Sinner ousts Zverev to book Miami Open final with Lehecka

-
 McKellar hails 'special memory' after Waratahs stun Brumbies
McKellar hails 'special memory' after Waratahs stun Brumbies
-
Best Crypto Roth IRA Company in the US Announced (2026 Update)

-
 Tuchel takes positives from scrappy England draw against Uruguay
Tuchel takes positives from scrappy England draw against Uruguay
-
Japanese star Sakamoto signs off with fourth world skating gold

-
 Tuchel disappointed after England fans boo White
Tuchel disappointed after England fans boo White
-
US envoy hopeful on Iran talks as strikes target nuclear facilities

-
 Controversial African champions Morocco salvage Ecuador draw on Ouahbi debut
Controversial African champions Morocco salvage Ecuador draw on Ouahbi debut
-
Dutch end Norway's unbeaten run as Haaland rests

-
 'Strait of Trump': US president says Iran must open key waterway
'Strait of Trump': US president says Iran must open key waterway
-
Wirtz steals show as Germany win thriller in Switzerland

-
 White jeered on England return as Uruguay snatch friendly draw
White jeered on England return as Uruguay snatch friendly draw
-
Tiger Woods arrested, charged with DUI after Florida crash: police

-
 Oyarzabal double fires Spain to win over Serbia
Oyarzabal double fires Spain to win over Serbia
-
More to IOC gender testing than appeasing Trump: ex-IOC executive

-
 Japan's Sakamoto ends career with fourth world skating title
Japan's Sakamoto ends career with fourth world skating title
-
'Whatever it takes' - Sabalenka faces Gauff for second straight Miami Open crown

-
 US hopes for Iran meetings 'this week': envoy Witkoff
US hopes for Iran meetings 'this week': envoy Witkoff
-
Uncertainty over war-induced oil crisis dominates key energy summit

-
 Czech Lehecka beats France's Fils to reach Miami Open final
Czech Lehecka beats France's Fils to reach Miami Open final
-
No pressure? Pochettino urges US co-hosts to 'play free' at World Cup

-
 Duckett eager to show hunger for England success after Ashes flop
Duckett eager to show hunger for England success after Ashes flop
-
'We are ready': astronauts arrive at launch site for Moon mission

-
 Fishy trades before major news spark insider trading allegations
Fishy trades before major news spark insider trading allegations
-
Tiger Woods involved in Florida car crash: reports

-
 WTO reform talks coming to the crunch
WTO reform talks coming to the crunch
-
Renaissance master Raphael honored at New York's Met museum

-
 At 'Davos of energy', AI looks to gas to power its rapid expansion
At 'Davos of energy', AI looks to gas to power its rapid expansion
-
Israel hits Iran nuclear sites as Washington trails end to war

-
 US court overturns $16.1 bn judgment against Argentina over oil firm seizure
US court overturns $16.1 bn judgment against Argentina over oil firm seizure
-
England quick Tongue backs Cooley to make him a better bowler


Congress Trying to Limit Marijuana Rescheduling, but DEA Can Still Act Now
Administrator Cole, you can fight illicit drugs and build a lawful, GMP-grade cannabinoid supply chain that shrinks the black market through science. Approve qualified marijuana manufacturers and research investigators now. Patients are waiting.
Administrator Cole, you can fight illicit drugs and build a lawful, GMP-grade cannabinoid supply chain that shrinks the black market through science. Approve qualified marijuana manufacturers and research investigators now. Patients are waiting.
WASHINGTON, DC / ACCESS Newswire / September 24, 2025 / An open letter to Administrator Terry Cole and the DEA leadership team.
First, credit where it's due. The DEA's frontline work-seizing fentanyl, shutting down cartel pipelines, stopping illicit shipments, and engaging communities all saves lives. Your teams of community outreach specialists, diversion investigators, and lab professionals do mission critical work every day. Thank you.

But there's a glaring gap in the current approach: patients and medical cannabis (MMJ).
While the public sees headlines about arrests and seizures, legitimate MMJ BioPharma Cultivation research and manufacturing are still waiting in line . Administrative delays and a broken Administrative Law system/registration process are holding back the one thing that can move cannabis out of the shadows and into modern medicine: clinical grade supply and FDA quality trial evidence .
Not Either/Or: Enforcement and Evidence
This is not an either/or proposition. The DEA can maintain vigorous enforcement against illicit fentanyl while simultaneously building a lawful, research grade cannabinoid supply chain that makes the black market less relevant. In fact, the best way to shrink the illicit sector is to scale the legitimate one -with Good Manufacturing Practice (GMP) standards and peer reviewed clinical evidence.
The FDA has already shown openness to cannabis derived medicines approving Epidiolex (cannabidiol) for seizure disorders and encouraging sound scientific research into medicinal uses. DEA's timely partnership is the missing link that turns scientific intent into actual trials, data, labels, and covered care.
What Patients and Researchers Are Saying
The frustration is palpable across the research community: too many applications sit on desks; investigators remain in an opaque queue with no timeline, no status, and no recourse. The result: trials delayed, data delayed, patient access delayed .
As Duane Boise , CEO of MMJ International Holdings, put it:
"We have followed every rule, spent millions building a compliant facility, and still the DEA has done everything in its power to delay and deny this life-saving research. This isn't about cannabis-it's about control, corruption, and constitutional failure."
You may disagree with the characterization-but you cannot ignore the bottleneck. And you don't need a new law to fix it.
The Bottleneck You Control-Today
Even with Congress constraining DOJ on rescheduling, DEA still has full authority right now to:
Grant Schedule I research registrations to qualified investigators and health systems
Approve bulk manufacturing (API) registrations for cannabinoid materials that supply clinical trials
Set quotas that match real research demand
Streamline amendments for protocol changes, sites, and dosage forms
The Registration Process Is Failing Stakeholders
No firm clocks : Months stretch into quarters; quarters into years
Opaque status : Applicants can't see where they are in the queue or what's missing
Duplicative reviews : Serial, not parallel, checks across agencies
ALJ lag : Timelines are elastic; outcomes arrive after grant windows and capital plans collapse
That machinery-built for a different era-does not serve a rapidly evolving research field with real public-health upside.
Six Actions DEA Can Take in 90 Days (No New Law Required)
Sign the stack : Immediately adjudicate pending MMJ BioPharma Cultivation API bulk manufacturing applications that meet security, GMP, and recordkeeping requirements. Use conditional approvals with remedial plans instead of open ended holds.
Publish a timeline & dashboard :
30-day administrative completeness check
90-day target for standard research registrations
Real-time, applicant-visible status and deficiency notices
Create a "Clinical Fast Lane" for FDA-aligned protocols: parallel reviews, a single point of contact, and quota setting matched to trial enrollment.
Issue clear GMP guidance for cannabinoid API: reference methods, stability, chain-of-custody, and validation packages-so applicants submit it right the first time.
Right-size quotas for research: adopt rolling, data-driven adjustments keyed to active INDs and enrollment curves, not calendar-year guesswork.
Stand up a public-private working group (DEA/FDA/NIH + investigators, sponsors, CROs, and patient orgs) to meet monthly and clear bottlenecks in real time.
What "Good" Looks Like by Year's End
New or expanded research registrations approved across major health systems and universities
Multiple API manufacturers producing GMP cannabinoid materials for Phase 2/3 trials
Quota policy aligned to real protocols and enrollment, not paperwork inertia
A public dashboard showing median review times, approvals, and trial-support metrics
The first wave of multicenter trials launched (oncology symptoms, chronic pain, PTSD, spasticity, sleep)
A Respectful Ask
Administrator Cole: your posts highlight enforcement, partnerships, and saving American lives . Approving research and API manufacturing for MMJ is exactly that. It saves lives by replacing conjecture with clinical data, stigma with standards, and street products with regulated, tested medicine .
Please direct your team to fix the bottleneck :
Put firm clocks on reviews
Communicate status transparently
Approve qualified manufacturers and investigators now
Celebrate the agents seizing poison and the scientists preparing medicine. America needs both. Patients are waiting. Let's get this done.
MMJ is represented by attorney Megan Sheehan .
CONTACT:
Madison Hisey
[email protected]
203-231-85832
SOURCE: MMJ International Holdings
View the original press release on ACCESS Newswire
J.Williams--AMWN
