
-
 Hollingsworth upsets Hunter Bell as Gout Gout fails to fire in Melbourne
Hollingsworth upsets Hunter Bell as Gout Gout fails to fire in Melbourne
-
Iran footballers pay tribute to victims of school strike

-
 Questions over Israel's interceptor stockpiles as Mideast war drags on
Questions over Israel's interceptor stockpiles as Mideast war drags on
-
Sweet heist? Nestle says 12 tonnes of KitKat stolen

-
 Pope denounces widening gap between the rich and poor on Monaco visit
Pope denounces widening gap between the rich and poor on Monaco visit
-
Yemen's Houthi enter war with missile targeting Israel

-
 USS Gerald Ford arrives in Croatia for maintenance
USS Gerald Ford arrives in Croatia for maintenance
-
Antonelli leads Mercedes 1-2 as Verstappen suffers qualifying shock

-
 Verstappen calls his Red Bull 'undriveable' after more woes
Verstappen calls his Red Bull 'undriveable' after more woes
-
Antonelli takes pole for Japanese Grand Prix in Mercedes 1-2

-
 Millions angry with Trump expected to fill American streets
Millions angry with Trump expected to fill American streets
-
Attacks across Middle East as Iran war enters second month

-
 Late surge lifts Thunder, Celtics rally to down Hawks
Late surge lifts Thunder, Celtics rally to down Hawks
-
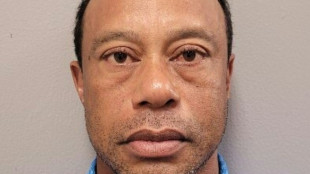
Tiger Woods arrested, charged with DUI after Florida crash
-
 Antonelli leads Mercedes one-two in final Japan practice
Antonelli leads Mercedes one-two in final Japan practice
-
Unease for Iranian-Canadians after shooting at ayatollah critic's gym

-
 Sequins, slogans, conspiracies: Inside the right-wing culture at CPAC
Sequins, slogans, conspiracies: Inside the right-wing culture at CPAC
-
NBA fines T-Wolves center Reid $50,000 for ripping refs

-
 Sinner ousts Zverev to book Miami Open final with Lehecka
Sinner ousts Zverev to book Miami Open final with Lehecka
-
McKellar hails 'special memory' after Waratahs stun Brumbies

-
 Best Crypto Roth IRA Company in the US Announced (2026 Update)
Best Crypto Roth IRA Company in the US Announced (2026 Update)
-
Tuchel takes positives from scrappy England draw against Uruguay

-
 Japanese star Sakamoto signs off with fourth world skating gold
Japanese star Sakamoto signs off with fourth world skating gold
-
Tuchel disappointed after England fans boo White

-
 US envoy hopeful on Iran talks as strikes target nuclear facilities
US envoy hopeful on Iran talks as strikes target nuclear facilities
-
Controversial African champions Morocco salvage Ecuador draw on Ouahbi debut

-
 Dutch end Norway's unbeaten run as Haaland rests
Dutch end Norway's unbeaten run as Haaland rests
-
'Strait of Trump': US president says Iran must open key waterway

-
 Wirtz steals show as Germany win thriller in Switzerland
Wirtz steals show as Germany win thriller in Switzerland
-
White jeered on England return as Uruguay snatch friendly draw

-
 Tiger Woods arrested, charged with DUI after Florida crash: police
Tiger Woods arrested, charged with DUI after Florida crash: police
-
Oyarzabal double fires Spain to win over Serbia

-
 More to IOC gender testing than appeasing Trump: ex-IOC executive
More to IOC gender testing than appeasing Trump: ex-IOC executive
-
Japan's Sakamoto ends career with fourth world skating title

-
 'Whatever it takes' - Sabalenka faces Gauff for second straight Miami Open crown
'Whatever it takes' - Sabalenka faces Gauff for second straight Miami Open crown
-
US hopes for Iran meetings 'this week': envoy Witkoff

-
 Uncertainty over war-induced oil crisis dominates key energy summit
Uncertainty over war-induced oil crisis dominates key energy summit
-
Czech Lehecka beats France's Fils to reach Miami Open final

-
 No pressure? Pochettino urges US co-hosts to 'play free' at World Cup
No pressure? Pochettino urges US co-hosts to 'play free' at World Cup
-
Duckett eager to show hunger for England success after Ashes flop

-
 'We are ready': astronauts arrive at launch site for Moon mission
'We are ready': astronauts arrive at launch site for Moon mission
-
Fishy trades before major news spark insider trading allegations

-
 Tiger Woods involved in Florida car crash: reports
Tiger Woods involved in Florida car crash: reports
-
WTO reform talks coming to the crunch

-
 Renaissance master Raphael honored at New York's Met museum
Renaissance master Raphael honored at New York's Met museum
-
At 'Davos of energy', AI looks to gas to power its rapid expansion

-
 Israel hits Iran nuclear sites as Washington trails end to war
Israel hits Iran nuclear sites as Washington trails end to war
-
US court overturns $16.1 bn judgment against Argentina over oil firm seizure

-
 England quick Tongue backs Cooley to make him a better bowler
England quick Tongue backs Cooley to make him a better bowler
-
Stand at new Inter Miami stadium to be named for Messi


Jaguar Health Submits Orphan Drug Designation Application to FDA for Crofelemer for Cancer Therapy-Related Diarrhea (CTD) in Patients with Breast Cancer with Metastasis Identified by FDA as a Distinct Condition
Potential treatment for patients with breast cancer with metastasis to the brain with crofelemer would augment the significant responder analysis results for crofelemer in the prespecified subgroup of patients with breast cancer from the OnTarget study that were reported at the Multinational Association of Supportive Care in Cancer 2025 Annual Meeting and the 2024 San Antonio Breast Cancer Symposium
Potential treatment for patients with breast cancer with metastasis to the brain with crofelemer would augment the significant responder analysis results for crofelemer in the prespecified subgroup of patients with breast cancer from the OnTarget study that were reported at the Multinational Association of Supportive Care in Cancer 2025 Annual Meeting and the 2024 San Antonio Breast Cancer Symposium
SAN FRANCISCO, CA / ACCESS Newswire / September 24, 2025 / Jaguar Health (NASDAQ:JAGX) today announced that Jaguar family company Napo Pharmaceuticals (Napo) has submitted an orphan drug designation (ODD) application to the U.S. Food and Drug Administration (FDA) for crofelemer, the company's novel prescription drug, for the treatment of diarrhea in adult patients with breast cancer that has metastasized to the brain receiving targeted therapy with or without standard chemotherapy.
Within the last ten years, the FDA's Office of Orphan Products Development (OOPD) has publicly acknowledged that brain metastasis of any cancer is considered a disease or condition separate and distinct from the primary site of origin. Accordingly, between 2015 and 2024 the OOPD awarded seven orphan drug designations for various therapies for the treatment of breast cancer that has metastasized to the brain.
"The FDA has identified the condition of breast cancer that has metastasized to the brain as an orphan indication. We have submitted this ODD application for crofelemer to treat diarrhea in these patients," said Lisa Conte, Jaguar's founder, president, and CEO. "Diarrhea is a common side effect of numerous targeted cancer therapies that can lead to dose changes, treatment delays, or cessation of treatment altogether, all of which impact patient outcomes. Given crofelemer's novel and physiological mechanism of action, we would plan to seek Breakthrough Therapy designation and/or Fast Track designation from the FDA to support potentially expedited regulatory pathways in the U.S. for crofelemer for this indication."
In both the US and European Union, crofelemer has previously received orphan drug designation for short bowel syndrome with intestinal failure and microvillus inclusion disease for a liquid formulation of crofelemer. Crofelemer has also been granted an orphan drug designation for treatment of diarrhea in cholera in the U.S. Orphan drug designation qualifies the drug sponsor for various development and/or regulatory incentives, such as tax credits for qualified clinical testing and relief from filing fees. Futhermore, orphan drug designation in the U.S. provides a seven-year period of marketing exclusivity to the first sponsor.
About Crofelemer
Crofelemer is a botanical (plant-based) drug extracted and purified from the red bark sap, also referred to as "dragon's blood," of the medicinal Croton lechleri tree in the Amazon Rainforest. Jaguar family company Napo Pharmaceuticals has established a sustainable harvesting program, under fair trade practices, for crofelemer to ensure a high degree of quality, ecological integrity, and support for indigenous communities.
About the Jaguar Health Family of Companies
Jaguar Health, Inc. (Jaguar) is a commercial stage pharmaceuticals company focused on developing novel proprietary prescription medicines sustainably derived from plants from rainforest areas for people and animals with gastrointestinal distress, specifically associated with overactive bowel, which includes symptoms such as chronic debilitating diarrhea, urgency, bowel incontinence, and cramping pain. Jaguar family company Napo Pharmaceuticals (Napo) focuses on developing and commercializing human prescription pharmaceuticals for essential supportive care and management of neglected gastrointestinal symptoms across multiple complicated disease states. Jaguar family company Napo Therapeutics is an Italian corporation Jaguar established in Milan, Italy in 2021 focused on expanding crofelemer access in Europe and specifically for orphan diseases.
For more information about:
Jaguar Health, visit https://jaguar.health
Napo Pharmaceuticals, visit www.napopharma.com
Napo Therapeutics, visit napotherapeutics.com
Visit the Make Cancer Less Shitty patient advocacy program on Bluesky, X, Facebook & Instagram
Forward-Looking Statements
Certain statements in this press release constitute "forward-looking statements." These include statements regarding Jaguar's expectation that Napo may seek Breakthrough Therapy designation and/or Fast Track designation from the FDA to support potentially expedited regulatory pathways in the U.S. for crofelemer for the treatment of diarrhea in adult patients with breast cancer that has metastasized to the brain. In some cases, you can identify forward-looking statements by terms such as "may," "will," "should," "expect," "plan," "aim," "anticipate," "could," "intend," "target," "project," "contemplate," "believe," "estimate," "predict," "potential" or "continue" or the negative of these terms or other similar expressions. The forward-looking statements in this release are only predictions. Jaguar has based these forward-looking statements largely on its current expectations and projections about future events. These forward-looking statements speak only as of the date of this release and are subject to a number of risks, uncertainties and assumptions, some of which cannot be predicted or quantified and some of which are beyond Jaguar's control. Except as required by applicable law, Jaguar does not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
CONTACT:
[email protected]
Jaguar-JAGX
SOURCE: Jaguar Health, Inc.
View the original press release on ACCESS Newswire
G.Stevens--AMWN
