
-
 Antonelli takes pole for Japanese Grand Prix in Mercedes 1-2
Antonelli takes pole for Japanese Grand Prix in Mercedes 1-2
-
Millions angry with Trump expected to fill American streets

-
 Attacks across Middle East as Iran war enters second month
Attacks across Middle East as Iran war enters second month
-
Late surge lifts Thunder, Celtics rally to down Hawks

-
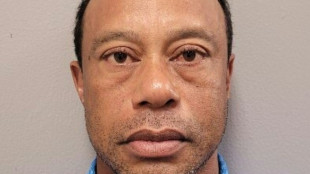 Tiger Woods arrested, charged with DUI after Florida crash
Tiger Woods arrested, charged with DUI after Florida crash
-
Antonelli leads Mercedes one-two in final Japan practice

-
 Unease for Iranian-Canadians after shooting at ayatollah critic's gym
Unease for Iranian-Canadians after shooting at ayatollah critic's gym
-
Sequins, slogans, conspiracies: Inside the right-wing culture at CPAC

-
 NBA fines T-Wolves center Reid $50,000 for ripping refs
NBA fines T-Wolves center Reid $50,000 for ripping refs
-
Sinner ousts Zverev to book Miami Open final with Lehecka

-
 McKellar hails 'special memory' after Waratahs stun Brumbies
McKellar hails 'special memory' after Waratahs stun Brumbies
-
Tuchel takes positives from scrappy England draw against Uruguay

-
 Japanese star Sakamoto signs off with fourth world skating gold
Japanese star Sakamoto signs off with fourth world skating gold
-
Tuchel disappointed after England fans boo White

-
 US envoy hopeful on Iran talks as strikes target nuclear facilities
US envoy hopeful on Iran talks as strikes target nuclear facilities
-
Controversial African champions Morocco salvage Ecuador draw on Ouahbi debut

-
 Dutch end Norway's unbeaten run as Haaland rests
Dutch end Norway's unbeaten run as Haaland rests
-
'Strait of Trump': US president says Iran must open key waterway

-
 Wirtz steals show as Germany win thriller in Switzerland
Wirtz steals show as Germany win thriller in Switzerland
-
White jeered on England return as Uruguay snatch friendly draw

-
 Tiger Woods arrested, charged with DUI after Florida crash: police
Tiger Woods arrested, charged with DUI after Florida crash: police
-
Oyarzabal double fires Spain to win over Serbia

-
 More to IOC gender testing than appeasing Trump: ex-IOC executive
More to IOC gender testing than appeasing Trump: ex-IOC executive
-
Japan's Sakamoto ends career with fourth world skating title

-
 'Whatever it takes' - Sabalenka faces Gauff for second straight Miami Open crown
'Whatever it takes' - Sabalenka faces Gauff for second straight Miami Open crown
-
US hopes for Iran meetings 'this week': envoy Witkoff

-
 Uncertainty over war-induced oil crisis dominates key energy summit
Uncertainty over war-induced oil crisis dominates key energy summit
-
Czech Lehecka beats France's Fils to reach Miami Open final

-
 No pressure? Pochettino urges US co-hosts to 'play free' at World Cup
No pressure? Pochettino urges US co-hosts to 'play free' at World Cup
-
Duckett eager to show hunger for England success after Ashes flop

-
 'We are ready': astronauts arrive at launch site for Moon mission
'We are ready': astronauts arrive at launch site for Moon mission
-
Fishy trades before major news spark insider trading allegations

-
 Tiger Woods involved in Florida car crash: reports
Tiger Woods involved in Florida car crash: reports
-
WTO reform talks coming to the crunch

-
 Renaissance master Raphael honored at New York's Met museum
Renaissance master Raphael honored at New York's Met museum
-
At 'Davos of energy', AI looks to gas to power its rapid expansion

-
 Israel hits Iran nuclear sites as Washington trails end to war
Israel hits Iran nuclear sites as Washington trails end to war
-
US court overturns $16.1 bn judgment against Argentina over oil firm seizure

-
 England quick Tongue backs Cooley to make him a better bowler
England quick Tongue backs Cooley to make him a better bowler
-
Stand at new Inter Miami stadium to be named for Messi

-
 G7 urges end to attacks on civilians in Middle East war
G7 urges end to attacks on civilians in Middle East war
-
Mideast war leaves 6,000 tonnes of tea stuck at Kenya port

-
 US and Israel hit nuclear sites as Rubio trails end to Iran war
US and Israel hit nuclear sites as Rubio trails end to Iran war
-
Van der Poel holds on for third straight E3 Classic victory

-
 Missing aid boats 'safely' crossed to Cuba: US Coast Guard
Missing aid boats 'safely' crossed to Cuba: US Coast Guard
-
'Everyone knows we are African champions', insists Senegal coach

-
 China used fake LinkedIn profiles to spy on NATO, EU: security source
China used fake LinkedIn profiles to spy on NATO, EU: security source
-
Djokovic withdraws from Monte-Carlo Masters

-
 English rugby chief says no talks with Farrell 'at present'
English rugby chief says no talks with Farrell 'at present'
-
G7 ministers urge end to attacks against civilians in Mideast war


Pentixapharm Announces Advancement of PentixaTher to Fourth Dose Level in Acute Myeloid Leukemia Trial
Achieves Key Milestone with CXCR4-based Radiotherapeutic
Favorable safety profile enables advancement of radiolabeled PentixaTher to fourth of five planned activity dose levels in investigator-initiated PENTILULA Phase 1/2 trial
Milestone marks important step for novel radiotherapeutic candidate in high unmet need indication with limited treatment options and poor prognosis
Achieves Key Milestone with CXCR4-based Radiotherapeutic
Favorable safety profile enables advancement of radiolabeled PentixaTher to fourth of five planned activity dose levels in investigator-initiated PENTILULA Phase 1/2 trial
Milestone marks important step for novel radiotherapeutic candidate in high unmet need indication with limited treatment options and poor prognosis
BERLIN, DE / ACCESS Newswire / September 26, 2025 / Pentixapharm AG (Frankfurt Prime Standard:PTP), an advanced clinical-stage biotech, developing novel radiopharmaceuticals, today announced that its CXCR4-targeting radiolabeled candidate PentixaTher has advanced to the fourth dose level in the investigator-initiated PENTILULA Phase 1/2 study in acute myeloid leukemia (AML).
Based on favorable safety findings at the third dose level of 7.5 GBq, the investigator-initiated trial received approval from the independent Data Safety Monitoring Board (DSMB) to advance to the fourth of five planned dose levels at 10 GBq, underscoring PentixaTher's tolerability to date. Importantly, this advancement not only reinforces the compound's safety profile but also moves the study into a dose range with a higher likelihood for meaningful clinical efficacy. If favorable safety signals continue, the study is expected to escalate to a fifth and final dose level of 12.5 GBq.
AML is the most common acute leukemia in adults, with approximately 20,000 new cases annually in the U.S. and 17,000 new cases annually in Europe. Five-year survival rates range between approximately 32 and 37%, respectively.
"Advancing to higher dose levels marks a key step in validating the therapeutic potential and promise of PentixaTher," said Dirk Pleimes, MD, CEO/CMO of Pentixapharm "The encouraging results in this investor-led effort highlight the potential of CXCR4 as a powerful target for hematologic cancers and demonstrate the promise of radiopharmaceuticals to expand treatment options where medical need is greatest. The trial is expanding the clinical evidence base for CXCR4-targeted therapy to expand the boundaries of radiopharmaceuticals."
Professor Françoise Kraeber-Bodéré, Nuclear Medicine Department, CHU Nantes, Principal Investigator, commented: "We are encouraged by the favorable safety profile observed to date, which has enabled us to move to higher activity dose levels. At dose level four, we are now entering a range considered of potentially higher effectiveness, and we look forward to evaluating the clinical impact in this high-need patient population."
Professor Patrice Chevallier, Hematology Department, CHU Nantes added: "For patients with advanced AML, treatment options are limited, and prognosis in this often heavily pretreated population is poor. Reaching this dose level with an acceptable safety profile provides new hope that CXCR4-targeted radiopharmaceuticals could one day become a meaningful addition to the therapeutic landscape."
About PentixaTher and the PENTILULA Phase 1/2 Study
Radiolabeled PentixaTher is a novel radiotherapeutic designed to selectively target the chemokine receptor CXCR4, a key player in the bone marrow microenvironment that is frequently overexpressed in aggressive hematological malignancies. The compound is labeled for the PENTILULA study with 177-lutetium, a clinically well-established isotope, enabling precise delivery of a targeted radiation payload. PentixaTher is currently being evaluated in the PENTILULA Phase 1/2 study (ClinicalTrials.gov ID: NCT06356922 ), initiated in November 2024 . The multicenter, open-label, dose-escalation trial is led by an experienced investigator team at the University Hospital of Nantes and conducted at three additional clinical sites in France. It aims primarily to assess the safety and tolerability of PentixaTher, with secondary objectives including preliminary measures of clinical activity such as overall response rate, complete response rate, and overall survival. The study is supported by the French Ministry of Health.
About Pentixapharm
Pentixapharm is an advanced clinical-stage biotech expanding the boundaries of radiopharmaceuticals. Headquartered in Berlin, Germany, the company develops first-in-class ligand- and antibody-based radiopharmaceuticals designed to transform patient care across oncology and beyond. Its late-stage pipeline is anchored by CXCR4-targeted programs, including a Phase 3-ready diagnostic candidate for primary aldosteronism and pioneering therapeutic programs in a number of hematological and solid cancers. Furthermore, Pentixapharm is advancing a next-generation antibody platform targeting CD24, an emerging immune-escape marker over-expressed in multiple hard-to-treat cancers. Complemented by reliable isotope supply from Eckert & Ziegler, and a robust global clinical network, Pentixapharm is uniquely positioned to deliver innovative radiopharmaceuticals that address high unmet need, improve patient outcomes, and create significant growth opportunities in one of the fastest-growing areas of precision medicine.
About Nantes University Hospital
Nantes University Hospital (CHU de Nantes) is one of France's leading healthcare institutions, recognized for its excellence in various medical specialties including cardiology, transplants, oncology, and neuroscience. The hospital is also a key player in medical research and innovation, conducting clinical trials and contributing to significant advancements in healthcare, in particular in nuclear medicine and hematology. [ 177 Lu]Lu-PentixaTher is produced by ARRONAX, the hospital radiopharmacy unit (APUI), which is specially authorized for the preparation of the radiopharmaceutical investigational product.
Pentixapharm Investor and Media Contact
SOURCE: Pentixapharm Holding AG
View the original press release on ACCESS Newswire
F.Schneider--AMWN

