
-
 Iran defies Trump Hormuz ultimatum with naval mine threat
Iran defies Trump Hormuz ultimatum with naval mine threat
-
African players in Europe: Awoniyi seals key win for lowly Forest

-
 France ex-PM Lionel Jospin dies aged 88
France ex-PM Lionel Jospin dies aged 88
-
Runway collision kills two pilots, shutters New York airport

-
 Hodgkinson in 'shape of her life' with eye on Kratochvilova's record
Hodgkinson in 'shape of her life' with eye on Kratochvilova's record
-
Griezmann given go-ahead to talk with Orlando City

-
 Mideast war threatens energy crisis worse than 1970s oil shocks
Mideast war threatens energy crisis worse than 1970s oil shocks
-
Pilot, co-pilot killed in runway collision at New York airport

-
 Plane, fire truck collide on runway at New York's LaGuardia Airport
Plane, fire truck collide on runway at New York's LaGuardia Airport
-
Russia's Max: The unencrypted super-app being forced on citizens

-
 EU chief in Australia with eyes on trade deal
EU chief in Australia with eyes on trade deal
-
Asia champions Japan need 'different tools' to win World Cup - coach

-
 Global economy under 'major threat' from Strait of Hormuz crisis: IEA chief
Global economy under 'major threat' from Strait of Hormuz crisis: IEA chief
-
Planet trapped record heat in 2025: UN

-
 Israel launches new strikes on Tehran as Iran takes aim at Gulf sites
Israel launches new strikes on Tehran as Iran takes aim at Gulf sites
-
German court to rule in climate case against automakers

-
 France's leftists win mayoral elections in largest cities
France's leftists win mayoral elections in largest cities
-
Asian stocks tumble as Trump gives Iran 48-hour ultimatum

-
 Wolves rally past Celtics, Nuggets sink Blazers
Wolves rally past Celtics, Nuggets sink Blazers
-
Middle East war to dominate Houston's 'Davos of Energy'

-
 Kim holds off Korda charge to win LPGA Founders Cup
Kim holds off Korda charge to win LPGA Founders Cup
-
Trump orders immigration agents to airports amid crippling budget standoff

-
 GAT Guns Delivers Standout Results at Factory Shoot 2026 with Champion Safe
GAT Guns Delivers Standout Results at Factory Shoot 2026 with Champion Safe
-
Digi Power X to Announce 2025 Year End Financial Results on March 31st

-
 Sinopec Announced 2025 Annual Results Annual Payout Ratio Reached 81%
Sinopec Announced 2025 Annual Results Annual Payout Ratio Reached 81%
-
Guanajuato Silver Adds Veteran Mine Operator to Board of Directors

-
 Snipp Interactive Secures US$3 Million Contract, Largest in Company History
Snipp Interactive Secures US$3 Million Contract, Largest in Company History
-
Atcom Recognized with 2026 Consumer Choice Award for Telephone Systems Sales & Service in Southern Alberta

-
 Announcing the 2026 Niagara Consumer Choice Award Winners
Announcing the 2026 Niagara Consumer Choice Award Winners
-
TaxBandits Partners with Protection Plus to Provide 940 and 941 Filers with $1 Million in Tax Protection(TM)

-
 TK Elevator Appoints Brad Selleck as Chief Operating Officer; Vincent Della Valle to Retire
TK Elevator Appoints Brad Selleck as Chief Operating Officer; Vincent Della Valle to Retire
-
TK Elevator Creates the Role of Chief Legal Officer, Appoints Lars Sjobring

-
 The Great Repricing of Plastic: How Recycling is Moving from ESG Narrative to Economic Reality
The Great Repricing of Plastic: How Recycling is Moving from ESG Narrative to Economic Reality
-
InterContinental Hotels Group PLC Announces Transaction in Own Shares - March 23

-
 Just - Evotec Biologics Enters Project Agreement with BARDA to Optimize Biomanufacturing of Antibodies Against Ebola and Related Viruses
Just - Evotec Biologics Enters Project Agreement with BARDA to Optimize Biomanufacturing of Antibodies Against Ebola and Related Viruses
-
ABRIDGED, PRELIMINARY AND UNAUDITED QUARTERLY AND FULL YEAR RESULTS AND DETAILS OF MANAGEMENT CONFERENCE Call for the Fourth Quarter and Full Year Ended December 31, 2025 ("The Quarter" or "Q4 2025" and "FY 2025" Respectively)

-
 Iran awaits Trump threat to blow up power plants
Iran awaits Trump threat to blow up power plants
-
Alcaraz eyes clay court season after early Miami exit

-
 Real Madrid down Atletico in derby, leaders Barca edge Rayo
Real Madrid down Atletico in derby, leaders Barca edge Rayo
-
Korda sends Alcaraz to another early exit in Miami

-
 Bordeaux-Begles hammer Toulouse in Dupont absence
Bordeaux-Begles hammer Toulouse in Dupont absence
-
Slovenia PM claims election win as results show neck and neck finish

-
 England's Fitzpatrick birdies 18th to win PGA Valspar title
England's Fitzpatrick birdies 18th to win PGA Valspar title
-
Man City's League Cup glory adds twist to title race

-
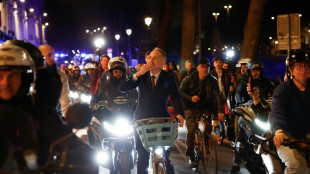 Leftists win mayoral elections in Paris and Marseille
Leftists win mayoral elections in Paris and Marseille
-
Vinicius double helps Real Madrid edge Atletico thriller

-
 Doncic cleared to face Pistons after foul rescinded: NBA
Doncic cleared to face Pistons after foul rescinded: NBA
-
Inter's Serie A lead cut to six with Fiorentina draw, Como march on

-
 World No.1 Alcaraz beaten by Korda in Miami Open third round
World No.1 Alcaraz beaten by Korda in Miami Open third round
-
Cuba starts to restore power after new blackout


Medicus Pharma Ltd. to Present at The Maxim Growth Summit 2025
EXECUTIVE CHAIRMAN & CEO OF MEDICUS TO PARTICIPATE IN A FIRESIDE CHAT WITH EQUITY RESEARCH ANALYST
PHILADELPHIA, PENNSYLVANIA / ACCESS Newswire / October 13, 2025 / Medicus Pharma Ltd. (NASDAQ:MDCX) ("Medicus" or the "Company"), a biotech/life sciences company focused on advancing the clinical development programs of novel and potentially disruptive therapeutics assets, is pleased to announce that it will present at the Maxim Growth Summit, being held October 22-23, 2025, in New York City.
This summit brings together industry leaders, innovators, thought leaders, opinion makers and premier institutions to explore the latest trends and advancements across several industries.
Event Details
Date: October 22-23, 2025
Location: Hard Rock Hotel, New York City
Dr. Raza Bokhari, Executive Chairman & CEO of Medicus, will deliver an overview of the Company in a Fireside Chat hosted by Maxim analyst, Dr. Jason McCarthy and provide an update on the clinical development program of its two therapeutic assets:
Skinject, a novel immuno-oncology precision product, focused on non-melanoma skin diseases, especially basal cell carcinoma (BCC) of the skin, representing ~$2 billion in potential market opportunity.
Teverelix, a next generation GnRH antagonist, as a first in market product for cardiovascular high-risk advanced prostate cancer patients and patients with acute urinary retention relapse (AURr) episodes due to enlarged prostate, collectively representing ~$6 billion in potential market opportunity.
A webcast of the Fireside Chat will be available on-demand to registered conference attendees. Dr. Bokhari will also meet with institutional investors registered for the conference in one-on-one format.
The conference will also feature roundtable discussions with CEOs from small and mid-cap companies, moderated by Maxim Research Analysts. Roundtable discussions will cover a range of sectors, including biotechnology, stem cell therapy, ophthalmology, artificial intelligence, and more.
For more information and a complete agenda of the Maxim Growth Summit, please visit www.maximgrp.com/2025-growth-summit.
Medicus is currently conducting a Phase 2 clinical study for SKNJCT-003 in nine (9) clinical sites across the United States which commenced randomizing patients in August 2024. SKNJCT-003 is a double blinded, placebo controlled triple arm proof of concept Phase 2 clinical study, designed to non-invasively treat basal cell carcinoma (BCC) of the skin using novel, patent protected, dissolvable Doxorubicin-containing microneedle arrays (D-MNA). In March 2025, the Company announced a positively trending interim analysis for SKNJCT-003 demonstrating more than 60% clinical clearance. The interim analysis was conducted after more than 50% of the then-targeted 60 patients in the study were randomized. The findings of the interim analysis are preliminary and may or may not correlate with the findings of the study once completed.In April 2025, the investigational review board approved to increase the number of participants in SKNJCT-003 to ninety (90) subjects. The Company is expanding its trial sites in Europe and has randomized more than 75% of the ninety (90) participants expected to be randomized in the study. In September 2025, the Company received positive feedback from the Food and Drug Administration (FDA) regarding its Type C meeting supporting the development of Skinject, indicating that the Company may follow 505(b)(2) regulatory pathway to non-invasively treat BCC using dissolvable D-MNA.
The Company also has a clinical study (SKNJCT-004) currently underway in the United Arab Emirates (UAE). The study is expected to randomize thirty-six (36) patients in six (6) sites in the UAE. Cleveland Clinic Abu Dhabi (CCAD) is the principal investigator, along with Sheikh Shakbout Medical City (SSMC), Burjeel Medical City (BMC), Rashid Hospital (RH), Clemenceau Medical Center (CMC) and American Hospital of Dubai (AHD). Insights Research Organization and Solutions (IROS), a UAE-based contract research organization, is coordinating the clinical study for the Company. IROS is a M42 portfolio company.
In August 2025, the Company completed the acquisition of Antev, a UK-based late clinical stage biotech company, developing Teverelix, a next generation GnRH antagonist, as a first in market product for cardiovascular high-risk advanced prostate cancer patients and patients with first acute urinary retention relapse (AURr) episodes due to enlarged prostate.
Antev's flagship drug candidate is Teverelix trifluoroacetate (Teverelix TFA), a long-acting gonadotrophin-releasing hormone (GnRH) antagonist. Unlike GnRH agonists, which can cause an initial surge in testosterone levels, Teverelix directly suppresses sex hormone production without this surge, potentially reducing cardiovascular risks. This mechanism is particularly beneficial for patients with existing cardiovascular conditions. Teverelix is formulated as a microcrystalline suspension, allowing for sustained release and a six-week dosing interval, which may improve patient compliance and outcomes.
For further information contact:
Carolyn Bonner, President and Acting Chief Financial Officer
(610) 636-0184
[email protected]
Anna Baran-Djokovic, SVP Investor Relations
(305) 615-9162
[email protected]
About Medicus Pharma Ltd.
Medicus Pharma Ltd. (Nasdaq: MDCX) is a biotech/life sciences company focused on accelerating the clinical development programs of novel and potentially disruptive therapeutics assets. The Company is actively engaged in multiple countries, spread over three continents.
SkinJect Inc. a wholly owned subsidiary of Medicus Pharma Ltd., is a development stage, life sciences company focused on commercializing novel, non-invasive treatment for basal cell skin cancer using a patented dissolvable microneedle patch to deliver a chemotherapeutic agent to eradicate tumors cells. The Company completed a phase 1 safety & tolerability study (SKNJCT-001) in March of 2021, which met its primary objective of safety and tolerability; the study also describes the efficacy of the investigational product D-MNA, with six (6) participants experiencing complete response on histological examination of the resected lesion. The Company is currently conducting a randomized, controlled, double-blind, multicenter clinical study (SKNJCT-003) in the United States and Europe. The Company has also commenced a randomized, controlled, double-blind, multicenter clinical study (SKNJCT-004) in the United Arab Emirates.
In August 2025, the Company announced its entry into a non-binding memorandum of understanding (the "MoU") with Helix Nanotechnologies, Inc. ("HelixNano"), a Boston Based biotech company focused on developing a proprietary advanced mRNA platform, in respect of their shared mutual interest in the development or commercial arrangement contemplated by the MoU. The MoU is non-binding and shall not be construed to obligate either party to proceed with a joint venture or any further development or commercial arrangement, unless and until definitive agreements are executed.
In August 2025, the Company completed the acquisition of Antev, a UK-based late clinical stage biotech company, developing Teverelix, a next generation GnRH antagonist, as a first in market product for cardiovascular high-risk advanced prostate cancer patients and patients with first acute urinary retention relapse (AURr) episodes due to enlarged prostate.
Antev's flagship drug candidate is Teverelix trifluoroacetate (Teverelix TFA), a long-acting gonadotrophin-releasing hormone (GnRH) antagonist. Unlike GnRH agonists, which can cause an initial surge in testosterone levels, Teverelix directly suppresses sex hormone production without this surge, potentially reducing cardiovascular risks. This mechanism is particularly beneficial for patients with existing cardiovascular conditions. Teverelix is formulated as a microcrystalline suspension, allowing for sustained release and a six-week dosing interval, which may improve patient compliance and outcomes
In September 2020, Antev completed a Phase 1 clinical trial in which Teverelix was shown to be well tolerated with no dose-limiting toxicities and demonstrated rapid testosterone suppression. The study included 48 healthy male volunteers. In February 2023, Antev also completed a Phase 2a study in fifty (50) patients with advanced prostate cancer (APC), where Teverelix achieved the primary endpoint of greater than 90% probability of castration levels of testosterone suppression (97.5%) but the secondary endpoint of maintaining this rate above 90% was not met with the probability dropping to 82.5% by Day 42.
In January 2023, the FDA, reviewed the Phase 1 and Phase 2a data and provided written guidance on Antev's proposed Phase 3 trial design for Teverelix. This milestone supports the Company's clinical plans to develop Teverelix as a treatment for advanced prostate cancer patients with increased cardiovascular risk.
In December 2023, FDA approved the Phase 2b study design in advanced prostate cancer covering 40 patients.
In November 2024, FDA approved the Phase 2b study design in acute urinary retention covering 390 patients.
Cautionary Notice on Forward-Looking Statements
Certain information in this news release constitutes "forward-looking information" under applicable securities laws. "Forward-looking information" is defined as disclosure regarding possible events, conditions or financial performance that is based on assumptions about future economic conditions and courses of action and includes, without limitation, the development of Teverelix and expectations concerning, and future outcomes relating to, the development, advancement and commercialization of Teverelix for AURr and high CV risk prostate cancer, and the potential market opportunities related thereto, the MOU, including the potential signing of definitive agreements between Medicus and HelixNano and the development of thermostable infectious diseases vaccines by combining HelixNano's proprietary mRNA vaccine platform with Medicus's proprietary microneedle array (MNA) delivery platform, the Company's aim to fast-track the clinical development program and convert the SKNJCT-003 exploratory clinical trial into a pivotal clinical trial, and approval from the FDA and the timing thereof, plans and expectations concerning, and future outcomes relating to, the development, advancement and commercialization of SkinJect through SKNJCT-003 and SKNJCT-004, and the potential market opportunities related thereto, the commencement of the SKNJCT-004 study and the potential results of and benefits of such study. Forward-looking statements are often but not always, identified by the use of such terms as "may", "on track", "aim", "might", "will", "will likely result", "could," "designed," "would", "should", "estimate", "plan", "project", "forecast", "intend", "expect", "anticipate", "believe", "seek", "continue", "target", "potential" or the negative and/or inverse of such terms or other similar expressions. These statements involve known and unknown risks, uncertainties and other factors, which may cause actual results, performance or achievements to differ materially from those expressed or implied by such statements, including those risk factors described in the Company's annual report on form 10-K for the year ended December 31, 2024 (the "Annual Report"), and in the Company's other public filings on EDGAR and SEDAR+, which may impact, among other things, the trading price and liquidity of the Company's common shares. Forward-looking statements contained in this news release are expressly qualified by this cautionary statement and reflect our expectations as of the date hereof and thus are subject to change thereafter. The Company disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. Readers are further cautioned not to place undue reliance on forward-looking statements as there can be no assurance that the plans, intentions or expectations upon which they are placed will occur. Such information, although considered reasonable by management at the time of preparation, may prove to be incorrect and actual results may differ materially from those anticipated.
SOURCE: Medicus Pharma Ltd
View the original press release on ACCESS Newswire
O.Johnson--AMWN
