
-
 Souped-up VPNs play 'cat and mouse' game with Iran censors
Souped-up VPNs play 'cat and mouse' game with Iran censors
-
Attacked Russian tanker drifting toward Libya: Italian authorities

-
 Coroner 'not satisfied' boxer Hatton intended to take own life
Coroner 'not satisfied' boxer Hatton intended to take own life
-
Stocks drop, as oil rises as Mideast war persists

-
 Vanishing glacier on Germany's highest peak prompts ski lift demolition
Vanishing glacier on Germany's highest peak prompts ski lift demolition
-
Chuck Norris, roundhouse-kicking action star, dead at 86: family

-
 Supreme leader says Iran dealt enemies 'dizzying blow'
Supreme leader says Iran dealt enemies 'dizzying blow'
-
Audi team principal Wheatley in shock exit after two races

-
 Spurs boss Tudor hopes for 'nice surprises' in relegation fight
Spurs boss Tudor hopes for 'nice surprises' in relegation fight
-
Arsenal must prove they are winners in League Cup final, says Arteta

-
 Record-breaking heat wave grips western US
Record-breaking heat wave grips western US
-
Liverpool showdown brings back 'beautiful memories' for PSG coach Luis Enrique

-
 IRA bomb victims drop civil court claim against Gerry Adams
IRA bomb victims drop civil court claim against Gerry Adams
-
Ntamack returns for Toulouse to face France rival Jalibert

-
 Trump calls NATO allies 'cowards' over Iran
Trump calls NATO allies 'cowards' over Iran
-
French jihadist jailed for life for Islamic State crimes against Yazidis

-
 Chuck Norris, action man who inspired endless memes, dead at 86: family
Chuck Norris, action man who inspired endless memes, dead at 86: family
-
Action movie star Chuck Norris has died: family statement

-
 England stars have 'last chance' to earn World Cup spots: Tuchel
England stars have 'last chance' to earn World Cup spots: Tuchel
-
League Cup final a 'big moment' for Man City, says Guardiola

-
 Injured Ronaldo misses Portugal World Cup friendlies
Injured Ronaldo misses Portugal World Cup friendlies
-
Liverpool condemn 'cowardly' racist abuse of Konate

-
 Far from war, global fuel frustrations mount
Far from war, global fuel frustrations mount
-
German auto exports to China plunged a third in 2025: study

-
 Coach Valverde to leave Bilbao at end of season
Coach Valverde to leave Bilbao at end of season
-
'Decimated'? The Iranian leaders killed in Israeli-US war

-
 Mistral chief calls for European AI levy to pay creatives
Mistral chief calls for European AI levy to pay creatives
-
Liverpool suffer Salah blow in chase for Champions League

-
 Mahuchikh soars to world indoor high jump gold, Hodgkinson cruises
Mahuchikh soars to world indoor high jump gold, Hodgkinson cruises
-
Spain include Joan Garcia as one of four new call-ups

-
 Salah ruled out of Liverpool's Brighton clash
Salah ruled out of Liverpool's Brighton clash
-
Ship crews ration food in Iran blockade: seafarers

-
 Kuwait refinery hit as Iran marks New Year under shadow of war
Kuwait refinery hit as Iran marks New Year under shadow of war
-
England recall Mainoo, Maguire for pre-World Cup matches

-
 Jerusalem's Muslims despair as war shuts Al-Aqsa Mosque for Eid
Jerusalem's Muslims despair as war shuts Al-Aqsa Mosque for Eid
-
'War has aged us': Lebanon's kids aren't alright

-
 Snooker great O'Sullivan makes history with highest-ever break
Snooker great O'Sullivan makes history with highest-ever break
-
Kuwait refinery hit as Iran says missile production 'no concern'

-
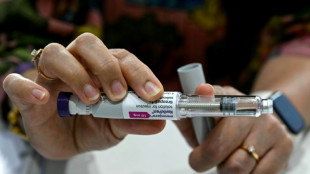 India to tackle global obesity with cheap fat-loss jabs
India to tackle global obesity with cheap fat-loss jabs
-
Somaliland centre saves cheetahs from trafficking to Gulf palaces

-
 China swim sensation Yu, 13, beats multiple Olympic medallist
China swim sensation Yu, 13, beats multiple Olympic medallist
-
North Korean leader, daughter try out new tank

-
 Israel strikes 'decimated' Iran as war roils markets
Israel strikes 'decimated' Iran as war roils markets
-
James ties NBA record for most regular-season games in latest milestone

-
 Trump's Mideast muddle could play into Xi's hands at planned summit
Trump's Mideast muddle could play into Xi's hands at planned summit
-
Wembanyama lifts playoff-bound Spurs, Doncic and James fuel Lakers

-
 Japan ski paradise faces strains of global acclaim
Japan ski paradise faces strains of global acclaim
-
Vinicius, Real Madrid must prove consistency in Atletico derby

-
 Kane credits Kompany's Bayern 'evolution' as treble beckons
Kane credits Kompany's Bayern 'evolution' as treble beckons
-
PSG look back to their best, but not yet out of sight in Ligue 1


Genflow Completes Dosing Phase of Canine Gene Therapy Trial
THE INFORMATION CONTAINED WITHIN THIS ANNOUNCEMENT IS DEEMED BY THE COMPANY TO CONSTITUTE INSIDE INFORMATION AS STIPULATED UNDER THE MARKET ABUSE REGULATION (EU) NO. 596/2014 AS IT FORMS PART OF UK DOMESTIC LAW PURSUANT TO THE EUROPEAN UNION (WITHDRAWAL) ACT 2018, AS AMENDED. UPON THE PUBLICATION OF THIS ANNOUNCEMENT VIA A REGULATORY INFORMATION SERVICE, THIS INFORMATION IS CONSIDERED TO BE IN THE PUBLIC DOMAIN.
Genflow Completes Dosing Phase of Canine Gene Therapy Trial - No Adverse Events Reported During Administration
LONDON, UK / ACCESS Newswire / December 16, 2025 /
Genflow Biosciences Plc (LSE:GENF)(OTCQB:GENFF) ("Genflow" or the "Company"), a biotechnology company focused on extending healthspan through advanced gene therapies, today announced that all dogs enrolled in its ongoing clinical trial have successfully received the full administration of the Company's investigational SIRT6-based gene therapy.
Importantly, no adverse events were reported during the dosing phase of the study. No serious, moderate, or minor side effects were observed, demonstrating a strong safety and tolerability profile for the therapy.
The study is being conducted as a blinded clinical trial and, as such, efficacy outcomes cannot yet be assessed. Genflow expects a first efficacy read in mid-to-late January 2026, which will include blood analyses and muscle biopsies to evaluate potential therapeutic benefits related to sarcopenia, overall healthspan, and lifespan-associated biomarkers.
The dogs enrolled in the trial will continue to be monitored for a total duration of 180 days. A second efficacy assessment is planned at the six-month timepoint, with results expected in June-July 2026, to evaluate durability and longer-term effects of the gene therapy.
Following analysis of the efficacy data, Genflow intends to resume discussions with several animal health companies, including parties currently engaged under confidentiality and data access agreements, to explore potential early-stage licensing opportunities.
Dr. Eric Leire, CEO of Genflow, commented: "These milestones represent an important step forward for our longevity program. The absence of any adverse events is highly encouraging and reinforces the strong safety profile of our approach. We look forward to sharing initial efficacy data in the coming months as the study progresses."
Contacts
Genflow Biosciences | Harbor Access |
Dr Eric Leire, CEO | Jonathan Paterson, Investor Relations |
+32-477-495-881 | +1 475 477 9401 |
About Genflow Biosciences
Founded in 2020, Genflow Biosciences Plc. (LSE:GENF)(OTCQB:GENFF), a biotechnology company headquartered in the UK with R&D facilities in Belgium, is pioneering gene therapies to decelerate the aging process, with the goal of promoting longer and healthier lives while mitigating the financial, emotional, and social impacts of a fast-growing aging global population. Genflow's lead compound, GF-1002, works through the delivery of a centenarian variant of the SIRT6 gene which has yielded promising preclinical results. Genflow's 12-month proof-of-concept clinical trial evaluating their SIRT6-centenarian gene therapy in aged dogs began in March 2025. Other programs, include a clinical trial that will explore the potential benefits of GF-1002 in treating MASH (Metabolic Dysfunction-Associated Steatohepatitis), the most prevalent chronic liver disease for which there is no effective treatments. Please visit www.genflowbio.com and follow the Company on LinkedIn and X.
DISCLAIMER
The contents of this announcement have been prepared by, and are the sole responsibility of, the Company.
This announcement may contain forward-looking statements. The forward-looking statements include, but are not limited to, statements regarding the Company's or the Directors' expectations, hopes, beliefs, intentions or strategies regarding the future. In addition, any statement that refers to projections, forecasts or other characterisations of future events or circumstances, including any underlying assumptions, is a forward-looking statement. The words "anticipate", "believe", "continue", "could", "estimate", "expect", "intend", "may", "might", "plan", "possible", "potential", "predict", "project", "seek", "should", "would" and similar expressions, or in each case their negatives, may identify forward-looking statements, but the absence of these words does not mean that a statement is not forward-looking.
Forward-looking statements include all matters that are not historical facts. Forward-looking statements are based on the current expectations and assumptions regarding the Company, the business, the economy and other future conditions. Because forward-looking statements relate to the future, by their nature, they are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict. Forward-looking statements are not guarantees of future performance and the Company's actual financial condition, actual results of operations and financial performance, and the development of the industries in which it operates or will operate, may differ materially from those made in or suggested by the forward-looking statements contained in this announcement. In addition, even if the Company's financial condition, results of operations and the development of the industries in which it operates or will operate, are consistent with the forward-looking statements contained in this announcement, those results or developments may not be indicative of financial condition, results of operations or developments in subsequent periods. Important factors that could cause actual results to differ materially from those in the forward-looking statements include regional, national or global, political, economic, social, business, technological, competitive, market and regulatory conditions.
Any forward-looking statement contained in this announcement applies only as of the date of this announcement and is expressly qualified in its entirety by these cautionary statements. Factors or events that could cause the Company's actual plans or results to differ may emerge from time to time, and it is not possible for the Company to predict all of them. The Company expressly disclaims any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements contained in this announcement to reflect any change in its expectations or any change in events, conditions or circumstances on which any forward-looking statement contained in this announcement is based, unless required to do so by applicable law, the Prospectus Regulation Rules, the Listing Rules, the Disclosure Guidance and Transparency Rules of the FCA or the UK Market Abuse Regulation.
This information is provided by RNS, the news service of the London Stock Exchange. RNS is approved by the Financial Conduct Authority to act as a Primary Information Provider in the United Kingdom. Terms and conditions relating to the use and distribution of this information may apply. For further information, please contact [email protected] or visit www.rns.com.
SOURCE: Genflow Biosciences PLC
View the original press release on ACCESS Newswire
T.Ward--AMWN

