
-
 Venezuela stun United States to win World Baseball Classic
Venezuela stun United States to win World Baseball Classic
-
Stocks extend gains and oil dips as US, Israel, Iran continue strikes
-
 Iran missile fire kills two in central Israel: medics
Iran missile fire kills two in central Israel: medics
-
Britain, Rwanda in £100m court clash over migrant deal

-
 'We will wait for each one': Ukrainians greet POWs with tears and cheers
'We will wait for each one': Ukrainians greet POWs with tears and cheers
-
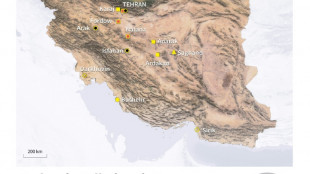
UN watchdog says projectile struck Iran nuclear power plant
-
 Trump faces impasse over Iran war
Trump faces impasse over Iran war
-
US Fed expected to hold rates steady as Iran war's shockwaves ripple

-
 Former Australian Test wicketkeeper Haddin to coach NSW
Former Australian Test wicketkeeper Haddin to coach NSW
-
China coach says team on right track despite Asian Cup heartache

-
 Oscars audience drops, viewing figures show
Oscars audience drops, viewing figures show
-
Resilient Australia 'need to be better' in Women's Asian Cup final

-
 Gio Reyna picked for US squad as Pochettino says World Cup roster still 'open'
Gio Reyna picked for US squad as Pochettino says World Cup roster still 'open'
-
Colombia, Ecuador leaders clash over bomb dropped near border

-
 PSG, Real Madrid and Arsenal march into Champions League last eight
PSG, Real Madrid and Arsenal march into Champions League last eight
-
'Incomplete' Man City not what they once were, says Guardiola

-
 US judge orders Trump admin to bring VOA employees back to work
US judge orders Trump admin to bring VOA employees back to work
-
White House pressure on Cuba mounts as island fights power cut

-
 Arteta hails 'magical' Eze after Arsenal star sinks Leverkusen
Arteta hails 'magical' Eze after Arsenal star sinks Leverkusen
-
Senegal stripped of AFCON title, Morocco declared champions

-
 Nvidia says restarting production of China-bound chips
Nvidia says restarting production of China-bound chips
-
Panic as Israel army urges residents to evacuate south Lebanon's Tyre area

-
 Real Madrid 'change' under Champions League spotlight: Vinicius
Real Madrid 'change' under Champions League spotlight: Vinicius
-
Real Madrid dump Man City out of Champions League once more

-
 Clinical PSG bury Chelsea to reach Champions League quarter-finals
Clinical PSG bury Chelsea to reach Champions League quarter-finals
-
Eze rocket fires Arsenal into Champions League quarters

-
 US airlines still see strong demand as jet fuel worries loom
US airlines still see strong demand as jet fuel worries loom
-
Milei blasts Iran on anniversary of attack on Israeli embassy

-
 USS Gerald R. Ford: the world's biggest aircraft carrier
USS Gerald R. Ford: the world's biggest aircraft carrier
-
US, European stocks rise despite latest jump in oil prices

-
 Sporting Lisbon thrash Bodo/Glimt to reach Champions League quarters
Sporting Lisbon thrash Bodo/Glimt to reach Champions League quarters
-
Irish PM pushes Trump on Iran -- politely

-
 Arizona charges prediction market Kalshi with illegal election betting
Arizona charges prediction market Kalshi with illegal election betting
-
Leftist New York mayor under pressure on Irish unity question

-
 Atletico boss Simeone defends Spurs star Romero
Atletico boss Simeone defends Spurs star Romero
-
Iran vets friendly ships for Hormuz passage: trackers

-
 Iran women's football team arrive in Turkey on way home
Iran women's football team arrive in Turkey on way home
-
Mexico prepared to host Iran World Cup games, says president

-
 Trump blasts 'foolish' NATO on Iran, says US needs no help
Trump blasts 'foolish' NATO on Iran, says US needs no help
-
Slot vows to win back support of frustrated Liverpool fans

-
 In Ukraine, Sean Penn gifted Oscar made from train carriage hit by Russia
In Ukraine, Sean Penn gifted Oscar made from train carriage hit by Russia
-
Ships in Gulf risk shortages on board, industry warns

-
 White House piles pressure on Cuba as island fights power cut
White House piles pressure on Cuba as island fights power cut
-
Newcastle must grow under Camp Nou pressure: Howe

-
 Trump says to make delayed China trip in 'five or six weeks'
Trump says to make delayed China trip in 'five or six weeks'
-
Kompany warns of complacency as injury-hit Bayern host Atalanta

-
 SAS cancels flights after fuel prices surge
SAS cancels flights after fuel prices surge
-
New particle discovered by Large Hadron Collider

-
 Lebanon says Israeli strikes kill soldiers, as shelters overflow
Lebanon says Israeli strikes kill soldiers, as shelters overflow
-
Van de Ven insists it's 'nonsense' to say players don't care about Spurs' plight


Moderna Announces the U.S. Food and Drug Administration will Initiate the Review of Its Investigational Seasonal Influenza Vaccine Submission
Following a Type A meeting, the FDA's Center for Biologics Evaluation and Research has notified Moderna that its biologics license application for mRNA-1010 will proceed to review
Moderna has received a Prescription Drug User Fee Act (PDUFA) goal date of August 5, 2026
CAMBRIDGE, MASSACHUSETTS / ACCESS Newswire / February 18, 2026 / Moderna, Inc. (NASDAQ:MRNA) today announced that, in response to a prior Refusal-to-File (RTF) letter, the Company engaged with the U.S. Food and Drug Administration (FDA) in a Type A meeting and proposed a revised regulatory approach for its investigational seasonal influenza vaccine candidate, mRNA-1010. To advance the review, Moderna proposed a regulatory pathway based on age, seeking full approval for adults 50 to 64 years of age and accelerated approval for adults 65 and older, along with a post-marketing requirement to conduct an additional study in older adults.
Following submission of the amended application, the FDA has accepted the biologics license application (BLA) for review and assigned a Prescription Drug User Fee Act (PDUFA) goal date of August 5, 2026. Pending review and FDA approval, mRNA-1010 would be available for U.S. adults 50 years of age and older, including adults 65 and older, for the 2026/2027 flu season.
"We appreciate the FDA's engagement in a constructive Type A meeting and its agreement to advance our application for review," said Stéphane Bancel, Chief Executive Officer of Moderna. "Pending FDA approval, we look forward to making our flu vaccine available later this year so that America's seniors have access to a new option to protect themselves against flu."
mRNA-1010 has now been accepted for review in the United States, Europe, Canada and Australia with further submissions planned in 2026. Moderna expects the first potential approvals for mRNA-1010 in 2026, subject to various ongoing regulatory reviews.
About Moderna
Moderna is a pioneer and leader in the field of mRNA medicine. Through the advancement of its technology platform, Moderna is reimagining how medicines are made to transform how we treat and prevent diseases. Since its founding, Moderna's mRNA platform has enabled the development of vaccines and therapeutics across infectious diseases, cancer, rare diseases and more.
With a global team and a unique culture, driven by the company's values and mindsets, Moderna's mission is to deliver the greatest possible impact to people through mRNA medicines. For more information about Moderna, please visit modernatx.com and connect with us on X, Facebook, Instagram, YouTube and LinkedIn.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including statements regarding: the FDA's review of mRNA-1010 and the potential for FDA approval; mRNA-1010's PDUFA goal date; the availability of mRNA-1010 for adults 50 years of age and older for the 2026/2027 flu season, pending FDA approval; Moderna's pending regulatory submissions for mRNA-1010 in Europe, Canada and Australia; Moderna's submissions in additional countries planned for 2026; and timing for the earliest potential approvals for mRNA-1010, subject to regulatory reviews. In some cases, forward-looking statements can be identified by terminology such as "will," "may," "should," "could," "expects," "intends," "plans," "aims," "anticipates," "believes," "estimates," "predicts," "potential," "continue," or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. The forward-looking statements in this press release are neither promises nor guarantees, and you should not place undue reliance on these forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond Moderna's control and which could cause actual results to differ materially from those expressed or implied by these forward-looking statements. These risks, uncertainties, and other factors include, among others, those risks and uncertainties described under the heading "Risk Factors" in Moderna's Annual Report on Form 10-K for the fiscal year ended December 31, 2024, filed with the U.S. Securities and Exchange Commission (SEC), and in subsequent filings made by Moderna with the SEC, which are available on the SEC's website at www.sec.gov. Except as required by law, Moderna disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this press release in the event of new information, future developments or otherwise. These forward-looking statements are based on Moderna's current expectations and speak only as of the date of this press release.
Moderna Contacts
Media:
Chris Ridley
Vice President, Global Head of Communications
+1 617-800-3651
[email protected]
Investors:
Lavina Talukdar
Senior Vice President & Head of Investor Relations
+1 617-209-5834
[email protected]
SOURCE: Moderna, Inc.
View the original press release on ACCESS Newswire
P.Costa--AMWN


