
-
 Mass MS-13 trial held at El Salvador mega-jail
Mass MS-13 trial held at El Salvador mega-jail
-
Barcelona must live without teen star Yamal for title run-in

-
 Hearts lead Old Firm as Scottish title race heads for tense finale
Hearts lead Old Firm as Scottish title race heads for tense finale
-
India criticizes 'poor taste' Trump post against immigrants

-
 China's DeepSeek says releases long-awaited new AI model
China's DeepSeek says releases long-awaited new AI model
-
Hawks fend off Knicks, Raptors pull away from Cavs to cut deficit

-
 Wildfires spread towards northern Japan town
Wildfires spread towards northern Japan town
-
Israel, Lebanon extend ceasefire as Iran peace talks stall

-
 'Clearly me': AI drama accused of stealing faces
'Clearly me': AI drama accused of stealing faces
-
Soviet architecture vanishes as Central Asia drifts from Moscow

-
 Oil extends gains, stocks sink as peace talk hopes fade
Oil extends gains, stocks sink as peace talk hopes fade
-
'Raw and honest': India climbers face obstacles in race to the top

-
 Cowgirls of Philippine rodeo tackle steers, stereotypes
Cowgirls of Philippine rodeo tackle steers, stereotypes
-
'Godzilla Minus Zero' will show monster up close, director says

-
 'Stigmatized' or 'sustainable'? Vintage sales boost sees fur return
'Stigmatized' or 'sustainable'? Vintage sales boost sees fur return
-
YouTube offers deepfake detection to Hollywood

-
 US soldier allegedly bet on Maduro operation using intel
US soldier allegedly bet on Maduro operation using intel
-
Bill to legalise assisted dying in England and Wales set to fail

-
 Arsenal eye return to top spot, Spurs fight for survival
Arsenal eye return to top spot, Spurs fight for survival
-
Child vaccine catch-up drive on course to hit target: UN
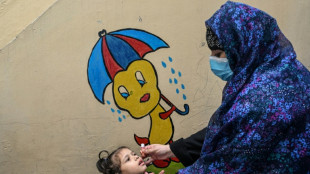
-
 Chinese EVs geared up to dominate world's biggest auto show
Chinese EVs geared up to dominate world's biggest auto show
-
No.2 Korda fires 65 to grab LPGA Chevron lead

-
 Raiders take quarterback Mendoza with No. 1 NFL draft pick
Raiders take quarterback Mendoza with No. 1 NFL draft pick
-
Lebanon leaders accuse Israel of war crime after journalist killed

-
 Stuffed toys in US capital symbolize displaced Ukrainian children
Stuffed toys in US capital symbolize displaced Ukrainian children
-
Lakers' Reaves could return for game three against Rockets

-
 US says Iran players welcome at World Cup amid Italy uproar
US says Iran players welcome at World Cup amid Italy uproar
-
Images of dead Maradona rock trial of medical team

-
 US invites Putin to G20 summit but Trump doubts he'll come
US invites Putin to G20 summit but Trump doubts he'll come
-
Israel, Lebanon extend ceasefire as Trump hopes for historic deal

-
 G20 summit invites to include Russia: US official
G20 summit invites to include Russia: US official
-
Last-gasp Tomas stunner sends Stuttgart into German Cup final

-
 Rights groups warn World Cup visitors over US travel
Rights groups warn World Cup visitors over US travel
-
Intel earnings signal recovery at US chip maker

-
 Trump rules out striking Iran with nuclear weapon
Trump rules out striking Iran with nuclear weapon
-
Stocks mostly fall as US-Iran peace talks stall and oil prices rise

-
 Meta plans 10% layoffs as AI spending soars: source
Meta plans 10% layoffs as AI spending soars: source
-
Trump 'gold card' visa granted to one person so far: US commerce chief

-
 EU unblocks funds as Ukraine presses for membership progress
EU unblocks funds as Ukraine presses for membership progress
-
Trump says US in no rush but 'clock is ticking' for Iran

-
 OpenAI says new model adept at making AI better
OpenAI says new model adept at making AI better
-
Child porn found on D4vd's phone: prosecutor in teen murder case

-
 Trump to meet Lebanon, Israel envoys on truce extension
Trump to meet Lebanon, Israel envoys on truce extension
-
Samson, Hosein star as Chennai hammer Mumbai by 103 runs in IPL

-
 Bolivia, Chile move to restore ties severed 50 years ago
Bolivia, Chile move to restore ties severed 50 years ago
-
Bayern fined but avoid fan ban over Champions League crowd incident

-
 Wembanyama will travel with Spurs but uncertain for next game
Wembanyama will travel with Spurs but uncertain for next game
-
Italy dismisses talk of replacing Iran at World Cup

-
 New multilateral force for gang-plagued Haiti to deploy soon, UN told
New multilateral force for gang-plagued Haiti to deploy soon, UN told
-
Canada not as reliant on US economy as some think: Carney


Amphastar Announces FDA Approval for Ipratropium Bromide HFA
RANCHO CUCAMONGA, CA / ACCESS Newswire / February 24, 2026 / Amphastar Pharmaceuticals, Inc. (NASDAQ:AMPH) announced that the U.S. Food and Drug Administration ("FDA") has approved the Company's Abbreviated New Drug Application ("ANDA") for Ipratropium Bromide HFA Inhalation Aerosol 17mcg/actuation. The FDA determined that Amphastar's Ipratropium Bromide HFA Inhalation Aerosol is bioequivalent and therapeutically equivalent to Boehringer Ingelheim's Atrovent® HFA Inhalation Aerosol.
Additionally, the FDA has confirmed that this product is eligible for 180-days of generic drug exclusivity for Ipratropium Bromide HFA Inhalation Aerosol as we were the first ANDA applicant with Paragraph IV certification. This exclusivity period will begin on the first day that Amphastar's Ipratropium Bromide HFA Inhalation Aerosol is commercially launched.
"We are excited to announce FDA approval of Ipratropium Bromide HFA Inhalation Aerosol, reinforcing the strength of our integrated R&D and manufacturing model and demonstrating our ability to deliver complex, high-value generics," said Dr. Jack Zhang, Amphastar's President and Chief Executive Officer. "We expect this launch to contribute meaningfully to our respiratory portfolio and to further strengthen our long-term growth strategy, capabilities and commitment to delivering impactful therapies to patients as we increase our efforts on the development of proprietary pipeline candidates."
Ipratropium is an anticholinergic indicated for the maintenance treatment of bronchospasm associated with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema. According to IQVIA, the U.S. sales for Atrovent® HFA were approximately $112 million for the 12 months ended December 31, 2025.
Amphastar plans to launch its Ipratropium Bromide HFA Inhalation Aerosol, previously referred to as AMP-007, early in the second quarter of 2026.
Pipeline Information
The Company currently has one ANDA and one biosimilar insulin filed with the FDA targeting products with a combined market size of over $1.7 billion, along with two biosimilar products in development targeting products with a market size exceeding $3.7 billion, and two generic products in development targeting products with a market size of over $1 billion. This market information is based on IQVIA data for the 12 months ended December 31, 2025. The Company is developing multiple proprietary products with injectable and intranasal dosage forms.
The Company's proprietary pipeline also includes four recently in-licensed products including three proprietary peptides targeting oncology and ophthalmology indications, and a fully synthetic corticotropin compound designed to address inflammatory and autoimmune conditions.
About Amphastar Pharmaceuticals, Inc.
Amphastar is a biopharmaceutical company that focuses on developing, manufacturing, and commercializing technically challenging generic and proprietary injectable, inhalation, and intranasal products. Additionally, the Company sells active pharmaceutical ingredient, or API products. Most of the Company's finished products are contracted and distributed through group purchasing organizations, drug wholesalers, and drug retailers. More information and resources are available at www.amphastar.com.
Amphastar's logo and other trademarks or service marks of Amphastar, including, but not limited to Amphastar ®, BAQSIMI ®, Primatene MIST ®, REXTOVY ®, Amphadase ®, AmpsilogTM and Cortrosyn ®, are the property of Amphastar.
Forward-Looking Statements
All statements in this press release referenced above that are not historical are forward-looking statements, including, among other things, statements relating to our expectations regarding future financial performance and business trends, our future growth and our ability to continue to scale, sales and marketing of our products, market size and expansion, product portfolio, product development, the timing of FDA filings or approvals, the timing of product launches, acquisitions and other matters related to our pipeline of product candidates, the timing and results of clinical trials, the impact of our products, including their potential for continued revenue growth, the strategic trajectory of and market for our product pipeline, our long-term strategic vision, our ability to leverage our existing expertise and technology, the impacts of any licensing agreements and ability to commercialize additional therapies, our in-house manufacturing expertise, our ability to deliver high-quality, affordable therapies to patients, our commercial momentum and position in the market. These statements are not facts but rather are based on Amphastar's historical performance and our current expectations, estimates, and projections regarding our business, operations, and other similar or related factors. Words such as "may," "might," "will," "could," "would," "should," "anticipate," "predict," "potential," "continue," "expect," "intend," "plan," "project," "believe," "estimate," and other similar or related expressions are used to identify these forward-looking statements, although not all forward-looking statements contain these words. You should not place undue reliance on forward-looking statements because they involve known and unknown risks, uncertainties, and assumptions that are difficult or impossible to predict and, in some cases, beyond Amphastar's control. Actual results may differ materially from those in the forward-looking statements as a result of a number of factors, including those described in Amphastar's filings with the Securities and Exchange Commission ("SEC"), including in our Annual Report on Form 10-K for the year ended December 31, 2024, filed with the SEC on March 3, 2025, in our Quarterly Report on Form 10-Q for the quarter ended March 31, 2025, filed with the SEC on May 8, 2025, in our Quarterly Report on Form 10-Q for the quarter ended June 30, 2025, filed with the SEC on August 7, 2025, in our Quarterly Report on Form 10-Q for the quarter ended September 30, 2025, filed with the SEC on November 6, 2025, and our other filings or reports that we may file with the SEC. In particular, there can be no guarantee that our sales strategies will be successful, or that we will continue to experience significant sales of BAQSIMI ®. You can locate these reports through our website at http://ir.amphastar.com and on the SEC's website at www.sec.gov. The forward-looking statements in this release speak only as of the date of the release. Amphastar undertakes no obligation to revise or update information or any forward-looking statements in this press release referenced above to reflect events or circumstances in the future, even if new information becomes available or if subsequent events cause our expectations to change.
Contact Information:
Amphastar Pharmaceuticals, Inc.
Bill Peters
Chief Financial Officer
(909) 476-3416
SOURCE: Amphastar Pharmaceuticals, Inc.
View the original press release on ACCESS Newswire
M.Thompson--AMWN
