
-
 Musk vs OpenAI trial enters second week
Musk vs OpenAI trial enters second week
-
India's Modi faces key test as vote count underway

-
 Japan PM says oil crisis has 'enormous impact' in Asia-Pacific
Japan PM says oil crisis has 'enormous impact' in Asia-Pacific
-
Badminton no.1 An brings 'fire' as South Korea win Uber Cup

-
 Saka sparks Arsenal attack into life ahead of Atletico showdown
Saka sparks Arsenal attack into life ahead of Atletico showdown
-
Atletico aim to show Alvarez their ambition in Arsenal semi

-
 Seoul, Taipei hit records as Asian stocks track Wall St tech rally
Seoul, Taipei hit records as Asian stocks track Wall St tech rally
-
Boeing faces civil trial over 737 MAX crash

-
 Australian inquiry opens public hearings into Bondi Beach shooting
Australian inquiry opens public hearings into Bondi Beach shooting
-
Iran warns of ceasefire violation as US plans to escort Hormuz ships

-
 North Korean club to play rare football match in South
North Korean club to play rare football match in South
-
Pistons rout Magic to cap comeback, book NBA playoff clash with Cavaliers

-
 Japan, Australia discuss energy, critical minerals
Japan, Australia discuss energy, critical minerals
-
Village braces for closure of Spain's largest nuclear plant

-
 GameStop makes $56 billion takeover bid for eBay
GameStop makes $56 billion takeover bid for eBay
-
Ex-NY mayor Giuliani hospitalized in 'critical' condition: spokesman

-
 Europe, Canada leaders hold Yerevan talks in Trump's shadow
Europe, Canada leaders hold Yerevan talks in Trump's shadow
-
'No pilgrims': regional war hushes Iraq's holy cities

-
 Israel court extends detention of two Gaza flotilla activists
Israel court extends detention of two Gaza flotilla activists
-
Massive search continues for two missing US soldiers in Morocco

-
 Players keep up battle with tennis majors as they decry Roland Garros prize money
Players keep up battle with tennis majors as they decry Roland Garros prize money
-
Evotec Announces Nomination of First Preclinical Development Candidate in Dermatology Collaboration with Almirall

-
 EB5 United Surpasses 800 I-526E Approvals in Post-RIA EB-5 Landscape
EB5 United Surpasses 800 I-526E Approvals in Post-RIA EB-5 Landscape
-
Pistons rout Magic to complete comeback, advance in NBA playoffs

-
 Trump says US and Iran in 'positive' talks, unveils plan to escort Hormuz ships
Trump says US and Iran in 'positive' talks, unveils plan to escort Hormuz ships
-
Talisman Endrick fires resurgent Lyon into third in France

-
 Verstappen laments spin and struggle for pace in Miami
Verstappen laments spin and struggle for pace in Miami
-
Teen Antonelli wins again in Miami to extend title race lead

-
 Ferrari's Leclerc admits he threw away Miami podium finish
Ferrari's Leclerc admits he threw away Miami podium finish
-
Cristian Chivu, a winner with Inter on the pitch and in the dugout

-
 Key players from Inter Milan's Serie A title triumph
Key players from Inter Milan's Serie A title triumph
-
No.4 Young cruises to PGA title at Doral

-
 Vinicius double delays Barca title as Real Madrid down Espanyol
Vinicius double delays Barca title as Real Madrid down Espanyol
-
Inter Milan win Italian title for third time in six seasons

-
 Spurs solved mental frailty to boost survival bid: De Zerbi
Spurs solved mental frailty to boost survival bid: De Zerbi
-
Miami champ Antonelli shrugs off success, vows 'back to work'

-
 Man Utd beat Liverpool, Spurs climb out of relegation zone
Man Utd beat Liverpool, Spurs climb out of relegation zone
-
Spurs out of relegation zone after vital win at Villa

-
 No.1 Korda cruises to LPGA Mexico crown
No.1 Korda cruises to LPGA Mexico crown
-
Thompson-Herah shines at world relays, Tebogo helps Botswana to win

-
 Three die on Atlantic cruise ship from suspected hantavirus: WHO
Three die on Atlantic cruise ship from suspected hantavirus: WHO
-
Germany's Merz says not 'giving up on working with Donald Trump'

-
 Mercedes' Kimi Antonelli wins Miami Grand Prix
Mercedes' Kimi Antonelli wins Miami Grand Prix
-
Man Utd job feels 'natural' to Carrick

-
 Ferguson taken to hospital before Man Utd win against Liverpool
Ferguson taken to hospital before Man Utd win against Liverpool
-
'Devil Wears Prada 2' takes top spot in N. America box office

-
 Iran weighs US response to peace plan after warning against military action
Iran weighs US response to peace plan after warning against military action
-
Gladbach sink Dortmund, St Pauli edge closer to drop

-
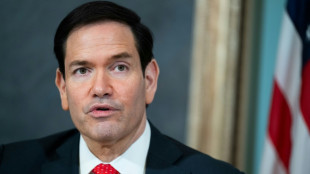 Rubio to visit Rome, meet Pope Leo after Trump row
Rubio to visit Rome, meet Pope Leo after Trump row
-
Kyiv hits Russian oil sites as eight killed in both countries


Ensysce Biosciences Initiates Live "Ask Me Anything" (AMA) Session Highlighting Growth and Future Plans
~ Underscores Strategic Execution Plans, Regulatory Risk Mitigation Efforts, Pipeline Prioritization, and the Commercial Potential of PF614 ~
SAN DIEGO, CALIFORNIA / ACCESS Newswire / March 4, 2026 / Ensysce Biosciences, Inc. (NASDAQ:ENSC) ("Ensysce" or the "Company"), a clinical-stage pharmaceutical company pioneering next-generation pain and central nervous system therapeutics designed to minimize risk of abuse and overdose, today announced the successful initiation of its live "Ask Me Anything" (AMA) series on March 4, 2024.The event reinforced the Company's commitment to transparency, accessibility, and open dialogue with its community. A replay of the AMA series is available on the Company's website here.
The AMA provided Ensysce shareholders and followers with the opportunity to engage directly with Dr. Lynn Kirkpatrick, Chief Executive Officer of Ensysce, covering topics such as Phase 3 execution and timeline visibility, capital discipline and shareholder alignment, as well as strategic priorities and long-term value creation. The event generated numerous distinct questions, arranged under 3 topics reflecting strong engagement and interest from the community.
"Our recent AMA session provided an important opportunity to engage directly with stakeholders and reinforce the strategic clarity behind our PF614 program," said Dr. Kirkpatrick. "We believe there is significant commercial potential for PF614 and that we are positioning the program to address unmet needs in the marketplace. We remain focused on disciplined execution and transparent communication as we advance these programs toward meaningful milestones."
Key highlights from the AMA included:
PF614-301 timeline sequencing
Efforts to reduce clinical and regulatory uncertainty
PF614, PF614-MPAR & ADHD pipeline prioritization
Potential PF614 commercial opportunities
For those who were unable to have their questions answered, please reach out for future opportunities to investor relations at [email protected].
About Ensysce Biosciences
Ensysce Biosciences is a clinical-stage company with a goal of disrupting the analgesic landscape by introducing a new class of highly novel opioids for the treatment of severe pain. Leveraging its Trypsin-Activated Abuse Protection (TAAP™) and Multi-Pill Abuse Resistance (MPAR®) platforms, the Company is developing unique, tamper-proof treatment options for pain that minimize the risk of both drug abuse and overdose. Ensysce's products are anticipated to provide safer options to treat patients suffering from severe pain and assist in preventing deaths caused by medication abuse. For more information, please visit www.ensysce.com.
Forward-Looking Statements
Statements contained in this press release that are not purely historical may be deemed to be forward-looking statements for the purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995 and other federal securities laws. Without limiting the foregoing, the use of words such as "may," "intends," "can," "might," "will," "expect," "plan," "possible," "believe" and other similar expressions are intended to identify forward-looking statements. The product candidates discussed are in clinic and not approved and there can be no assurance that the clinical programs will be successful in demonstrating safety and/or efficacy, that Ensysce will not encounter problems or delays in clinical development, or that any product candidate will ever receive regulatory approval or be successfully commercialized. All forward-looking statements are based on estimates and assumptions by Ensysce's management that, although Ensysce believes to be reasonable, are inherently uncertain. All forward-looking statements are subject to risks and uncertainties that may cause actual results to differ materially from those that Ensysce expected. In addition, Ensysce's business is subject to additional risks and uncertainties, including among others, the initiation and conduct of preclinical studies and clinical trials; the timing and availability of data from preclinical studies and clinical trials; expectations for regulatory submissions and approvals; potential safety concerns related to, or efficacy of, Ensysce's product candidates; the availability or commercial potential of product candidates; the ability of Ensysce to fund its continued operations, including its planned clinical trials; the dilutive effect of stock issuances from our fundraising; and Ensysce's and its partners' ability to perform under their license, collaboration and manufacturing arrangements. These statements are also subject to a number of material risks and uncertainties that are described in Ensysce's most recent quarterly report on Form 10-Q and current reports on Form 8-K, which are available, free of charge, at the SEC's website at www.sec.gov. Any forward-looking statement speaks only as of the date on which it was made. Ensysce undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except as required under applicable law.
Ensysce Biosciences Company Contact:
Lynn Kirkpatrick, Ph.D.
Chief Executive Officer
(858) 263-4196
Ensysce Biosciences Investor Relations Contact:
Shannon Devine
MZ North America
Main: 203-741-8811
[email protected]
SOURCE: Ensysce Biosciences
View the original press release on ACCESS Newswire
Ch.Kahalev--AMWN
