
-
 Musk vs OpenAI trial enters second week
Musk vs OpenAI trial enters second week
-
India's Modi faces key test as vote count underway

-
 Japan PM says oil crisis has 'enormous impact' in Asia-Pacific
Japan PM says oil crisis has 'enormous impact' in Asia-Pacific
-
Badminton no.1 An brings 'fire' as South Korea win Uber Cup

-
 Saka sparks Arsenal attack into life ahead of Atletico showdown
Saka sparks Arsenal attack into life ahead of Atletico showdown
-
Atletico aim to show Alvarez their ambition in Arsenal semi

-
 Seoul, Taipei hit records as Asian stocks track Wall St tech rally
Seoul, Taipei hit records as Asian stocks track Wall St tech rally
-
Boeing faces civil trial over 737 MAX crash

-
 Australian inquiry opens public hearings into Bondi Beach shooting
Australian inquiry opens public hearings into Bondi Beach shooting
-
Iran warns of ceasefire violation as US plans to escort Hormuz ships

-
 North Korean club to play rare football match in South
North Korean club to play rare football match in South
-
Pistons rout Magic to cap comeback, book NBA playoff clash with Cavaliers

-
 Japan, Australia discuss energy, critical minerals
Japan, Australia discuss energy, critical minerals
-
Village braces for closure of Spain's largest nuclear plant

-
 GameStop makes $56 billion takeover bid for eBay
GameStop makes $56 billion takeover bid for eBay
-
Ex-NY mayor Giuliani hospitalized in 'critical' condition: spokesman

-
 Europe, Canada leaders hold Yerevan talks in Trump's shadow
Europe, Canada leaders hold Yerevan talks in Trump's shadow
-
'No pilgrims': regional war hushes Iraq's holy cities

-
 Israel court extends detention of two Gaza flotilla activists
Israel court extends detention of two Gaza flotilla activists
-
Massive search continues for two missing US soldiers in Morocco

-
 Players keep up battle with tennis majors as they decry Roland Garros prize money
Players keep up battle with tennis majors as they decry Roland Garros prize money
-
Evotec Announces Nomination of First Preclinical Development Candidate in Dermatology Collaboration with Almirall

-
 EB5 United Surpasses 800 I-526E Approvals in Post-RIA EB-5 Landscape
EB5 United Surpasses 800 I-526E Approvals in Post-RIA EB-5 Landscape
-
Pistons rout Magic to complete comeback, advance in NBA playoffs

-
 Trump says US and Iran in 'positive' talks, unveils plan to escort Hormuz ships
Trump says US and Iran in 'positive' talks, unveils plan to escort Hormuz ships
-
Talisman Endrick fires resurgent Lyon into third in France

-
 Verstappen laments spin and struggle for pace in Miami
Verstappen laments spin and struggle for pace in Miami
-
Teen Antonelli wins again in Miami to extend title race lead

-
 Ferrari's Leclerc admits he threw away Miami podium finish
Ferrari's Leclerc admits he threw away Miami podium finish
-
Cristian Chivu, a winner with Inter on the pitch and in the dugout

-
 Key players from Inter Milan's Serie A title triumph
Key players from Inter Milan's Serie A title triumph
-
No.4 Young cruises to PGA title at Doral

-
 Vinicius double delays Barca title as Real Madrid down Espanyol
Vinicius double delays Barca title as Real Madrid down Espanyol
-
Inter Milan win Italian title for third time in six seasons

-
 Spurs solved mental frailty to boost survival bid: De Zerbi
Spurs solved mental frailty to boost survival bid: De Zerbi
-
Miami champ Antonelli shrugs off success, vows 'back to work'

-
 Man Utd beat Liverpool, Spurs climb out of relegation zone
Man Utd beat Liverpool, Spurs climb out of relegation zone
-
Spurs out of relegation zone after vital win at Villa

-
 No.1 Korda cruises to LPGA Mexico crown
No.1 Korda cruises to LPGA Mexico crown
-
Thompson-Herah shines at world relays, Tebogo helps Botswana to win

-
 Three die on Atlantic cruise ship from suspected hantavirus: WHO
Three die on Atlantic cruise ship from suspected hantavirus: WHO
-
Germany's Merz says not 'giving up on working with Donald Trump'

-
 Mercedes' Kimi Antonelli wins Miami Grand Prix
Mercedes' Kimi Antonelli wins Miami Grand Prix
-
Man Utd job feels 'natural' to Carrick

-
 Ferguson taken to hospital before Man Utd win against Liverpool
Ferguson taken to hospital before Man Utd win against Liverpool
-
'Devil Wears Prada 2' takes top spot in N. America box office

-
 Iran weighs US response to peace plan after warning against military action
Iran weighs US response to peace plan after warning against military action
-
Gladbach sink Dortmund, St Pauli edge closer to drop

-
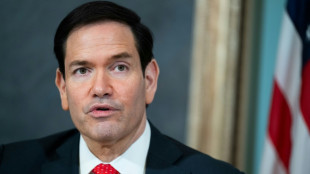 Rubio to visit Rome, meet Pope Leo after Trump row
Rubio to visit Rome, meet Pope Leo after Trump row
-
Kyiv hits Russian oil sites as eight killed in both countries


Pres. Trump's Marijuana Executive Mandate Accelerates MMJ International Holdings Federal Alignment Toward Pharmaceutical
Duane Boise, CEO, MMJ International Holdings stated, "Positioned with Active INDs, Orphan Drug Designations, and Standardized Soft-Gel Formulations, MMJ aligns with the FDA's Botanical Drug Pathway as Federal Oversight Expands. As Schedule III Normalization Hardens the Regulatory Landscape, MMJIH Secures the Lead in FDA Validated Medicine."
WASHINGTON, D.C. / ACCESS Newswire / March 4, 2026 / In the wake of President Donald J. Trump's December 2025 Executive Order, "Increasing Medical Marijuana and Cannabidiol Research," the American cannabis industry has reached a decisive inflection point.

The directive - issued from the White House and coordinated through the U.S. Department of Justice and the Department of Health and Human Services - accelerated the administrative pathway toward Schedule III reclassification under the Controlled Substances Act.
This shift does not represent deregulation. It represents federal normalization - and normalization inherently favors clinical structure over retail dispersion.
As regulatory gravity consolidates under the U.S. Food and Drug Administration and the Drug Enforcement Administration, MMJ International Holdings (MMJIH) emerges structurally aligned with the new architecture.
The Federalization of Cannabis: A Regulatory Hardening
Schedule III normalization signals that cannabis is recognized as having accepted medical use, but it does not legalize retail markets. Instead, it intensifies federal oversight.
The transition reframes cannabis as a regulated therapeutic input rather than a loosely governed botanical commodity.
Under this hardened environment:
Clinical validation replaces marketing claims
Dose reproducibility replaces strain variability
Federal manufacturing standards replace patchwork state rules
IND-backed trials replace anecdotal evidence
For much of the state-based cannabis industry, this creates operational friction.
For MMJIH, it validates a decade-long strategy.
MMJIH's Structural Moat
While many operators pursued state retail expansion, MMJIH built infrastructure aligned with federal drug-development requirements.
1. Advanced FDA Engagement
MMJIH holds active Investigational New Drug (IND) applications and Orphan Drug Designations for high-value neurological conditions, including Huntington's Disease and Multiple Sclerosis. These filings exist within the FDA's botanical drug development framework - the same pathway that led to approval of Epidiolex.
2. The Soft-Gel Advantage
Rather than relying on variable botanical flower, MMJIH developed standardized pharmaceutical soft-gel capsules (MMJ-001 and MMJ-002).
Soft-gel dosage forms enable:
Precise milligram-level dosing
Stability testing
Batch-to-batch reproducibility
Clinical trial consistency
Scalable GMP manufacturing
In pharmaceutical terms, formulation is control.
3. Federal Compliance Infrastructure
Operating within a DEA aligned research and manufacturing framework, MMJIH has structured its cultivation, lab, and processing operations to comply with federal standards - not merely state retail compliance.
Schedule III does not create this capability.
It rewards it.
A Message from the CEO
"Schedule III doesn't lift all boats; it separates medicine from merchandise," said Duane Boise, President & CEO of MMJ International Holdings. "This realignment is about allowing science to operate within a federally coherent structure. MMJ was built specifically for this architecture. We built real medicine while others built storefronts."
International Infrastructure: A Global Compliance Model
Federal normalization in the United States mirrors broader international trends.
MMJIH's strategic relationships in Europe and other regulated markets provide:
Pharmaceutical-grade logistics channels
Transparent regulatory counterparts
Supply-chain stability aligned with GMP expectations
Export readiness under treaty-compliant frameworks
As cannabinoid medicine migrates from fragmented retail to standardized healthcare models, international harmonization becomes essential.
MMJIH has already positioned itself within that framework.
Capital Migration Toward Compliance
With Schedule III normalization, capital allocation patterns are shifting.
Institutional investors, healthcare systems, and pharmaceutical distributors evaluate:
Clinical data
Regulatory durability
Intellectual property
Orphan exclusivity potential
Manufacturing defensibility
The speculative retail premium contracts.
The compliance premium expands.
In that environment, MMJIH is structurally differentiated.
The Realignment Has Begun
Rescheduling does not instantly resolve:
Federal-state conflicts
Banking statutes
Housing rules
DOT testing mandates
However, it resets the baseline classification for the first time in nearly 50 years.
That reset favors companies already integrated into the federal drug-development system.
MMJ International Holdings is not adapting to Schedule III.
It was built for it.
About MMJ International Holdings
MMJ International Holdings is a U.S.-based biopharmaceutical company focused on the development of plant-derived, FDA-aligned cannabinoid medicines for rare and chronic neurological diseases, including Huntington's Disease and Multiple Sclerosis. Through IND-backed research, orphan drug strategy, and standardized pharmaceutical dosage forms, the company operates within the federally regulated healthcare framework.
CONTACT:
Madison Hisey
[email protected]
203-231-8583
SOURCE: MMJ International Holdings
View the original press release on ACCESS Newswire
O.Karlsson--AMWN
