
-
 Seoul, Taipei hit records as Asian stocks track Wall St tech rally
Seoul, Taipei hit records as Asian stocks track Wall St tech rally
-
Boeing faces civil trial over 737 MAX crash

-
 Australian inquiry opens public hearings into Bondi Beach shooting
Australian inquiry opens public hearings into Bondi Beach shooting
-
Iran warns of ceasefire violation as US plans to escort Hormuz ships

-
 North Korean club to play rare football match in South
North Korean club to play rare football match in South
-
Pistons rout Magic to cap comeback, book NBA playoff clash with Cavaliers

-
 Japan, Australia discuss energy, critical minerals
Japan, Australia discuss energy, critical minerals
-
Village braces for closure of Spain's largest nuclear plant

-
 GameStop makes $56 billion takeover bid for eBay
GameStop makes $56 billion takeover bid for eBay
-
Ex-NY mayor Giuliani hospitalized in 'critical' condition: spokesman

-
 Europe, Canada leaders hold Yerevan talks in Trump's shadow
Europe, Canada leaders hold Yerevan talks in Trump's shadow
-
'No pilgrims': regional war hushes Iraq's holy cities

-
 Israel court extends detention of two Gaza flotilla activists
Israel court extends detention of two Gaza flotilla activists
-
Massive search continues for two missing US soldiers in Morocco

-
 Players keep up battle with tennis majors as they decry Roland Garros prize money
Players keep up battle with tennis majors as they decry Roland Garros prize money
-
Pistons rout Magic to complete comeback, advance in NBA playoffs

-
 Trump says US and Iran in 'positive' talks, unveils plan to escort Hormuz ships
Trump says US and Iran in 'positive' talks, unveils plan to escort Hormuz ships
-
Talisman Endrick fires resurgent Lyon into third in France

-
 Verstappen laments spin and struggle for pace in Miami
Verstappen laments spin and struggle for pace in Miami
-
Teen Antonelli wins again in Miami to extend title race lead

-
 Ferrari's Leclerc admits he threw away Miami podium finish
Ferrari's Leclerc admits he threw away Miami podium finish
-
Cristian Chivu, a winner with Inter on the pitch and in the dugout

-
 Key players from Inter Milan's Serie A title triumph
Key players from Inter Milan's Serie A title triumph
-
No.4 Young cruises to PGA title at Doral

-
 Vinicius double delays Barca title as Real Madrid down Espanyol
Vinicius double delays Barca title as Real Madrid down Espanyol
-
Inter Milan win Italian title for third time in six seasons

-
 Spurs solved mental frailty to boost survival bid: De Zerbi
Spurs solved mental frailty to boost survival bid: De Zerbi
-
Miami champ Antonelli shrugs off success, vows 'back to work'

-
 Man Utd beat Liverpool, Spurs climb out of relegation zone
Man Utd beat Liverpool, Spurs climb out of relegation zone
-
Spurs out of relegation zone after vital win at Villa

-
 No.1 Korda cruises to LPGA Mexico crown
No.1 Korda cruises to LPGA Mexico crown
-
Thompson-Herah shines at world relays, Tebogo helps Botswana to win

-
 Three die on Atlantic cruise ship from suspected hantavirus: WHO
Three die on Atlantic cruise ship from suspected hantavirus: WHO
-
Germany's Merz says not 'giving up on working with Donald Trump'

-
 Mercedes' Kimi Antonelli wins Miami Grand Prix
Mercedes' Kimi Antonelli wins Miami Grand Prix
-
Man Utd job feels 'natural' to Carrick

-
 Ferguson taken to hospital before Man Utd win against Liverpool
Ferguson taken to hospital before Man Utd win against Liverpool
-
'Devil Wears Prada 2' takes top spot in N. America box office

-
 Iran weighs US response to peace plan after warning against military action
Iran weighs US response to peace plan after warning against military action
-
Gladbach sink Dortmund, St Pauli edge closer to drop

-
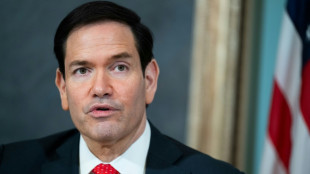 Rubio to visit Rome, meet Pope Leo after Trump row
Rubio to visit Rome, meet Pope Leo after Trump row
-
Kyiv hits Russian oil sites as eight killed in both countries

-
 Iran says US military operation 'impossible' as Trump mulls peace proposal
Iran says US military operation 'impossible' as Trump mulls peace proposal
-
Man Utd beat Liverpool to secure Champions League place

-
 Two die in 'respiratory illness' outbreak on Atlantic cruise ship
Two die in 'respiratory illness' outbreak on Atlantic cruise ship
-
Barcelona sink Bayern to reach women's Champions League final

-
 True Love lands eighth English 1000 Guineas for O'Brien
True Love lands eighth English 1000 Guineas for O'Brien
-
Sinner dismantles Zverev to win Madrid Open, set record

-
 Brilliant Bordeaux clean out Bath to reach Champions Cup final
Brilliant Bordeaux clean out Bath to reach Champions Cup final
-
Second unexploded shell found at illegal French rave: minister


From Billion-Dollar Hemp Drinks to FDA Cease-and-Desist Letters: The Cannabis Industry's Reckoning Is Coming
"Schedule III doesn't lift all boats; it separates medicine from merchandise," said Duane Boise, President & CEO of MMJ International Holdings. "This realignment is about allowing science to operate within a federally coherent structure. MMJ was built specifically for this architecture. We built real medicine while others built storefronts."
WASHINGTON, DC / ACCESS Newswire / March 6, 2026 / For the past several years, a growing segment of the cannabis industry has chased what many executives believed was the fastest route to profit: hemp-derived THC beverages, gummies, and intoxicating cannabinoid products sold under the Agriculture Improvement Act of 2018 loophole.

The business model looked irresistible.
Companies could manufacture intoxicating cannabinoid products derived from hemp, place them in convenience stores, liquor distributors, and e-commerce channels, and avoid the strict federal approval process required for pharmaceutical drugs.
But that gray zone is closing rapidly.
And when it does, many CEOs who rushed into hemp-THC beverages may find themselves wishing they had chosen a different strategy.
The Illusion of the "Hemp Loophole"
The intoxicating hemp market emerged from a narrow interpretation of the 2018 Farm Bill, which legalized hemp containing less than 0.3% delta-9 THC by dry weight.
Entrepreneurs quickly realized something important:
Chemically modified cannabinoids like delta-8 THC, delta-10 THC, and THC beverages could be produced from hemp-derived CBD and sold nationwide.
For a time, regulators struggled to respond.
But from a federal law standpoint, the situation has always been precarious.
The U.S. Food and Drug Administration has repeatedly stated that:
CBD cannot legally be marketed as a dietary supplement
Cannabinoids intended for therapeutic effects must go through the FDA drug approval process
Products making medical claims require clinical validation
Despite this, many companies raced ahead.
Why the FDA Is Likely to Act
Federal agencies are now under increasing pressure to restore order to the cannabinoid market.
Concerns include:
Mislabeling and inaccurate potency
Synthetic cannabinoid conversion processes
Youth marketing concerns
Lack of safety testing
Interstate commerce violations
Once the federal government begins issuing warning letters and cease-and-desist actions, the entire hemp intoxicant category could face rapid contraction.
And historically, that is how the FDA regulates new markets:
First warnings.
Then enforcement.
The CEOs Who Chased the Quick Buck
Many executives focused on short-term consumer trends.
Cannabis beverages appeared attractive because they resemble alcohol distribution models.
But there is a fundamental regulatory difference:
Alcohol is federally legal.
Intoxicating cannabinoids remain regulated under the Controlled Substances Act.
Companies building billion-dollar beverage brands may soon discover that federal drug law ultimately determines the rules of the game.
When enforcement begins, the industry narrative will shift quickly from celebration to regret.
The Pharmaceutical Path Most Companies Ignored
While the hemp beverage boom unfolded, a very small number of companies pursued a far more difficult path.
The FDA drug development pathway.
That path requires:
Pre-clinical toxicology studies
Investigational New Drug (IND) applications
Controlled clinical trials
Standardized dosage forms
Pharmaceutical manufacturing controls
Most cannabis companies avoided this route because it takes years of scientific work and regulatory discipline.
But one company committed to it early.
Why MMJ International Holdings Took the Hard Road
MMJ International Holdings focused on building cannabinoid medicines that could meet federal drug standards.
Rather than selling unregulated consumer products, the company pursued:
• FDA IND filings
Orphan Drug Designations for Huntington's disease and Multiple Sclerosis
Pharmaceutical-grade manufacturing
Standardized soft-gel dosage forms
This strategy aligns with the same regulatory framework that produced the FDA-approved cannabinoid drug Epidiolex.
The difference is simple.
Most companies built brands.
MMJ built medicine.
Schedule III Could Accelerate the Divide
If cannabis is ultimately reclassified under the Controlled Substances Act, the industry could split into two very different sectors.
One side:
Retail cannabis products
beverages
gummies
consumer brands
The other:
Clinical cannabinoid medicines
pharmaceutical formulations
FDA-approved therapies
History suggests that the second category ultimately commands far greater long-term value.
The Moment of Realization
When regulators finally draw a clear line between consumer cannabinoids and pharmaceutical cannabinoids, many executives may realize something uncomfortable:
They spent years building products that could disappear with a single regulatory action.
Meanwhile, companies that invested in clinical research may emerge as the long-term winners.
The Lesson for the Industry
Regulatory shortcuts rarely last forever.
Science-based medicine does.
And as federal oversight tightens, the companies that invested in clinical validation rather than gray-market loopholes will be the ones defining the next era of cannabinoid therapeutics.
CONTACT:
Madison Hisey
[email protected]
203-231-8583
SOURCE: MMJ International Holdings
View the original press release on ACCESS Newswire
P.Stevenson--AMWN
