
-
 Messi shines as Inter Miami finally win at new stadium
Messi shines as Inter Miami finally win at new stadium
-
Shai Gilgeous-Alexander wins second straight NBA MVP award

-
 White House mass prayer event seeks to reclaim US Christian roots
White House mass prayer event seeks to reclaim US Christian roots
-
International dive group joins Maldives search for missing Italians

-
 'Staggering' Iran toll drives up global executions: Amnesty
'Staggering' Iran toll drives up global executions: Amnesty
-
Rai wins first major at PGA with back-nine birdie blitz

-
 Woad bags second LPGA title at Queen City Championship
Woad bags second LPGA title at Queen City Championship
-
Lebanon says Israeli strikes kill 7 as Hezbollah condemns talks

-
 Revived La Rochelle trounce Top 14 leaders Toulouse
Revived La Rochelle trounce Top 14 leaders Toulouse
-
PSG beaten by Paris FC in Ligue 1 as Lille qualify for Champions League

-
 Griezmann apologetic on emotional Atletico Madrid farewell
Griezmann apologetic on emotional Atletico Madrid farewell
-
Raging Neymar forced off by refereeing error as Santos lose

-
 Sinner extends Masters tournament streak on home turf, eyes French Open
Sinner extends Masters tournament streak on home turf, eyes French Open
-
Canadian cruise passenger confirmed positive for hantavirus

-
 England see off gutsy France to clinch another Women's Six Nations
England see off gutsy France to clinch another Women's Six Nations
-
Sevilla safe despite Real Madrid defeat, Mallorca on brink

-
 UK police detail arrests after far-right rally and counter demo
UK police detail arrests after far-right rally and counter demo
-
Smalley tees off with PGA lead and stars in hot pursuit

-
 Trump issues dire warning to Iran to accept peace deal
Trump issues dire warning to Iran to accept peace deal
-
West Ham on brink of Premier League relegation, Man Utd seal third

-
 Bulgaria's Eurovision winner flies home to rapturous welcome
Bulgaria's Eurovision winner flies home to rapturous welcome
-
Starc takes four to keep Delhi alive in IPL

-
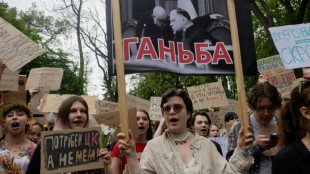 Kyiv residents protest 'dangerous' civil code, call for LGBTQ rights
Kyiv residents protest 'dangerous' civil code, call for LGBTQ rights
-
Modiba thunderbolt gives Sundowns victory in African final first leg

-
 World champions England see off France to clinch another Women's Six Nations
World champions England see off France to clinch another Women's Six Nations
-
Taiwan's leader says island will not be 'traded away'

-
 Sinner wins Italian Open, extends Masters tournament streak
Sinner wins Italian Open, extends Masters tournament streak
-
'Michael' moonwalks back to top of N. America box office

-
 Putter powers sizzling Kitayama to record 63 at PGA
Putter powers sizzling Kitayama to record 63 at PGA
-
Travolta channelled film greats in low-thrust plane movie

-
 Scotland rugby great Scott Hastings dead at 61 - SRU
Scotland rugby great Scott Hastings dead at 61 - SRU
-
Fujimori and Sanchez advance to Peru runoff: official results

-
 Italian PM meets victims of Modena car incident
Italian PM meets victims of Modena car incident
-
'Fight relentlessly': Ukraine commander vows strikes into Russia

-
 Kitayama fires sizzling 63 at PGA as No.1 Scheffler starts
Kitayama fires sizzling 63 at PGA as No.1 Scheffler starts
-
Fernandes equals Premier League assist record in Man Utd win, West Ham brace for Newcastle

-
 Ireland thrash Scotland 54-5 in Women's Six Nations to finish third
Ireland thrash Scotland 54-5 in Women's Six Nations to finish third
-
Vingegaard climbs to victory as Eulalio holds firm in pink

-
 Carrick expects clarity on Man Utd future in 'coming days'
Carrick expects clarity on Man Utd future in 'coming days'
-
Eyewitness says Modena tragedy could have been even worse

-
 Around 10 'new' victims in France's Epstein probe: prosecutor
Around 10 'new' victims in France's Epstein probe: prosecutor
-
Shock threat by billionaire Bollore's Canal+ group rocks French cinema

-
 Kohli, Venkatesh dazzle as Bengaluru qualify for IPL play-offs
Kohli, Venkatesh dazzle as Bengaluru qualify for IPL play-offs
-
Probes ongoing into alleged abuse at 84 Paris preschools: prosecutor

-
 Di Giannantonio wins Catalan MotoGP Grand Prix, Alex Marquez injured in horror crash
Di Giannantonio wins Catalan MotoGP Grand Prix, Alex Marquez injured in horror crash
-
Fernandes equals assist record as Man Utd edge Forest thriller

-
 Earps to leave PSG, in talks with London City Lionesses
Earps to leave PSG, in talks with London City Lionesses
-
Bowlers, Joy put Bangladesh on top in second Pakistan Test

-
 Alex Marquez injured in horrific Catalan MotoGP crash
Alex Marquez injured in horrific Catalan MotoGP crash
-
'Message for friends and foes': Libyan National Army conducts grand exercises


Biogen pulls controversial Alzheimer's drug Aduhelm
A controversial Alzheimer's drug that was trumpeted as the first to ever treat the cognitive decline associated with the devastating brain disorder has been pulled from the market, its maker Biogen announced Wednesday.
The US Food and Drug Administration awarded accelerated approval to Aduhelm in June 2021, a decision that was highly contentious at the time because the agency overruled its own independent advisors, who found there was insufficient evidence of benefit.
At least three of the 11-member independent committee that voted unanimously against recommending the drug subsequently resigned, and US congressional investigators slammed the accelerated approval as "rife with irregularities."
Biogen said it was discontinuing Aduhelm to put more resources into Leqembi, a newer Alzheimer's medicine that was fully approved last year under the traditional regulatory pathway.
"When searching for new medicines, one breakthrough can be the foundation that triggers future medicines to be developed," said Christopher Viehbacher, president and CEO of the Cambridge, Massachusetts-based biotech firm.
"Aduhelm was that groundbreaking discovery that paved the way for a new class of drugs and reinvigorated investments in the field."
Aduhelm, a monoclonal antibody that targets the build-up of a protein called amyloid beta in the brain tissue which is thought to be a cause of Alzheimer's, was tested in two late-stage human trials.
It showed a reduction in cognitive decline in one of the studies, but not the other.
According to a congressional report from December 2022, the FDA "considered Aduhelm under the traditional approval pathway used for most drugs for nine months, before abruptly changing course and granting approval under the accelerated approval pathway after a three-week review period."
The report said that FDA interactions with Biogen were "atypical" and included a failure to properly document contacts between agency staff and the drug maker.
The FDA and Biogen had also "inappropriately collaborated" on a joint briefing document for a key advisory committee, it said.
"FDA's approval process was rife with irregularities."
As for Biogen, the report said the company "viewed Aduhelm as an unprecedented financial opportunity -- estimating a potential peak revenue of $18 billion per year."
The congressional panel pointed to an "unjustifiably high price" for Aduhelm of $56,000 a year for patients.
Biogen's Leqembi, which it co-manufactures with Eisai of Japan, is now the only US approved treatment for Alzheimer's. It also targets amyloid beta and has been found to modestly reduce cognitive decline in patients with early stage disease.
Donanemab, developed by Eli Lilly, could be next to get the green light after performing similarly in clinical trials.
Alzheimer's is the most common form of dementia. More than one in nine people over 65 develop the condition, which worsens over time, robbing them of their memories and independence, according to the US Alzheimer's Association.
Ch.Kahalev--AMWN


