-
 Starc takes four to keep Delhi alive in IPL
Starc takes four to keep Delhi alive in IPL
-
Kyiv residents protest 'dangerous' civil code, call for LGBTQ rights
-
 Modiba thunderbolt gives Sundowns victory in African final first leg
Modiba thunderbolt gives Sundowns victory in African final first leg
-
World champions England see off France to clinch another Women's Six Nations

-
 Taiwan's leader says island will not be 'traded away'
Taiwan's leader says island will not be 'traded away'
-
Sinner wins Italian Open, extends Masters tournament streak

-
 'Michael' moonwalks back to top of N. America box office
'Michael' moonwalks back to top of N. America box office
-
Putter powers sizzling Kitayama to record 63 at PGA

-
 Travolta channelled film greats in low-thrust plane movie
Travolta channelled film greats in low-thrust plane movie
-
Scotland rugby great Scott Hastings dead at 61 - SRU

-
 Fujimori and Sanchez advance to Peru runoff: official results
Fujimori and Sanchez advance to Peru runoff: official results
-
Italian PM meets victims of Modena car incident

-
 'Fight relentlessly': Ukraine commander vows strikes into Russia
'Fight relentlessly': Ukraine commander vows strikes into Russia
-
Kitayama fires sizzling 63 at PGA as No.1 Scheffler starts

-
 Fernandes equals Premier League assist record in Man Utd win, West Ham brace for Newcastle
Fernandes equals Premier League assist record in Man Utd win, West Ham brace for Newcastle
-
Ireland thrash Scotland 54-5 in Women's Six Nations to finish third

-
 Vingegaard climbs to victory as Eulalio holds firm in pink
Vingegaard climbs to victory as Eulalio holds firm in pink
-
Carrick expects clarity on Man Utd future in 'coming days'

-
 Eyewitness says Modena tragedy could have been even worse
Eyewitness says Modena tragedy could have been even worse
-
Around 10 'new' victims in France's Epstein probe: prosecutor

-
 Shock threat by billionaire Bollore's Canal+ group rocks French cinema
Shock threat by billionaire Bollore's Canal+ group rocks French cinema
-
Kohli, Venkatesh dazzle as Bengaluru qualify for IPL play-offs

-
 Probes ongoing into alleged abuse at 84 Paris preschools: prosecutor
Probes ongoing into alleged abuse at 84 Paris preschools: prosecutor
-
Di Giannantonio wins Catalan MotoGP Grand Prix, Alex Marquez injured in horror crash

-
 Fernandes equals assist record as Man Utd edge Forest thriller
Fernandes equals assist record as Man Utd edge Forest thriller
-
Earps to leave PSG, in talks with London City Lionesses

-
 Bowlers, Joy put Bangladesh on top in second Pakistan Test
Bowlers, Joy put Bangladesh on top in second Pakistan Test
-
Alex Marquez injured in horrific Catalan MotoGP crash

-
 'Message for friends and foes': Libyan National Army conducts grand exercises
'Message for friends and foes': Libyan National Army conducts grand exercises
-
Bayern's Neuer sidelined again with leg issue

-
 Adam Driver shuts down question about clashes with Lena Dunham
Adam Driver shuts down question about clashes with Lena Dunham
-
British soprano Felicity Lott dies aged 79

-
 Roma near Champions League return with derby triumph, Napoli secure top four
Roma near Champions League return with derby triumph, Napoli secure top four
-
Denmark's Antonsen wins badminton Thailand Open title

-
 'Toxic' males Trump, Putin, Netanyahu to blame for wars, says star Bardem
'Toxic' males Trump, Putin, Netanyahu to blame for wars, says star Bardem
-
Iran have 'constructive' meeting with FIFA over World Cup preparations

-
 'Peaky Blinders' creator says he has licence to reinvent James Bond
'Peaky Blinders' creator says he has licence to reinvent James Bond
-
Xabi Alonso appointed Chelsea manager on four-year deal

-
 Mass Ukraine drone barrage kills 4 in Russia: Moscow
Mass Ukraine drone barrage kills 4 in Russia: Moscow
-
Gucci takes over New York's Times Square for fashion show

-
 Lyles says 'well worth the journey' after winning 100m in Tokyo
Lyles says 'well worth the journey' after winning 100m in Tokyo
-
Nepali duo break own records on Everest

-
 North Korean women footballers land in South ahead of rare match
North Korean women footballers land in South ahead of rare match
-
North Korean women footballers arrive in South Korea: AFP

-
 Rousey demolishes Carano in MMA comeback fight
Rousey demolishes Carano in MMA comeback fight
-
German 'chemical town' fears impact of industrial decline

-
 Qantas flight diverted after man bites flight attendant
Qantas flight diverted after man bites flight attendant
-
India scrambles to steady rupee as oil shock bites

-
 McGregor to make UFC return with Holloway rematch
McGregor to make UFC return with Holloway rematch
-
WHO declares international emergency as Ebola outbreak kills more than 80 in DR Congo


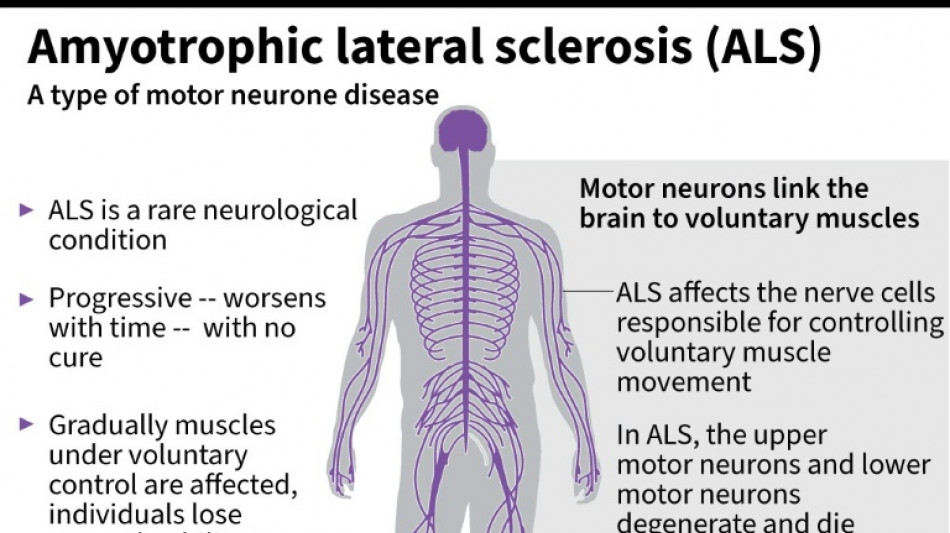
US company withdraws ALS drug after it fails in trial
Amylyx Pharmaceuticals announced Thursday it was withdrawing its approved treatment against the deadly neurodegenerative disease ALS after clinical data found no evidence the drug worked.
In a statement, the US company said it would discontinue its market authorizations for Relyvrio/Albrioza, using the brand names of the medicine in the US and Canadian markets.
"While this is a difficult moment for the ALS community, we reached this path forward in partnership with the stakeholders who will be impacted and in line with our steadfast commitment to people living with ALS and other neurodegenerative diseases," said the company's co-CEOs Joshua Cohen and Justin Klee in a statement.
The company also said it was reducing its workforce "by approximately 70 percent" as it focused on another experimental drug for use against ALS, and on repurposing Relyvrio for other conditions. It added it would continue to make Relyvrio available for patients who wish to keep using the treatment, through a "free drug program."
The news follows data from a clinical trial of 664 ALS patients announced in March, which found no significant differences in outcomes between those on the treatment group and those who received a placebo.
It was a big blow for patients with amyotrophic lateral sclerosis, sometimes called Lou Gehrig's disease after the famous baseball player, which devastates nerve cells in the brain and spinal cord.
ALS affects about two people per 100,000 every year, causing progressive loss of motor and cognitive function. Most patients die within five years of their diagnosis.
Relyvrio's approval by the US Food and Drug Administration in 2022 was controversial and based on the results of a single trial that involved just 137 participants.
The FDA itself noted there was "residual uncertainty about the evidence of effectiveness" -- but "given the serious and life-threatening nature of ALS and the substantial unmet need, this level of uncertainty is acceptable in this instance and consideration of these results in the context of regulatory flexibility is appropriate."
- Patient groups backed approval -
Advocacy groups also mounted a major campaign sending a petition to the FDA with tens of thousands of signatures urging approval. Once it became available, Amylyx reportedly announced an eye-watering list price of $158,000 per year in the US, drawing criticism.
Patient groups in Europe watched with desperation at the bureaucratic delays.
When the European Union drug watchdog later announced it was rejecting Relyvrio, the decision was slammed as "an affront" by angry French patients, who say they "don't have time to wait." France later relented, offering conditional approval in November.
"We commend Amylyx for pulling Relyvrio off the market, while still ensuring that people living with ALS can access the drug if they believe it is helping them," said the US-based ALS association, which had lobbied for the drug's approval and funded its research.
"Safe and potentially effective treatments can be made accessible rapidly until further research can confirm their efficacy," it added.
For now, there remain only a handful of treatments available.
Riluzole, FDA approved in 1995, prolongs life approximately three months. Edaravone, FDA approved in 2017, has been found to slow disease progression and improve survival.
And in 2023, the regulatory body approved tofersen, a gene therapy treatment that targets those ALS cases that are caused by mutations in the SOD1 gene.
Th.Berger--AMWN



