-
 Lyon a different proposition for Arsenal this time round, says Giraldez
Lyon a different proposition for Arsenal this time round, says Giraldez
-
Marc Marquez wins chaotic rain-affected Spanish MotoGP sprint

-
 Gunfire in Mali as army battles 'terrorist groups'
Gunfire in Mali as army battles 'terrorist groups'
-
Gunfire rocks Mali districts, including junta stronghold: witnesses

-
 Welsh football icon Ramsey takes on marathon challenge for charity
Welsh football icon Ramsey takes on marathon challenge for charity
-
Aussie Rules fires appeals chair over ruling on anti-gay slur

-
 Lakers' OT win puts Rockets on brink of NBA playoff elimination
Lakers' OT win puts Rockets on brink of NBA playoff elimination
-
From radiation to invasion: a Chernobyl worker's two wars

-
 AI firms flex lobbying muscle on both side of Atlantic
AI firms flex lobbying muscle on both side of Atlantic
-
First female Archbishop of Canterbury to meet Pope Leo

-
 Hundreds of firefighters battle Japan forest blazes
Hundreds of firefighters battle Japan forest blazes
-
Lakers down Rockets in overtime for 3-0 series lead, Celtics hold off Sixers

-
 US envoys heading to Pakistan for uncertain Iran talks
US envoys heading to Pakistan for uncertain Iran talks
-
'Hockey is religion': Montreal fans pack church for playoff push

-
 Billionaire Elon Musk enters courtroom showdown with OpenAI
Billionaire Elon Musk enters courtroom showdown with OpenAI
-
Crunch nuclear proliferation meeting at UN amid raging global wars

-
 Awkward debut for Trump at correspondents' dinner
Awkward debut for Trump at correspondents' dinner
-
Under blackout threat, Wikimedia reaches compromise with Indonesia

-
 'Going to the moon': Irish footballers return to China 50 years after historic tour
'Going to the moon': Irish footballers return to China 50 years after historic tour
-
Spurs' Wembanyama ruled out of game 3 after concussion

-
 Palestinians to vote in first elections since Gaza war
Palestinians to vote in first elections since Gaza war
-
Pragmatism, not patriotism, pushes young Lithuanians to military service

-
 Good Driver Club Redesigns Its Website and App to Bring Transparency into Clearer View
Good Driver Club Redesigns Its Website and App to Bring Transparency into Clearer View
-
Good Driver Club Publishes Eligible Events in Full Each Monday

-
 The Story Behind Good Driver Club: Why Good Drivers Deserve to Keep More
The Story Behind Good Driver Club: Why Good Drivers Deserve to Keep More
-
Group Seeking Court Order to Halt CMS Medicare THC Hemp Marijuana Program

-
 Peru confirms election runoff date, court says no to Lima re-vote
Peru confirms election runoff date, court says no to Lima re-vote
-
Venezuela, Colombia pledge military cooperation on first post-Maduro visit

-
 US hopes for progress, but Iran says not direct talks
US hopes for progress, but Iran says not direct talks
-
Maine governor nixes data center moratorium in state

-
 Betis's Bellerin further dents Real Madrid title hopes
Betis's Bellerin further dents Real Madrid title hopes
-
Lens rally but title bid fades after draw at Brest

-
 OpenAI CEO apologizes to Canada town for not reporting mass shooter
OpenAI CEO apologizes to Canada town for not reporting mass shooter
-
UK PM vows legislation to ban Iran Guards: report

-
 Leipzig tighten top-four grip as Union's Eta suffers second loss
Leipzig tighten top-four grip as Union's Eta suffers second loss
-
Furyk named USA captain for 2027 Ryder Cup

-
 EU, US sign critical minerals plan to counter China reliance
EU, US sign critical minerals plan to counter China reliance
-
The 'housewives' did well -- Ukraine takes drone know-how abroad

-
 Court removes US businessman from managing his Brazilian football team
Court removes US businessman from managing his Brazilian football team
-
'Natural' birth control risks unwanted pregnancy, experts warn

-
 No.2 Korda boosts LPGA Chevron lead to seven
No.2 Korda boosts LPGA Chevron lead to seven
-
EU trade chief seeks 'positive traction' on US steel tariffs

-
 Anthropic says Google to pump $40 bn into AI startup
Anthropic says Google to pump $40 bn into AI startup
-
Kohli makes Gujarat pay as Bengaluru cruise to IPL win

-
 One injured in bomb attack on Colombia military base
One injured in bomb attack on Colombia military base
-
Envoys from Iran, US expected in Pakistan for new talks

-
 ILO names US official as number two amid grumbling over unpaid dues
ILO names US official as number two amid grumbling over unpaid dues
-
Son of director Rob Reiner pays tribute to slain parents

-
 AI united Altman and Musk, then drove them apart
AI united Altman and Musk, then drove them apart
-
Sinner overcomes Bonzi in record hunt at Madrid Open


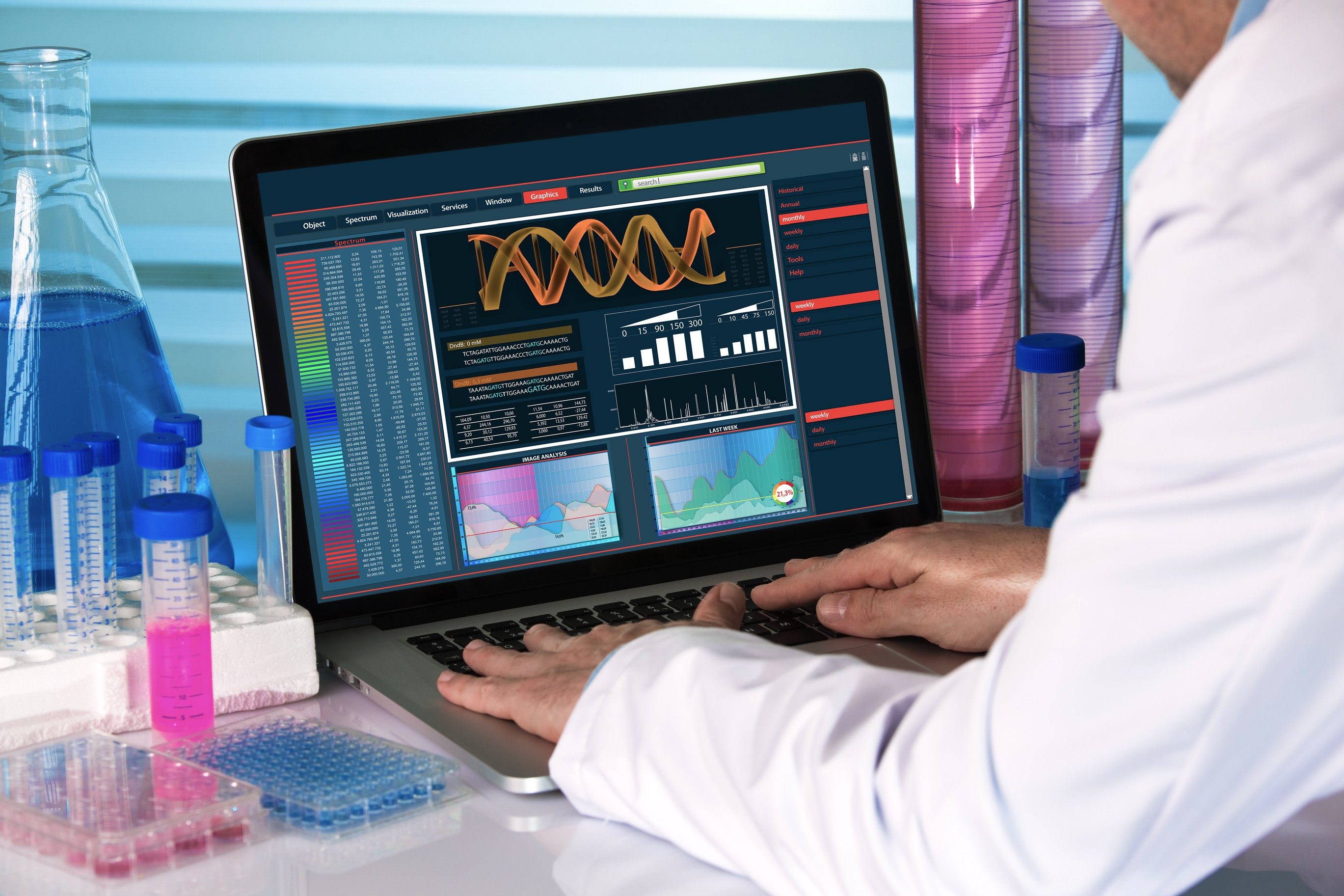
Adolore BioTherapeutics to Present at Festival of Biologics USA 2025
DELRAY BEACH, FL / ACCESS Newswire / April 23, 2025 / Adolore BioTherapeutics ("Adolore" or the "Company") is developing intracellularly-delivered neuromodulators that naturally reduce neuronal excitability to restore balance to the PNS/CNS to treat the underlying root cause of serious neurological disorders, including chronic pain and announced today Mr. Roelof Rongen, the Company's CEO, is a presenter at the Festival of Biologics USA 2025 meeting, "FOB", on April 24, 2025 in San Diego, Ca.
Adolore is delighted to present and highlight Adolore's innovative CA8* therapeutics in development (Kv7 activating carbonic anhydrase-like neuromodulating peptides, CA8* variants). Neuromodulation using gene therapy is an exciting new area that has widespread applications in treating serious undertreated neurological disorders including chronic pain. There is a paucity of safe, efficacious non-opioid analgesic treatments for chronic pain, creating a large and very urgent unmet medical need given the ongoing opioid crisis.
Adolore currently has two therapeutic programs in development, including peripheral neuropathy caused by erythromelalgia, which is an orphan disease, (ADB-101), and Adolore's lead program (ADB-102) for chronic pain caused by moderate-to-severe knee osteoarthritis. Based on compelling preclinical data, the Company is progressing these programs toward Investigational New Drug, ("IND"), filings and first-in-human clinical studies. These CA8* therapies were licensed in 2023 by Adolore from the University of Miami, Miami, FL. The rdHSV intracellular biotherapeutics delivery technology was licensed by Adolore from the University of Pittsburgh, in 2023.
The Company's neuromodulation development program for the treatment of chronic pain due to moderate-to-severe knee osteoarthritis is supported by the NIH/NINDS HEAL UH3 Award to the University of Miami that funds all formal pre-clinical GLP/GMP/GCP development work through a first-in-human clinical study in patients, which is expected to commence in 2026.
About Adolore BioTherapeutics, Inc.
Adolore BioTherapeutics, Inc., is focused on developing novel intracellularly delivered treatments for chronic pain, drug-resistant epilepsy, seizure disorders, neurodegeneration and neurotrauma/neuroprotection, e.g., hearing loss. The Company's chronic pain treatment platform comprises two highly innovative technologies: our CA8* analgesic peptides, and our cutting-edge, disease-free, nontoxic rdHSV vector-based intracellular biotherapeutics delivery technology. Our best-in-class programs are long-acting, localized gene-therapies that are opioid-free Disease Modifying Anti-Pain therapies (DMAPs).
The Company's two current CA8* DMAP programs are in preclinical development for treating patients suffering from erythromelalgia, a life-long heritable chronic pain condition representing an orphan drug disease with no approved therapy, and patients suffering with chronic pain due to moderate-to-severe knee osteoarthritis, which affects a large number of patients that are often treated with opioids due to the lack of good alternatives, thus contributing to the ongoing opioid crisis.
For more information, visit adolore.com.
Forward Looking Statements
To the extent this announcement contains information and statements that are not historical, they are considered forward-looking statements within the meaning of the federal securities laws. You can identify forward-looking statements by the use of the words "believe," "expect," "anticipate," "intend," "estimate," "project," "will," "should," "may," "plan," "intend," "assume" and other expressions which predict or indicate future events and trends and which do not relate to historical matters. You should not rely on forward-looking statements, because they involve known and unknown risks, uncertainties and other factors, some of which are beyond the control of the Company. These risks and uncertainties include, but are not limited to, those associated with drug development. These risks, uncertainties and other factors may cause the actual results, performance or achievements of the Company to be materially different from the anticipated future results, performance or achievements expressed or implied by the forward-looking statements.
Investor Relations Contact
Paul Barone
(215)622-4542
[email protected]
SOURCE: Adolore Biotherapeutics, Inc.
View the original press release on ACCESS Newswire
J.Oliveira--AMWN