
-
 From sun to subsoil, how countries are moving away from fossil fuels
From sun to subsoil, how countries are moving away from fossil fuels
-
London's Jewish community on edge amid attacks

-
 Ranieri's Roma role ends after spat with coach Gasperini: club
Ranieri's Roma role ends after spat with coach Gasperini: club
-
Warming El Nino set to return in mid-2026: UN

-
 Porsche exits sports car maker Bugatti Rimac
Porsche exits sports car maker Bugatti Rimac
-
Bill legalising assisted dying in England and Wales set to fail

-
 Chinese EVs, flying cars take centre stage at world's biggest auto show
Chinese EVs, flying cars take centre stage at world's biggest auto show
-
Macron says still sees France, Germany developing European fighter jet

-
 Al Ahli star Mahrez warns team-mates not to take Japanese rivals for granted
Al Ahli star Mahrez warns team-mates not to take Japanese rivals for granted
-
Greece expands sunbed-free beach list for 2026

-
 Rugby legend McCaw hails 'spectacular' NZ stadium built after deadly quake
Rugby legend McCaw hails 'spectacular' NZ stadium built after deadly quake
-
Mideast war drives up condom, rubber glove prices: manufacturers

-
 Gulf states in limbo as US-Iran crisis drags on
Gulf states in limbo as US-Iran crisis drags on
-
Liverpool's Slot warns 'margins are small' in Champions League push

-
 Musk says Tesla has started 'robotaxi' production
Musk says Tesla has started 'robotaxi' production
-
Suspected Nazi-looted Stradivarius reappears in France, says expert

-
 Glacier block delays route-setting on Everest
Glacier block delays route-setting on Everest
-
Appeal board says homophobia 'commonplace' in Aussie Rules

-
 Hot pants: Tokyo government workers swap suits for shorts
Hot pants: Tokyo government workers swap suits for shorts
-
Chinese EV makers take centre stage at world's biggest auto show

-
 Concern stirs Lula camp as election bid loses momentum
Concern stirs Lula camp as election bid loses momentum
-
China's top AI players

-
 Five things to know about Chinese AI startup DeepSeek
Five things to know about Chinese AI startup DeepSeek
-
Possible Trump rescue of Spirit Airlines spurs debate

-
 Wild Balkan berries keep gin taste steady as climate shifts
Wild Balkan berries keep gin taste steady as climate shifts
-
Mass MS-13 trial held at El Salvador mega-jail

-
 Barcelona must live without teen star Yamal for title run-in
Barcelona must live without teen star Yamal for title run-in
-
Hearts lead Old Firm as Scottish title race heads for tense finale

-
 India criticizes 'poor taste' Trump post against immigrants
India criticizes 'poor taste' Trump post against immigrants
-
China's DeepSeek says releases long-awaited new AI model

-
 Hawks fend off Knicks, Raptors pull away from Cavs to cut deficit
Hawks fend off Knicks, Raptors pull away from Cavs to cut deficit
-
Wildfires spread towards northern Japan town

-
 Israel, Lebanon extend ceasefire as Iran peace talks stall
Israel, Lebanon extend ceasefire as Iran peace talks stall
-
'Clearly me': AI drama accused of stealing faces

-
 Soviet architecture vanishes as Central Asia drifts from Moscow
Soviet architecture vanishes as Central Asia drifts from Moscow
-
Oil extends gains, stocks sink as peace talk hopes fade

-
 'Raw and honest': India climbers face obstacles in race to the top
'Raw and honest': India climbers face obstacles in race to the top
-
Cowgirls of Philippine rodeo tackle steers, stereotypes

-
 'Godzilla Minus Zero' will show monster up close, director says
'Godzilla Minus Zero' will show monster up close, director says
-
'Stigmatized' or 'sustainable'? Vintage sales boost sees fur return

-
 YouTube offers deepfake detection to Hollywood
YouTube offers deepfake detection to Hollywood
-
US soldier allegedly bet on Maduro operation using intel

-
 Bill to legalise assisted dying in England and Wales set to fail
Bill to legalise assisted dying in England and Wales set to fail
-
Arsenal eye return to top spot, Spurs fight for survival

-
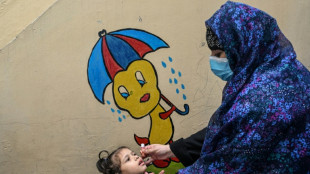 Child vaccine catch-up drive on course to hit target: UN
Child vaccine catch-up drive on course to hit target: UN
-
Chinese EVs geared up to dominate world's biggest auto show

-
 No.2 Korda fires 65 to grab LPGA Chevron lead
No.2 Korda fires 65 to grab LPGA Chevron lead
-
Raiders take quarterback Mendoza with No. 1 NFL draft pick

-
 Lebanon leaders accuse Israel of war crime after journalist killed
Lebanon leaders accuse Israel of war crime after journalist killed
-
Critical Minerals Such as Lithium and Rare Earth Elements Reach All-Time High Demand - Elektros Moves Forward with U.S. Lithium Refinery Search


Aspira Women's Health Provides Update on ARPA-H Sprint Program Partnership to Advance Women's Health
AUSTIN, TX AND WASHINGTON, DC / ACCESS Newswire / June 11, 2025 / Aspira Women's Health Inc., ("Aspira") (OTCQB:AWHL), an AI enhanced bio-analytics based women's health company focused on delivering leading noninvasive gynecologic disease diagnostic and disease management tools, announces an update from the Advanced Research Projects Agency for Health's ( ARPA-H ) Sprint for Women's Health program initiative to address critical unmet needs in women's health.
Aspira was originally awarded a $10 Million award through ARPA-H's Sprint for Women's Health to support the development of Aspira's ENDOinform test, a non-invasive blood draw-based multi-omics test utilizing protein and microRNA biomarkers, along with patient-specific data, to fuel a powerful, AI-powered algorithm to aid in the diagnosis of endometriosis in patients with symptoms of the disease.
This test, leveraging technology and expertise originally developed by Aspira for ovarian cancer risk assessment diagnostics, promises the potential to non-invasively confirm presence of suspected endometriosis in patients, enabling physicians to identify patients who may benefit from endometriosis-controlling therapeutics currently available.
The original ARPA-H contract was announced on October 24, 2024, and reflected $10 million divided into eight payments. The first two of those milestone payments, for $2 million and $1.5 million, were received on November 29, 2024, and March 28, 2025, respectively, for a total of $ 3.5 million from the ARPA-H program. On June 9, 2025, Aspira received notice from ARPA-H that ARPA-H and the assigned managing contractor, VentureWell, have determined that Aspira had not met the specifications of Milestone 3, and have therefore elected to terminate the ENDOinform development program contract.
Aspira's CEO, Mike Buhle, commented, "While we are certainly disappointed in ARPA-H's termination notice, we are highly confident that our team fully achieved all specifications requirements in the Milestone 3 scope of work provisions. We achieved these requirements in early May and are well underway in achieving Milestone 4 targeted specifications. We are confident in our quality of work, and even more so in our understanding of the technical requirements to complete the ENDOinform program."
Buhle further commented, "Our R&D team has been excited about the progress we have made, as well as the early indications of our potential success with the ENDOinform program overall. We plan to continue development of this critical women's health program with the help and support of our shareholders, our research & collaboration partners, and the ongoing dedication of our highly talented and missional teammates. ENDOinform promises a dramatic improvement in health outcomes and quality of life for millions of women suffering from endometriosis, both in the U.S. and abroad. The program remains targeted for 2026 completion goals. We look forward to continuing our development work to completion. In fact, compliance with the ARPA-H program involved significant time investment for our R&D team, and this may well offer us a material acceleration in the pace of achieving our goals."
About Aspira Women's Health Inc.
Aspira Women's Health Inc. is dedicated to the discovery, development, and commercialization of noninvasive, AI-powered tests to aid in the diagnosis of gynecologic diseases. OvaWatch® and Ova1Plus® are offered to clinicians as OvaSuiteSM. Together, they provide the only comprehensive portfolio of blood tests to aid in the detection of ovarian cancer risk for the 1.2+ million American women diagnosed with an adnexal mass each year.
OvaWatch provides a negative predictive value of 99% and is used to assess ovarian cancer risk for women where initial clinical assessment indicates the mass is indeterminate or benign, and thus surgery may be premature or unnecessary. Ova1Plus is a reflex process of two FDA-cleared tests, Ova1® and Overa®, to assess the risk of ovarian malignancy in women with an adnexal mass planned for surgery.
Our in-development test pipeline will expand our ovarian cancer portfolio and address the tremendous need for non-invasive diagnostics for endometriosis, a debilitating disease that impacts millions of women worldwide. In ovarian cancer, we intend to combine microRNA and protein biomarkers with patient data to further enhance the sensitivity and specificity of our current tests. In endometriosis, we have developed the first-ever non-invasive test designed to identify endometriomas, one of the most commonly occurring forms of severe endometriosis. Through our ongoing endometriosis development program, we are combining microRNA and protein biomarkers with patient data, with the intent of identifying all endometriosis independent of disease location or severity.
Forward-Looking Statements
This press release contains forward-looking statements, as defined in the Private Securities Litigation Reform Act of 1995. Forward-looking statements involve a number of risks and uncertainties. Such forward-looking statements include statements regarding, among other things, the timing and completion of any products in the development pipeline and other statements that are predictive in nature, and whether the marketing of the OvaSuite portfolio will prove successful. Actual results could differ materially from those discussed due to known and unknown risks, uncertainties, and other factors. These forward-looking statements generally can be identified by the use of words such as "designed to," "expect," "plan," "anticipate," "could," "may," "intend," "will," "continue," "future," and other words of similar meaning and the use of future dates. These and additional risks and uncertainties are described more fully in the Company's filings with the Securities and Exchange Commission (SEC), including those factors identified as "Risk Factors" in our most recent Annual Report on Form 10-K for the fiscal year ended December 31, 2024, and subsequent Quarterly Reports on Form 10-Q. If any of these risks materialize or our assumptions prove incorrect, actual results could differ materially from the results implied by these forward-looking statements. There may be additional risks that Aspira presently does not know, or that Aspira currently believes are immaterial, that could also cause actual results to differ from those contained in the forward-looking statements. In addition, forward-looking statements reflect Aspira's expectations, plans, or forecasts of future events and views as of the date of this press release. Subsequent events and developments may cause the Company's assessments to change. However, while Aspira may elect to update these forward-looking statements at some point in the future, Aspira expressly disclaims any obligation to do so, except as required by law. These forward-looking statements should not be relied upon as representing Aspira's assessments of any date after the date of this press release. Accordingly, undue reliance should not be placed upon the forward-looking statements.
Investor Relations Contact:
SOURCE: Aspira Women's Health
View the original press release on ACCESS Newswire
P.Santos--AMWN