
-
 Tourists trickle back to Kashmir, one year after deadly attack
Tourists trickle back to Kashmir, one year after deadly attack
-
Inside the world of ultra-luxury wedding cakes

-
 Chinese AI circuit board maker soars on Hong Kong debut
Chinese AI circuit board maker soars on Hong Kong debut
-
Oil prices dip, most stocks rise on lingering Iran peace hopes

-
 Tim Cook's time as Apple chief marked by profit absent awe
Tim Cook's time as Apple chief marked by profit absent awe
-
Mitchell, Harden shine as Cavs down Raptors for 2-0 series lead

-
 El Salvador's missing thousands buried by official indifference
El Salvador's missing thousands buried by official indifference
-
Trump's Fed chair pick to face lawmakers at key confirmation hearing

-
 PGA Tour to scrap Hawaii opening events from 2027
PGA Tour to scrap Hawaii opening events from 2027
-
Amazon invests another $5 bn in Anthropic

-
 Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
-
Wembanyama wins NBA defensive player of the year
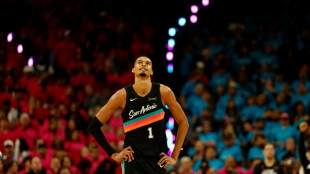
-
 'The Devil Wears Prada 2' stars reunite for glamorous premiere
'The Devil Wears Prada 2' stars reunite for glamorous premiere
-
El Salvador holds mass trial of nearly 500 alleged gang members

-
 Apple's Tim Cook to step down as CEO in September
Apple's Tim Cook to step down as CEO in September
-
West Ham's draw at Palace relegates Wolves, piles pressure on Spurs

-
 Canadian tourist killed in Mexico archaeological site shooting
Canadian tourist killed in Mexico archaeological site shooting
-
Wolves relegated from Premier League

-
 Oil jumps on Hormuz tensions, stocks mostly retreat
Oil jumps on Hormuz tensions, stocks mostly retreat
-
Colombian environmental activist honored amid threats and exile

-
 Gun battle traps more than 200 tourists at Rio viewpoint
Gun battle traps more than 200 tourists at Rio viewpoint
-
Alcaraz may skip French Open rather than rush injury comeback

-
 Top US court to hear case of Catholic schools excluded from state funding
Top US court to hear case of Catholic schools excluded from state funding
-
Trump Fed chair pick to vow interest rate independence at key hearing

-
 EU to host Taliban officials for talks on deporting Afghans
EU to host Taliban officials for talks on deporting Afghans
-
Blue Origin probing rocket's failure to deliver satellite

-
 Pope blasts 'exploitation' as he wraps up tour of Angola
Pope blasts 'exploitation' as he wraps up tour of Angola
-
Wembanyama 'changing the game as we speak', says Nowitzki

-
 Singer D4vd charged with murder after teen's body found in Tesla
Singer D4vd charged with murder after teen's body found in Tesla
-
Swiss football club turn down Kanye West concert approach

-
 Leicester fairytale turns sour as relegation to third tier looms
Leicester fairytale turns sour as relegation to third tier looms
-
Pope Leo blasts 'exploitation' as he wrap up tour of resource-rich Angola

-
 Varma ton revives Mumbai's IPL hopes with win over Gujarat
Varma ton revives Mumbai's IPL hopes with win over Gujarat
-
Formula One makes rule changes after drivers' criticism

-
 Singer D4vd charged with murder over teen's body found in Tesla
Singer D4vd charged with murder over teen's body found in Tesla
-
UK PM denies misleading MPs, says officials hid Mandelson info

-
 Tit-for-tat blockades once again cripple traffic in Hormuz
Tit-for-tat blockades once again cripple traffic in Hormuz
-
Cafu says 2026 World Cup is perfect time for Brazil to win again

-
 Erdogan vows new measures after deadly Turkey school shootings
Erdogan vows new measures after deadly Turkey school shootings
-
Rose to take charge at Bournemouth after Iraola exit

-
 Olympic status a massive 'boost' for squash says European champion Crouin
Olympic status a massive 'boost' for squash says European champion Crouin
-
Kenyan double-double as Korir, Lokedi defend Boston Marathon crowns

-
 Whale stranded on German coast swims off, gets stuck again
Whale stranded on German coast swims off, gets stuck again
-
Iran pulling Hormuz 'lever' to maximum in US standoff

-
 Argentine film and theater great Luis Brandoni dies at 86
Argentine film and theater great Luis Brandoni dies at 86
-
French Open sensation Boisson returns to action after 'most difficult' spell

-
 Desmond Morris: from 'Naked Ape' to watching 'Big Brother'
Desmond Morris: from 'Naked Ape' to watching 'Big Brother'
-
Rosenior says Chelsea owners supportive despite slump

-
 Oil jumps on Hormuz tensions, stocks retreat
Oil jumps on Hormuz tensions, stocks retreat
-
Romania legend Hagi eyes 'winning every game' on return as coach


IGC Pharma Expands CALMA Phase 2 Trial with Site at Lynn Health Science Institute in Oklahoma City
POTOMAC, MD / ACCESS Newswire / August 25, 2025 / IGC Pharma, Inc. (NYSE American:IGC) today announced the expansion of its ongoing Phase 2 clinical trial evaluating IGC-AD1, an investigational drug candidate for treating agitation in patients with Alzheimer's disease. The Company has added a clinical site at the Lynn Health Science Institute (LHSI) in Oklahoma City, Oklahoma.
Dr. Carl Griffin, MD, will serve as the Principal Investigator for the site. Dr. Griffin brings extensive experience in conducting clinical trials and a deep commitment to improving outcomes for patients affected by neurodegenerative diseases. He and the team at LHSI will begin recruiting eligible patients immediately.
"Expanding our clinical footprint to Oklahoma represents another important step toward reaching more patients and families affected by this devastating condition," said Ram Mukunda, CEO of IGC Pharma. "We are pleased to welcome Dr. Griffin and the Lynn Health Science Institute to the CALMA trial and look forward to their contributions in advancing our mission."
The CALMA trial is a randomized, double-blind, placebo-controlled Phase 2 study designed to evaluate the safety and efficacy of IGC-AD1 in reducing agitation in individuals diagnosed with Alzheimer's disease. Agitation is one of the most challenging and disruptive symptoms associated with Alzheimer's, often leading to earlier institutionalization and significant caregiver stress.
Lynn Health Science Institute is now actively enrolling participants. For more information, please contact:
Mikayla Dade
Clinical Research Coordinator - Lynn Health Science Institute
Email: [email protected]
Office: (405) 602-3939 ext. 106
To learn more about IGC Pharma and the CALMA trial, please visit www.igcpharma.com.
About Lynn Health Science Institute OKC
Lynn Health Science Institute (LHSI), located in Oklahoma City, Oklahoma, is a premier clinical research facility dedicated to advancing medical science through high-quality clinical trials. With over two decades of experience, LHSI partners with pharmaceutical and biotechnology companies to conduct Phase I-IV trials across a wide range of therapeutic areas. The institute is recognized for its commitment to patient care, data integrity, and research excellence. Under the leadership of seasoned investigators, LHSI strives to contribute to the development of innovative treatments that improve lives and strengthen the future of medicine.
About IGC Pharma (dba IGC):
IGC Pharma (NYSE American: IGC) is a clinical-stage biotechnology company leveraging AI to develop innovative treatments for Alzheimer's and metabolic disorders. Our lead asset, IGC-AD1, is a cannabinoid-based therapy currently in a Phase 2 trial (CALMA) for agitation in Alzheimer's dementia. Our pipeline includes TGR-63, targeting amyloid plaques, and early-stage programs focused on neurodegeneration, tau proteins, and metabolic dysfunctions. We integrate AI to accelerate drug discovery, optimize clinical trials, and enhance patient targeting. With 30 patent filings and a commitment to innovation, IGC Pharma is advancing breakthrough therapies.
Forward-Looking Statements:
This press release contains forward-looking statements. These forward-looking statements are based largely on IGC Pharma's expectations and are subject to several risks and uncertainties, certain of which are beyond IGC Pharma's control. Actual results could differ materially from these forward-looking statements as a result of, among other factors, the Company's failure or inability to commercialize one or more of the Company's products or technologies, including the products or formulations described in this release, or failure to obtain regulatory approval for the products or formulations, where required, or government regulations affecting AI or the AI algorithms not working as intended or producing accurate predictions; general economic conditions that are less favorable than expected; the FDA's general position regarding cannabis- and hemp-based products; and other factors, many of which are discussed in IGC Pharma's U.S. Securities and Exchange Commission ("SEC") filings. IGC incorporates by reference its Annual Report on Form 10-K filed with the SEC on June 27, 2025, as if fully incorporated and restated herein. Considering these risks and uncertainties, there can be no assurance that the forward-looking information contained in this release will occur. IGC Pharma, Inc. assumes no obligation to update forward-looking statements contained in this release as the result of new information or future events or developments.
Contact Information
Rosalyn Christian / John Nesbett
IMS Investor Relations
[email protected]
(203) 972-9200
SOURCE: IGC Pharma, Inc.
View the original press release on ACCESS Newswire
G.Stevens--AMWN

