
-
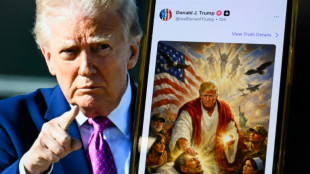 Trump deletes Jesus post of himself after outcry
Trump deletes Jesus post of himself after outcry
-
Lufthansa pilots strike as cabin crew call further stoppage

-
 SCANDIC COIN — цифровая валюта в рамках закрытой экосистемы
SCANDIC COIN — цифровая валюта в рамках закрытой экосистемы
-
Opposition candidate concedes defeat in Benin presidential election
-
 SCANDIC COIN, a digital currency within a closed ecosystem
SCANDIC COIN, a digital currency within a closed ecosystem
-
'Beautiful' battle with Sinner extra motivation: Alcaraz

-
 Szoboszlai says sorry to Liverpool fans after Man City incident
Szoboszlai says sorry to Liverpool fans after Man City incident
-
Goldman Sachs eyes more corporate mergers despite war uncertainty

-
 Star names inspiring Barca teen Yamal for Atletico comeback
Star names inspiring Barca teen Yamal for Atletico comeback
-
LVMH sales feel impact from war

-
 Satisfaction as Rolling Stones drop track under Cockroaches name
Satisfaction as Rolling Stones drop track under Cockroaches name
-
Serie A clubs endorse Milan-Cortina chief Malago as football federation president

-
 Liverpool need 'very special' night to stun PSG, says Slot
Liverpool need 'very special' night to stun PSG, says Slot
-
Russian, Belarusian swimmers free to compete under own flag

-
 Trump vows US will sink any Iran boats that challenge blockade
Trump vows US will sink any Iran boats that challenge blockade
-
Right-wing candidates tipped for runoff in Peru presidential poll

-
 Norwegian effectively cured of HIV after transplant from brother
Norwegian effectively cured of HIV after transplant from brother
-
French court gives teacher suspended sentence over pupil's suicide

-
 'No warning': Survivors say Nigerian air force bombed packed market
'No warning': Survivors say Nigerian air force bombed packed market
-
Pope says doesn't fear Trump, has 'moral duty to speak out' against war

-
 'No fun': French hospital confronts laughing gas abuse
'No fun': French hospital confronts laughing gas abuse
-
Pro-EU Magyar vows 'new era' in Hungary after ousting Orban in vote

-
 UK Taylor Swift dance party stabbing spree 'avoidable': inquiry
UK Taylor Swift dance party stabbing spree 'avoidable': inquiry
-
Iran releases assets of football captain in Australia asylum row

-
 French court jails Lafarge ex-CEO for funding IS in Syria
French court jails Lafarge ex-CEO for funding IS in Syria
-
Atletico need 'personality' to prevent Barca comeback: Koke

-
 Cameroon's Catholics divided on papal visit
Cameroon's Catholics divided on papal visit
-
South Africa's new DA leader vows to shed party's white image

-
 Karol G honors Latinos in Coachella headline performance: 'Feel proud'
Karol G honors Latinos in Coachella headline performance: 'Feel proud'
-
Pope's African tour begins in shadow of Trump ire

-
 'Help me!': family's anguish over Equatorial Guinean lured into Ukraine war
'Help me!': family's anguish over Equatorial Guinean lured into Ukraine war
-
Germany unveils 1.6 bn euro fuel price relief to tackle energy shock

-
 Ukraine loan, frozen funds: how could Orban's ouster unblock EU?
Ukraine loan, frozen funds: how could Orban's ouster unblock EU?
-
What next for Pogacar, Van der Poel after Roubaix blow?

-
 Orban loses Hungary vote to pro-Europe newcomer Magyar
Orban loses Hungary vote to pro-Europe newcomer Magyar
-
US says to begin blockade of Iranian ports

-
 Germany to cut fuel taxes amid Iran war energy shock
Germany to cut fuel taxes amid Iran war energy shock
-
Pope Leo kicks off African tour under shadow of Trump's ire

-
 Singer Luisa Sonza shares 'unique experience' of Coachella debut
Singer Luisa Sonza shares 'unique experience' of Coachella debut
-
Australia names Coyle first woman to lead army

-
 Rashford with point to prove as Barca target Atletico comeback
Rashford with point to prove as Barca target Atletico comeback
-
Iran executed at least 1,639 people in 2025, most since 1989: NGOs

-
 Nuggets roll into NBA playoffs, Raptors clinch berth
Nuggets roll into NBA playoffs, Raptors clinch berth
-
Flagg's sensational rookie season ends with injury

-
 Trump says 'not a big fan' of Pope Leo after his anti-war message
Trump says 'not a big fan' of Pope Leo after his anti-war message
-
Spain's Sanchez calls China trade imbalance with EU 'unsustainable'

-
 Oil surges, stocks fall as Trump says to blockade Strait of Hormuz
Oil surges, stocks fall as Trump says to blockade Strait of Hormuz
-
Rivers departing as Bucks coach after disappointing season

-
 Raptors top Nets, grab No. 5 seed on last day of NBA regular season
Raptors top Nets, grab No. 5 seed on last day of NBA regular season
-
Greece's ancient sites get climate-change checkup


IGC Pharma Expands Phase 2 CALMA Trial with New Clinical Trial Site at Integrative Clinical Trials in New York
- Multi-Specialty CNS Research Center Strengthens Trial Execution and Patient Diversity in Alzheimer's Agitation Study -
POTOMAC, MD / ACCESS Newswire / January 22, 2026 / IGC Pharma, Inc. (NYSE American:IGC) ("IGC" or the "Company"), a clinical-stage pharmaceutical company leveraging Artificial Intelligence ("AI") to develop innovative treatments for Alzheimer's disease, today announced the addition of Integrative Clinical Trials, LLC, located in Brooklyn, New York, as a new clinical site participating in the Company's Phase 2 CALMA clinical trial evaluating IGC-AD1 for the treatment of agitation associated with Alzheimer's disease.

Integrative Clinical Trials is a clinical research center conducting studies across a broad range of Central Nervous System ("CNS") disorders, including Alzheimer's disease and mild cognitive impairment. The site is led by Dr. Inna Yuryev-Golger, MD, who serves as Principal Investigator and brings more than 10 years of experience conducting clinical research. Bella Yevseva serves as the Clinical Trial Coordinator.
"Adding Integrative Clinical Trials further strengthens our CALMA clinical network with a site that has deep expertise across CNS disorders," said Ram Mukunda, CEO of IGC Pharma. "Their experience in neuropsychiatric and neurodegenerative research supports our commitment to high-quality data generation and efficient trial execution as we advance IGC-AD1."
Located at 2409 Ocean Avenue, Unit 1F, Brooklyn, New York, Integrative Clinical Trials expands IGC Pharma's presence in a major metropolitan area with access to a diverse patient population. The Company believes that incorporating sites with broad CNS experience is important to supporting enrollment and patient engagement.
IGC-AD1 is an investigational cannabinoid-based investigational therapy being evaluated in a randomized, double-blind, placebo-controlled Phase 2 clinical trial for agitation in patients with Alzheimer's disease. The continued expansion of the CALMA trial's clinical footprint reflects IGC Pharma's strategy to support enrollment and maintain rigorous clinical oversight.
To learn more about the trial and participation, visit https://igcpharma.com/igc-ad1-phase-ii/.
About IGC Pharma (dba IGC):
IGC Pharma (NYSE American:IGC) is a clinical-stage biotechnology company leveraging AI to develop innovative treatments for Alzheimer's and metabolic disorders. Our lead asset, IGC-AD1, is a cannabinoid-based therapy currently in a Phase 2 trial (CALMA) for agitation in Alzheimer's dementia. Our pipeline includes TGR-63, targeting amyloid plaques, and early-stage programs focused on neurodegeneration, tau proteins, and metabolic dysfunctions. We integrate AI to accelerate drug discovery, optimize clinical trials, and enhance patient targeting. With a complete patent portfolio and a commitment to innovation, IGC Pharma is advancing breakthrough therapies.
Forward-Looking Statements:
This press release contains forward-looking statements. These forward-looking statements are based largely on IGC Pharma's expectations and are subject to several risks and uncertainties, certain of which are beyond IGC Pharma's control. Actual results could differ materially from these forward-looking statements as a result of, among other factors, the Company's failure or inability to commercialize one or more of the Company's products or technologies, including the products or formulations described in this release, or failure to obtain regulatory approval for the products or formulations, where required, or government regulations affecting AI or the AI algorithms not working as intended or producing accurate predictions; general economic conditions that are less favorable than expected; the FDA's general position regarding cannabis- and hemp-based products; and other factors, many of which are discussed in IGC Pharma's U.S. Securities and Exchange Commission ("SEC") filings. IGC incorporates by reference its Annual Report on Form 10-K filed with the SEC on June 27, 2025, as if fully incorporated and restated herein. Considering these risks and uncertainties, there can be no assurance that the forward-looking information contained in this release will occur. IGC Pharma, Inc. assumes no obligation to update forward-looking statements contained in this release as the result of new information or future events or developments.
Contact Information:
Rosalyn Christian / John Nesbett
IMS Investor Relations
[email protected]
(203) 972-9200
SOURCE: IGC Pharma, Inc.
View the original press release on ACCESS Newswire
F.Bennett--AMWN


