
-
 'Shame on Hollywood': Cannes-winning writer rails at stance on Gaza
'Shame on Hollywood': Cannes-winning writer rails at stance on Gaza
-
Singaporean, Indian firms face criminal charges over Maryland bridge crash

-
 Arsenal's White out for rest of the season with knee injury
Arsenal's White out for rest of the season with knee injury
-
Germany wants to put TikTok 'in European hands'

-
 Rahm has faith LIV will develop good survival plan
Rahm has faith LIV will develop good survival plan
-
Sinner dismisses Pellegrino to reach Italian Open quarter-finals

-
 Sam Altman to testify at California tech titan trial
Sam Altman to testify at California tech titan trial
-
McIlroy has 'clear road ahead' to win more majors

-
 Rome derby row as authorities reschedule Serie A to avoid tennis clash
Rome derby row as authorities reschedule Serie A to avoid tennis clash
-
Georgia enthrones new leader of powerful Orthodox Church

-
 French court convicts VW for 'consumer harm' in 'Dieselgate' scandal
French court convicts VW for 'consumer harm' in 'Dieselgate' scandal
-
US consumer inflation hits three-year high fuelled by Iran war

-
 Cannes honours Jackson, Middle Earth wizard who 'transformed' cinema
Cannes honours Jackson, Middle Earth wizard who 'transformed' cinema
-
Vladimir Weiss returns as Slovakia coach

-
 Iran says US must accept peace plan or face 'failure'
Iran says US must accept peace plan or face 'failure'
-
UK PM Starmer defiant as calls to quit grow

-
 Spain coach counting on Yamal and Williams fitness for World Cup
Spain coach counting on Yamal and Williams fitness for World Cup
-
Guardiola says Man City 'still fighting' for Premier League title

-
 Singer FKA twigs to play Josephine Baker in biopic of anti-racist legend
Singer FKA twigs to play Josephine Baker in biopic of anti-racist legend
-
Flick extends contract with Barcelona

-
 Rana stars as Bangladesh down Pakistan in 1st Test thriller
Rana stars as Bangladesh down Pakistan in 1st Test thriller
-
Oil prices jump, stocks retreat on US-Iran deadlock

-
 South Korea official floats AI profit social tax as tech giants boom
South Korea official floats AI profit social tax as tech giants boom
-
Kremlin says no 'specifics' on ending Ukraine war despite Putin's words

-
 Vodafone sees signs of recovery amid turnaround plan
Vodafone sees signs of recovery amid turnaround plan
-
Ruud crushes Musetti to reach Italian Open quarters, Sinner awaits derby

-
 Japan Olympic official resigns after 'utterly unacceptable' remarks
Japan Olympic official resigns after 'utterly unacceptable' remarks
-
Australia's economy 'hostage' to Mideast war: treasurer

-
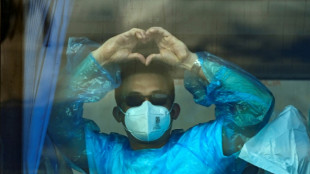 WHO chief says 'work not over' after hantavirus evacuation
WHO chief says 'work not over' after hantavirus evacuation
-
UK PM Starmer defiant as quit calls grow

-
 Indigenous Australians awarded major compensation in mining dispute
Indigenous Australians awarded major compensation in mining dispute
-
Bayer profit up but glyphosate sales struggle

-
 New London museum woos younger visitors
New London museum woos younger visitors
-
Japan crisp packs to go colourless due to Iran war crunch

-
 Mosquitoes: bloodsuckers and flower lovers
Mosquitoes: bloodsuckers and flower lovers
-
Russia, Ukraine end US-brokered truce with fresh attacks

-
 Over 370 Afghan civilians killed in Pakistan conflict in three months: UN
Over 370 Afghan civilians killed in Pakistan conflict in three months: UN
-
Japan Olympic official sorry for 'utterly unacceptable' remarks

-
 'Genuine urgency': China's underlying concerns at the Xi-Trump talks
'Genuine urgency': China's underlying concerns at the Xi-Trump talks
-
Bayer profit up on seed business but glyphosate sales struggle

-
 James undecided on future after Lakers bow out of NBA playoffs
James undecided on future after Lakers bow out of NBA playoffs
-
Japan baseball to punish dangerous swings after umpire hit

-
 Israel takes the stage in semis of boycotted Eurovision
Israel takes the stage in semis of boycotted Eurovision
-
Even DJs don't escape junta's 'revolution' in Burkina Faso

-
 Antarctic talks in Japan: key things to know
Antarctic talks in Japan: key things to know
-
Thyssenkrupp cuts sales outlook on Mideast war

-
 LeBron's Lakers eliminated from NBA playoffs as Thunder seal sweep
LeBron's Lakers eliminated from NBA playoffs as Thunder seal sweep
-
South Korea floats AI profit social tax as tech giants boom

-
 'Big hug' or colder shoulder? Xi-Trump talks spotlight contrasting styles, expectations
'Big hug' or colder shoulder? Xi-Trump talks spotlight contrasting styles, expectations
-
New Zealand moves to halt lawsuits over climate damage


enVVeno Medical Discusses FDA IDE Approval in Virtual Investor "What This Means" Segment
First-Ever FDA IDE Approval for a U.S. Pivotal Study of a Non-surgical Replacement Venous Valve
IDE approval positions enVVeno to advance what could become the first effective treatment option for the approximately 3 million U.S. patients suffering from DVI
Watch the "What This Means" video here
IRVINE, CA / ACCESS Newswire / May 12, 2026 / enVVeno Medical Corporation (Nasdaq:NVNO) ("enVVeno Medical" or the "Company"), today announced that Robert Berman, Chief Executive Officer of enVVeno Medical participated in a Virtual Investor "What This Means" segment.

As part of the segment, Mr. Berman discussed the Company's recent news announcing the U.S. Food and Drug Administration's (FDA) first-ever IDE approval awarded to enVVeno to proceed with a U.S. pivotal study of a non-surgical replacement venous valve.
The Virtual Investor "What This Means" segment featuring enVVeno Medical is now available here.
About enVVeno Medical Corporation
enVVeno Medical (NASDAQ:NVNO) is an Irvine, California-based, clinical-stage medical device Company focused on the advancement of innovative bioprosthetic (tissue-based) solutions to improve the standard of care for the treatment of deep venous disease. The Company is currently developing the enVVe system, which includes a first-in-class non-surgical replacement venous valve being developed for the treatment of severe Deep Venous Insufficiency (DVI). The enVVe valve is designed to act as one-way valve, to help assist in propelling blood up the leg, and back to the heart and lungs. As a transcatheter delivered device, the enVVe procedure will be performed under light sedation and should not require an over-night hospital stay.
Cautionary Note on Forward-Looking Statements
This press release and any statements of stockholders, directors, employees, representatives and partners of enVVeno Medical Corporation (the "Company") related thereto contain, or may contain, among other things, certain "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements involve significant risks and uncertainties. Such statements may include, without limitation, statements identified by words such as "projects," "may," "will," "could," "would," "should," "believes," "expects," "anticipates," "estimates," "intends," "plans," "potential" or similar expressions. These statements are based upon the current beliefs and expectations of the Company's management and are subject to significant risks and uncertainties, including those detailed in the Company's filings with the Securities and Exchange Commission. Actual results and timing (may differ significantly from those set forth or implied in the forward-looking statements. Forward-looking statements involve certain risks and uncertainties that are subject to change based on various factors (many of which are beyond the Company's control). The Company undertakes no obligation to publicly update any forward-looking statements, whether as a result of new information, future presentations or otherwise, except as required by applicable law.
INVESTOR CONTACT:
JTC Team, LLC
Jenene Thomas
[email protected]
(908) 824-0775
SOURCE: enVVeno Medical Corporation
View the original press release on ACCESS Newswire
G.Stevens--AMWN


