
-
 Inter close in on Serie A title by thumping Roma
Inter close in on Serie A title by thumping Roma
-
Trump makes foul-mouthed threat to Iran after US airman rescued

-
 Monaco sink Marseille for seventh Ligue 1 win in a row
Monaco sink Marseille for seventh Ligue 1 win in a row
-
Inter thump Roma to extend Serie A lead to nine points

-
 Lebanon's Christians mark Easter in solidarity with war-hit south
Lebanon's Christians mark Easter in solidarity with war-hit south
-
Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years

-
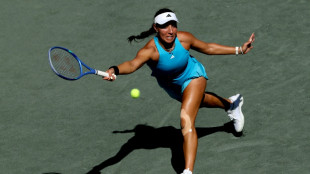 Pegula romps to WTA Charleston Open victory
Pegula romps to WTA Charleston Open victory
-
David six-hitting spree powers Bengaluru to IPL win

-
 Union draw leaves St Pauli stranded in Bundesliga drop zone
Union draw leaves St Pauli stranded in Bundesliga drop zone
-
UK police arrest protesters near base used by US

-
 Trump issues foul-mouthed threat to Iran after US airman rescued
Trump issues foul-mouthed threat to Iran after US airman rescued
-
Alcaraz plans to play full clay-court season, get 'socks dirty'

-
 'Super Mario Galaxy' blasts off in N. America box office debut
'Super Mario Galaxy' blasts off in N. America box office debut
-
Artemis astronauts begin fifth day on historic Moon mission

-
 Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup
Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup
-
Trump draws criticism with fiery Easter message on Iran

-
 OPEC+ hikes oil production quotas, issues warning
OPEC+ hikes oil production quotas, issues warning
-
British PM slams London event for booking Kanye West, sponsor quits

-
 Pogacar wins joint-record third Tour of Flanders
Pogacar wins joint-record third Tour of Flanders
-
Trump threatens 'hell' for Iran over Strait of Hormuz

-
 Shami, Pant help Lucknow beat Hyderabad in nervy IPL clash
Shami, Pant help Lucknow beat Hyderabad in nervy IPL clash
-
What we know about the race to rescue downed US airman in Iran

-
 US commandos went deep into Iran to rescue downed airman: media
US commandos went deep into Iran to rescue downed airman: media
-
Liberated McIlroy eyes more Masters magic after career Slam

-
 Van Dijk apologises for Liverpool thumping by Man City
Van Dijk apologises for Liverpool thumping by Man City
-
British PM slams London festival for booking Kanye West

-
 'Choose peace': Pope marks first Easter under cloud of Mideast war
'Choose peace': Pope marks first Easter under cloud of Mideast war
-
British royals attend Easter service without Andrew

-
 US media says commandos probed deep into Iran to rescue downed airman
US media says commandos probed deep into Iran to rescue downed airman
-
Revellers parade giant penises to dash stigma in Japan's fertility festival

-
 Artemis astronauts glimpse Moon's 'Grand Canyon' ahead of historic lunar flyby
Artemis astronauts glimpse Moon's 'Grand Canyon' ahead of historic lunar flyby
-
Middle East war hits Britain's fish and chip shops

-
 Artemis astronauts to study the Moon's surface using mainly their eyes
Artemis astronauts to study the Moon's surface using mainly their eyes
-
Second US airman downed over Iran 'SAFE and SOUND': Trump

-
 Indonesia lays to rest peacekeepers killed in Lebanon
Indonesia lays to rest peacekeepers killed in Lebanon
-
Pharmaceutical logistics in demand as war rattles supply chains

-
 Messi marks new stadium with goal but Miami held by Austin
Messi marks new stadium with goal but Miami held by Austin
-
Afghan mother seeks justice after Pakistani bombing kills hundreds

-
 UK royal family's dilemma over Andrew's daughters
UK royal family's dilemma over Andrew's daughters
-
Pope marks first Easter under cloud of Mideast war

-
 AI at war: Five things to know about Project Maven
AI at war: Five things to know about Project Maven
-
In the online 'maxxing' era, what's the deal with fiber and protein?

-
 At Met Opera, life after a school shooting takes center stage
At Met Opera, life after a school shooting takes center stage
-
Taiwan opposition leader to make 'peace' visit to China, first in 10 years

-
 McIlroy seeks rare Masters repeat in wide-open Augusta fight
McIlroy seeks rare Masters repeat in wide-open Augusta fight
-
Israel says will strike Lebanon-Syria border crossing

-
 Global Energy Shift Accelerates: Surging Gas Prices Drive Mass EV Adoption - Elektros Advances Patented Technology Aimed at Transforming Charging Efficiency
Global Energy Shift Accelerates: Surging Gas Prices Drive Mass EV Adoption - Elektros Advances Patented Technology Aimed at Transforming Charging Efficiency
-
Paul topples Tiafoe to book Houston ATP final against Burruchaga

-
 Jokic out-duels Wemby as Nuggets down Spurs in overtime
Jokic out-duels Wemby as Nuggets down Spurs in overtime
-
Trump gives Iran 48 hours to make deal, search for missing airman continues


Dermata Therapeutics Announces Presentation of Abstract at the European Academy of Dermatology and Venereology Congress 2025
- Abstract highlights additional primary and secondary data from Phase 3 STAR-1 clinical study of XYNGARI™ for the treatment of moderate-to-severe acne -
- Abstract highlights additional primary and secondary data from Phase 3 STAR-1 clinical study of XYNGARI™ for the treatment of moderate-to-severe acne -
SAN DIEGO, CA / ACCESS Newswire / September 17, 2025 / Dermata Therapeutics, Inc. (Nasdaq:DRMA)(Nasdaq:DRMAW) ("Dermata" or the "Company"), a science-driven leader in dermatologic solutions, today announced the presentation of an abstract from its XYNGARI™ (also known as DMT310) Phase 3 Spongilla Treatment of Acne Research clinical trial (STAR-1) will be presented at the European Academy of Dermatology and Venereology (EADV) Congress 2025 being held in Paris, France, from September 17-20th.
Details of the abstract are as follows:
Abstract Title: Once Weekly Topical Treatment with DMT310 Demonstrates Significant and Early Onset of Effect in Patients with Moderate to Severe Acne Vulgaris - Results from the STAR-1 Phase 3 Study
Abstract Number: P3243
The full abstract will be released on Tuesday, September 16, 2025, at 10:00pm (PST). For more information about the EADV Congress 2025, please visit https://eadv.org/congress/
Click here to view the EADV abstract.
Forward-Looking Statements
Statements in this press release that are not strictly historical in nature are forward-looking statements. These statements are based on the Company's current beliefs and expectations and new risks may emerge from time to time. Forward-looking statements are subject to known and unknown risks, uncertainties, assumptions and other factors including, but are not limited to, statements related to: Dermata's shift to prioritize OTC dermatology products; the anticipated benefits of the strategic shift; the anticipated benefits of Dermata's strategic shift, including acceleration of its path to commercialization, reduction of regulatory burdens, and expansion into broader consumer markets; the expected timing and success of any planned or future OTC product launches; risks that clinical trials may not be predictive of real-world results or of results of subsequent clinical trials; risks that current clinical trials will lead to further product development by the Company; and other factors described in the Company's filings with the Securities and Exchange Commission. These forward-looking statements are generally identified by the use of such words as "may," "could," "should," "would," "believe," "anticipate," "forecast," "estimate," "expect," "intend," "plan," "continue," "outlook," "will," "potential" and similar statements of a future or forward-looking nature. These statements are only predictions based on current information and expectations and involve a number of risks and uncertainties. Actual events or results may differ materially from those projected in any of such statements due to various factors, including the risks and uncertainties inherent in drug development, approval and commercialization, and the fact that past results of clinical trials may not be indicative of future trial results. For a discussion of these and other factors, please refer to Dermata's filings with the Securities and Exchange Commission. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof. This caution is made under the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. All forward-looking statements are qualified in their entirety by this cautionary statement and Dermata undertakes no obligation to revise or update this press release to reflect events or circumstances after the date hereof, except as required by law.
Dermata Investors:
Cliff Mastricola
Investor Relations
[email protected]
SOURCE: Dermata Therapeutics
View the original press release on ACCESS Newswire
O.Karlsson--AMWN