
-
 Lebanon's Christians mark Easter in solidarity with war-hit south
Lebanon's Christians mark Easter in solidarity with war-hit south
-
Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years

-
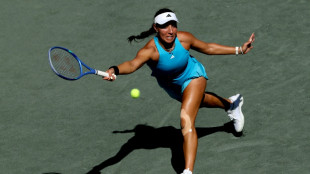 Pegula romps to WTA Charleston Open victory
Pegula romps to WTA Charleston Open victory
-
David six-hitting spree powers Bengaluru to IPL win

-
 Union draw leaves St Pauli stranded in Bundesliga drop zone
Union draw leaves St Pauli stranded in Bundesliga drop zone
-
UK police arrest protesters near base used by US

-
 Trump issues foul-mouthed threat to Iran after US airman rescued
Trump issues foul-mouthed threat to Iran after US airman rescued
-
Alcaraz plans to play full clay-court season, get 'socks dirty'

-
 'Super Mario Galaxy' blasts off in N. America box office debut
'Super Mario Galaxy' blasts off in N. America box office debut
-
Artemis astronauts begin fifth day on historic Moon mission

-
 Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup
Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup
-
Trump draws criticism with fiery Easter message on Iran

-
 OPEC+ hikes oil production quotas, issues warning
OPEC+ hikes oil production quotas, issues warning
-
British PM slams London event for booking Kanye West, sponsor quits

-
 Pogacar wins joint-record third Tour of Flanders
Pogacar wins joint-record third Tour of Flanders
-
Trump threatens 'hell' for Iran over Strait of Hormuz

-
 Shami, Pant help Lucknow beat Hyderabad in nervy IPL clash
Shami, Pant help Lucknow beat Hyderabad in nervy IPL clash
-
What we know about the race to rescue downed US airman in Iran

-
 US commandos went deep into Iran to rescue downed airman: media
US commandos went deep into Iran to rescue downed airman: media
-
Liberated McIlroy eyes more Masters magic after career Slam

-
 Van Dijk apologises for Liverpool thumping by Man City
Van Dijk apologises for Liverpool thumping by Man City
-
British PM slams London festival for booking Kanye West

-
 'Choose peace': Pope marks first Easter under cloud of Mideast war
'Choose peace': Pope marks first Easter under cloud of Mideast war
-
British royals attend Easter service without Andrew

-
 US media says commandos probed deep into Iran to rescue downed airman
US media says commandos probed deep into Iran to rescue downed airman
-
Revellers parade giant penises to dash stigma in Japan's fertility festival

-
 Artemis astronauts glimpse Moon's 'Grand Canyon' ahead of historic lunar flyby
Artemis astronauts glimpse Moon's 'Grand Canyon' ahead of historic lunar flyby
-
Middle East war hits Britain's fish and chip shops

-
 Artemis astronauts to study the Moon's surface using mainly their eyes
Artemis astronauts to study the Moon's surface using mainly their eyes
-
Second US airman downed over Iran 'SAFE and SOUND': Trump

-
 Indonesia lays to rest peacekeepers killed in Lebanon
Indonesia lays to rest peacekeepers killed in Lebanon
-
Pharmaceutical logistics in demand as war rattles supply chains

-
 Messi marks new stadium with goal but Miami held by Austin
Messi marks new stadium with goal but Miami held by Austin
-
Afghan mother seeks justice after Pakistani bombing kills hundreds

-
 UK royal family's dilemma over Andrew's daughters
UK royal family's dilemma over Andrew's daughters
-
Pope marks first Easter under cloud of Mideast war

-
 AI at war: Five things to know about Project Maven
AI at war: Five things to know about Project Maven
-
In the online 'maxxing' era, what's the deal with fiber and protein?

-
 At Met Opera, life after a school shooting takes center stage
At Met Opera, life after a school shooting takes center stage
-
Taiwan opposition leader to make 'peace' visit to China, first in 10 years

-
 McIlroy seeks rare Masters repeat in wide-open Augusta fight
McIlroy seeks rare Masters repeat in wide-open Augusta fight
-
Israel says will strike Lebanon-Syria border crossing

-
 Global Energy Shift Accelerates: Surging Gas Prices Drive Mass EV Adoption - Elektros Advances Patented Technology Aimed at Transforming Charging Efficiency
Global Energy Shift Accelerates: Surging Gas Prices Drive Mass EV Adoption - Elektros Advances Patented Technology Aimed at Transforming Charging Efficiency
-
Paul topples Tiafoe to book Houston ATP final against Burruchaga

-
 Jokic out-duels Wemby as Nuggets down Spurs in overtime
Jokic out-duels Wemby as Nuggets down Spurs in overtime
-
Trump gives Iran 48 hours to make deal, search for missing airman continues

-
 Lens' title push in Ligue 1 hit hard by Lille defeat
Lens' title push in Ligue 1 hit hard by Lille defeat
-
Arteta demands Arsenal response after FA Cup shocker at Southampton

-
 Barca move clear in La Liga as Real Madrid stumble
Barca move clear in La Liga as Real Madrid stumble
-
Lakers injury crisis deepens as Reaves out for regular season


Moderna Announces First Made-in-Canada mRNA Vaccines Delivered to Canadian Provinces and Territories
CAMBRIDGE, MA / ACCESS Newswire / September 19, 2025 / Moderna announced today that the first mRNA vaccines fully manufactured in Canada are being shipped to provinces and territories. This milestone advances Canada's domestic vaccine production capacity and strengthens national health security.
"The delivery of the first made-in-Canada mRNA vaccines to provinces and territories across the country marks a major milestone in our partnership with Canada," said Stéphane Bancel, Chief Executive Officer of Moderna. "This is not just about local manufacturing, it's about scientific leadership, national health security and ensuring Canadians have timely access to the latest mRNA medicines. We're proud to stand alongside Canada in advancing innovation, driving economic growth and investing in a resilient future for public health."
All drug substance of Moderna's authorized Spikevax® (COVID-19 mRNA vaccine) product targeting the SARS-CoV-2 LP.8.1 variant is manufactured at the facility in Laval, Quebec. Fill-and-finish of the new single-use pre-filled syringes is completed by Novocol Pharma in Cambridge, Ontario.
Moderna's updated COVID-19 vaccine targeting the LP.8.1 variant was approved by Health Canada on August 21, 2025 [1], and has also received authorization from regulators in the United States, Europe, Japan, Switzerland and several other countries. Additional regulatory submissions are under review globally in preparation for the 2025-2026 respiratory virus season.
Canadian Access and Eligibility
Timing and eligibility for the COVID-19 public vaccination program in Canada is set by each province and territory. Those who meet provincial criteria will receive COVID-19 vaccines free of charge. For individuals not covered under the public programs, efforts are ongoing with private insurers and payers to streamline access and reimbursement within the private sector. Canadians are encouraged to consult their provincial or territorial health authorities for the latest information on eligibility and availability.
Additional Quotes:
Stefan Raos, General Manager, Moderna Canada, said:
"As a Canadian, it's incredibly meaningful to see our first made-in-Canada COVID-19 vaccines in pre-filled syringes now reaching across the country to help protect Canadians starting this fall. This marks a critical shift, demonstrating that Canada's domestic manufacturing capability is real, operational and serving Canadians today. It reflects what's possible when science, industry and government work together with purpose."
The Honourable Mélanie Joly, Minister of Industry and Minister responsible for Canada Economic Development for Quebec Regions, said:
"Today's milestone of the production of the first made-in-Canada doses of Moderna's COVID-19 vaccine represents a major achievement for Canada's biomanufacturing and life sciences sector. Our government is building a resilient, forward-looking economy that positions Canada as a global leader in scientific research and innovation. Moderna's investments in Canada strengthen our national life sciences industry, create good-paying new jobs for Canadians and bolster our national security."
The Honourable Marjorie Michel, Minister of Health, said:
"Manufacturing the first-ever doses of a fully made-in-Canada COVID-19 vaccine demonstrates the power of collaboration between government, industry and science to help protect the health of Canadians. By producing vaccines here at home, we are supporting our economy while strengthening Canada's ability to respond quickly and effectively to future public health emergencies."
Images and b-roll can be found here.
[1] Moderna Canada. SPIKEVAX® Product Monograph. August 21, 2025.
About Moderna
Moderna is a leader in the creation of the field of mRNA medicine. Through the advancement of mRNA technology, Moderna is reimagining how medicines are made and transforming how we treat and prevent disease for everyone. By working at the intersection of science, technology and health for more than a decade, the company has developed medicines at unprecedented speed and efficiency, including one of the earliest and most effective COVID-19 vaccines.
Moderna's mRNA platform has enabled the development of therapeutics and vaccines for infectious diseases, immuno-oncology, rare diseases and autoimmune diseases. With a unique culture and a global team driven by the Moderna values and mindsets to responsibly change the future of human health, Moderna strives to deliver the greatest possible impact to people through mRNA medicines. For more information about Moderna, please visit modernatx.com and connect with us on X (formerly Twitter), Facebook, Instagram, YouTube and LinkedIn.
Spikevax® is a registered trademark of Moderna.
Moderna Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including statements regarding: the availability in Canadian provinces and territories of Moderna's updated COVID vaccine for the 2025-2026 vaccination season; Moderna's Canadian manufacturing; and Moderna's pending regulatory applications for approval of its COVID vaccine globally. The forward-looking statements in this press release are neither promises nor guarantees, and you should not place undue reliance on these forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond Moderna's control and which could cause actual results to differ materially from those expressed or implied by these forward-looking statements. These risks, uncertainties, and other factors include, among others, those risks and uncertainties described under the heading "Risk Factors" in Moderna's Annual Report on Form 10-K for the fiscal year ended December 31, 2024, and in subsequent filings made by Moderna with the U.S. Securities and Exchange Commission, which are available on the SEC's website at www.sec.gov. Except as required by law, Moderna disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this press release in the event of new information, future developments or otherwise. These forward-looking statements are based on Moderna's current expectations and speak only as of the date of this press release.
Moderna Contacts
Media:
Sacha Kennedy
Director, Communications and Media
+1 647-385-6846
[email protected]
Investors:
Lavina Talukdar
Senior Vice President & Head of Investor Relations
+1 617-209-5834
[email protected]
SOURCE: Moderna, Inc.
View the original press release on ACCESS Newswire
M.Fischer--AMWN