
-
 Sinner, defending champ Mensik advance to third round at Miami Open
Sinner, defending champ Mensik advance to third round at Miami Open
-
Iran missile strikes wound over 100 in two south Israel towns

-
 Shai hits 40 as Thunder win despite NBA melee with four ejected
Shai hits 40 as Thunder win despite NBA melee with four ejected
-
Records shattered as US heatwave moves eastward

-
 Iran missiles hit southern Israel, injuring more than 100
Iran missiles hit southern Israel, injuring more than 100
-
LeBron James breaks record for most NBA games played

-
 'Perfect' PSG sweep past Nice to reclaim top spot in Ligue 1
'Perfect' PSG sweep past Nice to reclaim top spot in Ligue 1
-
Japan coach says Asian Cup crown 'well-deserved' for inspirational team

-
 PSG sweep past Nice to reclaim top spot in Ligue 1
PSG sweep past Nice to reclaim top spot in Ligue 1
-
Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81

-
 Milan move to within five points of Serie A leaders Inter
Milan move to within five points of Serie A leaders Inter
-
Duplantis masterclass as Kerr and record-setter Ehammer shine

-
 Rosenior urges Chelsea to 'forget the noise' after damaging loss
Rosenior urges Chelsea to 'forget the noise' after damaging loss
-
Marquez ambushed Di Giannantonio to win Brazil sprint

-
 Sweden's Duplantis wins fourth world indoor pole vault title
Sweden's Duplantis wins fourth world indoor pole vault title
-
Liverpool, Chelsea slip up in Champions League race

-
 WHO sends first overland convoy from emergencies hub to Beirut
WHO sends first overland convoy from emergencies hub to Beirut
-
Everton rub salt in Chelsea wounds as Champions League race tightens

-
 Coach Mignoni returns but Toulon crash to Stade Francais
Coach Mignoni returns but Toulon crash to Stade Francais
-
Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81

-
 Sinner and Pegula advance to third round at Miami Open
Sinner and Pegula advance to third round at Miami Open
-
Britain's Kerr outsprints Hocker for world indoor 3,000m gold

-
 Kane backs Tuchel's call to rest him from England friendly
Kane backs Tuchel's call to rest him from England friendly
-
NBA fines 76ers' Drummond, Magic's Suggs $25,000 each

-
 Switzerland's Ehammer sets indoor heptathlon world record
Switzerland's Ehammer sets indoor heptathlon world record
-
Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set

-
 World Athletics decision to hand Asia two world indoors 'strategic' - Coe
World Athletics decision to hand Asia two world indoors 'strategic' - Coe
-
Trump threatens to use ICE agents for airport security control

-
 Kane moves closer to goals record as Bayern sink Union
Kane moves closer to goals record as Bayern sink Union
-
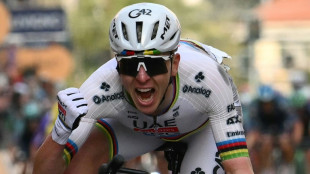
Pogacar ends long wait for Milan-San Remo glory after edging epic
-
 US says 'took out' Iran base threatening blocked Hormuz oil route
US says 'took out' Iran base threatening blocked Hormuz oil route
-
Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez

-
 Welbeck scores twice to dent Liverpool's top-five hopes
Welbeck scores twice to dent Liverpool's top-five hopes
-
US strikes Iran bases threatening blocked Hormuz oil route

-
 Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin
Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin
-
Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast

-
 K-pop kings BTS stun Seoul in '2.0' comeback concert
K-pop kings BTS stun Seoul in '2.0' comeback concert
-
French prosecutors suspect Musk encouraged deepfakes row to inflate X value

-
 Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa
Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa
-
Iranians mark Eid as Tehran reports strike on nuclear plant

-
 Kenya, Uganda open rail extension burdened by Chinese debt
Kenya, Uganda open rail extension burdened by Chinese debt
-
K-pop kings BTS rock Seoul in comeback concert

-
 Invincible Japan edge Australia to win Women's Asian Cup
Invincible Japan edge Australia to win Women's Asian Cup
-
Italy's Paris claims first win of season in World Cup downhill finale

-
 In Finland, divers learn to explore icy polar waters
In Finland, divers learn to explore icy polar waters
-
Dortmund extend injured captain Can's contract

-
 Iranians mark Eid as Trump mulls winding down war
Iranians mark Eid as Trump mulls winding down war
-
Matisse's last years cut out -- but not pasted -- at Paris expo

-
 BTS fans take over central Seoul for K-pop kings' comeback
BTS fans take over central Seoul for K-pop kings' comeback
-
Star jockey McDonald becomes horse racing's most prolific Group 1 winner


Tivic Health Reports Findings of Clinical Optimization Trial of Non-Invasive Cervical Vagus Nerve Stimulation (ncVNS)
The study demonstrated that personalizing the stimulation parameters to each user dramatically enhanced autonomic effects
FREMONT, CA / ACCESS Newswire / November 13, 2025 / Tivic Health Systems, Inc. (Nasdaq:TIVC), a diversified immunotherapeutics company, announced today it has optimized key device and treatment parameters including frequency, amplitude, electrode positioning, and duration of stimulation as a result of its collaborative study with The Feinstein Institutes for Medical Research at Northwell Health.
Physiological measurements taken before, during, and after ncVNS treatment found the following:
Personalizing the frequency of ncVNS stimulation to each study subject resulted in a 46% increase in heart rate variability, which was 8.9x more effective than applying the same frequency to all subjects
The optimal ncVNS frequency for each subject was found to be different between visits, indicating the importance of personalizing the stimulation parameters before each treatment
Certain placement and configurations of electrodes were found to be more effective than others, with some placements increasing heart rate variability and others markedly reduce heart rate variability
Four minutes of ncVNS stimulation was sufficient to drive a large increase in heart rate variability and was more effective than twenty minutes of stimulation.
While previous studies of non-invasive VNS devices have reported mixed results for autonomic nervous system changes, Tivic's study demonstrated that specific parameters could reproducibly impact outcomes.
"What makes our findings so compelling is that they advance the field of neurostimulation by underscoring the importance of stimulation frequency personalization, laterality and placement of stimulation electrodes, and duration of treatment," stated Jennifer Ernst, CEO of Tivic Health. "While these results were in healthy subjects, this data combined with our past studies suggest our ncVNS approach may have clinical utility in several very large patient populations including ischemic stroke, post-traumatic stress disorder, and inflammatory diseases, among others."
Ernst continued, "Our mission is to harness the immune system to improve clinical outcomes and save lives. We are energized by these results and we believe that this new data will enable us to unlock valuable new opportunities that can be developed either in house or with partners as funding is allocated across a growing pipeline of late-, mid- and early stage product candidates."
About Tivic Health Systems, Inc.
Tivic's dual platform utilizes the body's biopharmaceutical and bioelectronic systems to treat unmet medical needs through targeting the immune system.
Tivic's biologics compounds activate an innate immune pathway to prevent cell death in the bone marrow and epithelial tissues across systems impacted by radiation and age. The company's lead drug candidate, Entolimod™ for acute radiation syndrome, is a novel TLR5 agonist that has been granted Fast Track and Orphan Drug designations and is in late-stage development.
Tivic's bioelectronic program is developing a novel, non-invasive medical device designed to target the neural pathways implicated in many prevalent and debilitating diseases. Early trials show promising signals that Tivic's approach may regulate specific biologic responses, and the company believes its early-stage vagus nerve stimulation device has the potential to deliver clinical outcomes similar to or better than those of surgically implanted devices. To learn more about Tivic, visit: https://ir.tivichealth.com.
Forward-Looking Statements
This press release may contain "forward-looking statements" that are subject to substantial risks and uncertainties. All statements, other than statements of historical fact, contained in this press release are forward-looking statements. Forward-looking statements contained in this press release may be identified by the use of words such as "anticipate," "believe," "contemplate," "could," "estimate," "expect," "intend," "seek," "may," "might," "plan," "potential," "predict," "project," "target," "aim," "should," "will," "would," or the negative of these words or other similar expressions, although not all forward-looking statements contain these words. Forward-looking statements are based on Tivic Health Systems Inc.'s current expectations and are subject to inherent uncertainties, risks, and assumptions that are difficult to predict. Further, certain forward-looking statements are based on assumptions as to future events that may not prove to be accurate, including as a result of interactions with and guidance from the FDA and other regulatory authorities; changes to the company's relationship with the its partners; timing and success of clinical trials and study results; the indications for which the company's ncVNS approach may ultimately be found to have clinical utility based on additional studies and trials; the company's ability to unlock new opportunities for the further development of its ncVNS platform; the failure to obtain FDA or similar clearances or approvals and noncompliance with FDA or similar regulations; the company's future development of its ncVNS treatment, Entolimod and Entolasta; changes to the company's business strategy; regulatory requirements and pathways for approval; the company's ability to successfully commercialize its product candidates in the future; the potential opportunities that may be available to the company and its product candidates in the future; the company's need for, and ability to secure when needed, additional working capital; the company's ability to maintain its Nasdaq listing; and changes in tariffs, inflation, legal, regulatory, political and economic risks. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors. Accordingly, you are cautioned not to place undue reliance on such forward-looking statements. For a discussion of risks and uncertainties relevant to the company, and other important factors, see Tivic Health's filings with the SEC, including, its Annual Report on Form 10-K for the year ended December 31, 2024, filed with the SEC on March 21, 2025, under the heading "Risk Factors," as well as the company's subsequent filings with the SEC. Forward-looking statements contained in this press release are made as of this date, and Tivic Health Systems, Inc. undertakes no duty to update such information except as required by applicable law.
# # #
Investor Contact:
Hanover International, Inc.
[email protected]
Media Contact:
Deanne Eagle or Laura Min Jackson
[email protected]
SOURCE: Tivic Health Systems
View the original press release on ACCESS Newswire
A.Mahlangu--AMWN
