
-
 Milan move to within five points of Serie A leaders Inter
Milan move to within five points of Serie A leaders Inter
-
Duplantis masterclass as Kerr and record-setter Ehammer shine

-
 Rosenior urges Chelsea to 'forget the noise' after damaging loss
Rosenior urges Chelsea to 'forget the noise' after damaging loss
-
Marquez ambushed Di Giannantonio to win Brazil sprint

-
 Sweden's Duplantis wins fourth world indoor pole vault title
Sweden's Duplantis wins fourth world indoor pole vault title
-
Liverpool, Chelsea slip up in Champions League race

-
 WHO sends first overland convoy from emergencies hub to Beirut
WHO sends first overland convoy from emergencies hub to Beirut
-
Everton rub salt in Chelsea wounds as Champions League race tightens

-
 Coach Mignoni returns but Toulon crash to Stade Francais
Coach Mignoni returns but Toulon crash to Stade Francais
-
Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81

-
 Sinner and Pegula advance to third round at Miami Open
Sinner and Pegula advance to third round at Miami Open
-
Britain's Kerr outsprints Hocker for world indoor 3,000m gold

-
 Kane backs Tuchel's call to rest him from England friendly
Kane backs Tuchel's call to rest him from England friendly
-
NBA fines 76ers' Drummond, Magic's Suggs $25,000 each

-
 Switzerland's Ehammer sets indoor heptathlon world record
Switzerland's Ehammer sets indoor heptathlon world record
-
Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set

-
 World Athletics decision to hand Asia two world indoors 'strategic' - Coe
World Athletics decision to hand Asia two world indoors 'strategic' - Coe
-
Trump threatens to use ICE agents for airport security control

-
 Kane moves closer to goals record as Bayern sink Union
Kane moves closer to goals record as Bayern sink Union
-
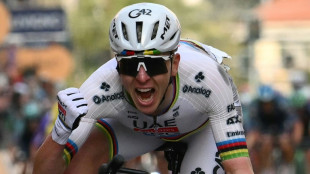
Pogacar ends long wait for Milan-San Remo glory after edging epic
-
 US says 'took out' Iran base threatening blocked Hormuz oil route
US says 'took out' Iran base threatening blocked Hormuz oil route
-
Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez

-
 Welbeck scores twice to dent Liverpool's top-five hopes
Welbeck scores twice to dent Liverpool's top-five hopes
-
US strikes Iran bases threatening blocked Hormuz oil route

-
 Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin
Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin
-
Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast

-
 K-pop kings BTS stun Seoul in '2.0' comeback concert
K-pop kings BTS stun Seoul in '2.0' comeback concert
-
French prosecutors suspect Musk encouraged deepfakes row to inflate X value

-
 Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa
Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa
-
Iranians mark Eid as Tehran reports strike on nuclear plant

-
 Kenya, Uganda open rail extension burdened by Chinese debt
Kenya, Uganda open rail extension burdened by Chinese debt
-
K-pop kings BTS rock Seoul in comeback concert

-
 Invincible Japan edge Australia to win Women's Asian Cup
Invincible Japan edge Australia to win Women's Asian Cup
-
Italy's Paris claims first win of season in World Cup downhill finale

-
 In Finland, divers learn to explore icy polar waters
In Finland, divers learn to explore icy polar waters
-
Dortmund extend injured captain Can's contract

-
 Iranians mark Eid as Trump mulls winding down war
Iranians mark Eid as Trump mulls winding down war
-
Matisse's last years cut out -- but not pasted -- at Paris expo

-
 BTS fans take over central Seoul for K-pop kings' comeback
BTS fans take over central Seoul for K-pop kings' comeback
-
Star jockey McDonald becomes horse racing's most prolific Group 1 winner

-
 Israel strikes Tehran, Beirut as Trump mulls 'winding down' war
Israel strikes Tehran, Beirut as Trump mulls 'winding down' war
-
Pistons top Warriors to clinch NBA playoff berth

-
 Tickets to toothbrushes: BTS's money-making machine
Tickets to toothbrushes: BTS's money-making machine
-
Top-ranked Alcaraz, Sabalenka win Miami openers

-
 After Cuba beckons, Miami entrepreneurs are mostly reluctant to invest in the island
After Cuba beckons, Miami entrepreneurs are mostly reluctant to invest in the island
-
Peru's crowded presidential race zeroes in on organized crime

-
 Taiwan's Lin to compete in first international event since Paris gender row
Taiwan's Lin to compete in first international event since Paris gender row
-
BTS takes over central Seoul for comeback concert

-
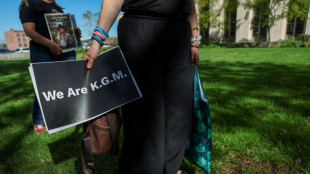 Jury signals tech titans on hook for social media addiction
Jury signals tech titans on hook for social media addiction
-
Brumbies mark Slipper record in thriller against Chiefs


Ensysce Biosciences Secures $4 Million Financing, Unlocking Up to $20 Million in Total Financing to Advance Breakthrough Pain Programs
~Funding supports Phase 3 clinical development of PF614 and continued innovation in abuse- and overdose-resistant analgesics~
SAN DIEGO, CA / ACCESS Newswire / November 17, 2025 / Ensysce Biosciences, Inc. (NASDAQ:ENSC)("Ensysce" or the "Company"), pioneering novel solutions for severe pain with built-in abuse and overdose protection, today announced the closing of a $4 million convertible preferred stock financing, with upside to potentially $16 million of additional funding available through future tranches over the next 24 months.
The capital will accelerate the Phase 3 clinical program for PF614, Ensysce's flagship analgesic candidate, while supporting general corporate initiatives. The initial $4 million tranche includes a fixed conversion price of $2.50 per share, with an alternate conversion price based upon average common stock prices prior to conversion. The financing includes 50% warrant coverage on each takedown, exercisable at the fixed conversion price for five years, subject to adjustment.
Key Highlights:
$4 Million Closed Now, $16 Million Available: Providing funding flexibility to advance critical programs.
Phase 3 Acceleration: PF614 clinical trials moving toward market readiness within 18-24 months.
Investor Confidence: Current backers continue to support Ensysce's mission-driven innovation.
Dr. Kirkpatrick, CEO of Ensysce Biosciences, stated, "This financing demonstrates strong investor confidence in Ensysce's vision: safe, effective analgesics that re-invent pain management. With PF614 on track for Phase 3 completion in the next 18 months and federal grant support for our MPAR program, we are moving closer to delivering transformative treatments for patients in urgent need."
About Ensysce Biosciences
Ensysce Biosciences is a clinical stage company with a goal of disrupting the analgesic landscape by introducing a new class of highly novel opioids for the treatment of severe pain. Leveraging its Trypsin-Activated Abuse Protection (TAAPTM) and Multi-Pill Abuse Resistance (MPAR®) platforms, the Company is developing unique, tamper-proof treatment options for pain that minimize the risk of both drug abuse and overdose. Ensysce's products are anticipated to provide safer options to treat patients suffering from severe pain and assist in preventing deaths caused by medication abuse. Ensysce's pipeline is backed by a robust global intellectual property portfolio, offering hope to patients and providers confronting the challenges of pain management. Learn more at www.ensysce.com.
Forward-Looking Statements
Statements contained in this press release that are not purely historical may be deemed to be forward-looking statements for the purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995 and other federal securities laws. Without limiting the foregoing, the use of words such as "may," "intends," "can," "might," "will," "expect," "plan," "possible," "believe" and other similar expressions are intended to identify forward-looking statements. The product candidates discussed are in clinic and not approved and there can be no assurance that the clinical programs will be successful in demonstrating safety and/or efficacy, that Ensysce will not encounter problems or delays in clinical development, or that any product candidate will ever receive regulatory approval or be successfully commercialized. All forward-looking statements are based on estimates and assumptions by Ensysce's management that, although Ensysce believes to be reasonable, are inherently uncertain. All forward-looking statements are subject to risks and uncertainties that may cause actual results to differ materially from those that Ensysce expected. In addition, Ensysce's business is subject to additional risks and uncertainties, including among others, the initiation and conduct of preclinical studies and clinical trials; the timing and availability of data from preclinical studies and clinical trials; expectations for regulatory submissions and approvals; potential safety concerns related to, or efficacy of, Ensysce's product candidates; the availability or commercial potential of product candidates; the ability of Ensysce to fund its continued operations, including its planned clinical trials; the dilutive effect of stock issuances from our fundraising; and Ensysce's and its partners' ability to perform under their license, collaboration and manufacturing arrangements. These statements are also subject to a number of material risks and uncertainties that are described in Ensysce's most recent quarterly report on Form 10-Q and current reports on Form 8-K, available free of charge at the SEC's website at www.sec.gov. Any forward-looking statement speaks only as of the date on which it was made. Ensysce undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except as required under applicable law.
Ensysce Biosciences Company Contact:
Lynn Kirkpatrick, Ph.D.
Chief Executive Officer
(858) 263-4196
Ensysce Biosciences Investor Relations Contact:
Shannon Devine
MZ North America
Main: 203-741-8811
[email protected]
SOURCE: Ensysce Biosciences Inc.
View the original press release on ACCESS Newswire
Y.Aukaiv--AMWN
