
-
 Coach Mignoni returns but Toulon crash to Stade Francais
Coach Mignoni returns but Toulon crash to Stade Francais
-
Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81

-
 Sinner and Pegula advance to third round at Miami Open
Sinner and Pegula advance to third round at Miami Open
-
Britain's Kerr outsprints Hocker for world indoor 3,000m gold

-
 Kane backs Tuchel's call to rest him from England friendly
Kane backs Tuchel's call to rest him from England friendly
-
NBA fines 76ers' Drummond, Magic's Suggs $25,000 each

-
 Switzerland's Ehammer sets indoor heptathlon world record
Switzerland's Ehammer sets indoor heptathlon world record
-
Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set

-
 World Athletics decision to hand Asia two world indoors 'strategic' - Coe
World Athletics decision to hand Asia two world indoors 'strategic' - Coe
-
Trump threatens to use ICE agents for airport security control

-
 Kane moves closer to goals record as Bayern sink Union
Kane moves closer to goals record as Bayern sink Union
-
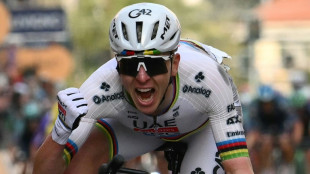
Pogacar ends long wait for Milan-San Remo glory after edging epic
-
 US says 'took out' Iran base threatening blocked Hormuz oil route
US says 'took out' Iran base threatening blocked Hormuz oil route
-
Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez

-
 Welbeck scores twice to dent Liverpool's top-five hopes
Welbeck scores twice to dent Liverpool's top-five hopes
-
US strikes Iran bases threatening blocked Hormuz oil route

-
 Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin
Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin
-
Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast

-
 K-pop kings BTS stun Seoul in '2.0' comeback concert
K-pop kings BTS stun Seoul in '2.0' comeback concert
-
French prosecutors suspect Musk encouraged deepfakes row to inflate X value

-
 Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa
Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa
-
Iranians mark Eid as Tehran reports strike on nuclear plant

-
 Kenya, Uganda open rail extension burdened by Chinese debt
Kenya, Uganda open rail extension burdened by Chinese debt
-
K-pop kings BTS rock Seoul in comeback concert

-
 Invincible Japan edge Australia to win Women's Asian Cup
Invincible Japan edge Australia to win Women's Asian Cup
-
Italy's Paris claims first win of season in World Cup downhill finale

-
 In Finland, divers learn to explore icy polar waters
In Finland, divers learn to explore icy polar waters
-
Dortmund extend injured captain Can's contract

-
 Iranians mark Eid as Trump mulls winding down war
Iranians mark Eid as Trump mulls winding down war
-
Matisse's last years cut out -- but not pasted -- at Paris expo

-
 BTS fans take over central Seoul for K-pop kings' comeback
BTS fans take over central Seoul for K-pop kings' comeback
-
Star jockey McDonald becomes horse racing's most prolific Group 1 winner

-
 Israel strikes Tehran, Beirut as Trump mulls 'winding down' war
Israel strikes Tehran, Beirut as Trump mulls 'winding down' war
-
Pistons top Warriors to clinch NBA playoff berth

-
 Tickets to toothbrushes: BTS's money-making machine
Tickets to toothbrushes: BTS's money-making machine
-
Top-ranked Alcaraz, Sabalenka win Miami openers

-
 After Cuba beckons, Miami entrepreneurs are mostly reluctant to invest in the island
After Cuba beckons, Miami entrepreneurs are mostly reluctant to invest in the island
-
Peru's crowded presidential race zeroes in on organized crime

-
 Taiwan's Lin to compete in first international event since Paris gender row
Taiwan's Lin to compete in first international event since Paris gender row
-
BTS takes over central Seoul for comeback concert

-
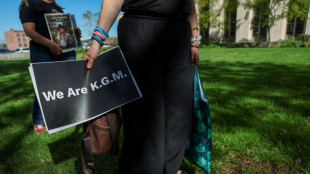 Jury signals tech titans on hook for social media addiction
Jury signals tech titans on hook for social media addiction
-
Brumbies mark Slipper record in thriller against Chiefs

-
 US jury finds Elon Musk misled Twitter shareholders
US jury finds Elon Musk misled Twitter shareholders
-
New to The Street Announces Broadcast of Show #739 on Bloomberg Television Across the U.S. at 6:30 PM EST

-
 SMX Reinforces Trust, Traceability, and Market Value Across Rare Earths and Precious Metals
SMX Reinforces Trust, Traceability, and Market Value Across Rare Earths and Precious Metals
-
SMX Redefines Trust, Provenance, and Transparency in the Global Luxury Market

-
 SMX Establishes a New Framework for Verification and Visibility Across Global Energy Supply Chains
SMX Establishes a New Framework for Verification and Visibility Across Global Energy Supply Chains
-
Gauff rallies to avance at Miami Open

-
 WNBA, players union confirm agreement on 'groundbreaking' labor deal
WNBA, players union confirm agreement on 'groundbreaking' labor deal
-
Carrick 'baffled' by inconsistent penalty calls as Man Utd held


Altamira Therapeutics Announces Certification of Bentrio Nasal Spray under Medical Device Regulation
Conformity with stringent new EU regulations for medical devices confirmed
Allows for commercialization of Bentrio in EU member states; facilitates regulatory approval in various other countries
Major milestone in international growth strategy
HAMILTON, BERMUDA / ACCESS Newswire / November 20, 2025 / Altamira Therapeutics Ltd. ("Altamira" or the "Company") (OTCQB:CYTOF), a company dedicated to developing and supplying nucleic acid delivery technology for partners in biotech and pharma, today announced that its associate company Altamira Medica ("Medica") obtained certification of its Bentrio® nasal spray under the European Union's Medical Device Regulation (MDR). Bentrio is a drug-free, preservative-free nasal spray for protection against airborne allergens which is commercialized by Medica through a network of marketing and distribution partners.
"The MDR certification represents a major milestone for our innovative Bentrio nasal spray as it is a prerequisite for commercializing the product in the European Union and beyond," commented Thomas Meyer, Medica's Chairman and CEO. "The certification declares Bentrio's conformity with the EU's new rigorous requirements for demonstrating and ensuring the safety, performance and quality of medical devices. Further, it serves as a key reference for certain regulatory agencies outside the EU, thus facilitating marketing clearance in various countries. We look forward to moving forward with our growth strategy with Bentrio in close collaboration with our international marketing and distribution partners."
The EU started to implement the MDR framework in 2021, thus gradually replacing the Medical Device Directive (MDD). Bentrio was launched in certain EU markets in 2021 as a "Class I" medical device under MDD, based on a self-declaration of conformity. Under MDR, nasal sprays such as Bentrio are considered "Class IIa" medical devices, implying additional regulatory requirements. The new regulations, together with the up-classification, required substantial efforts and investments by Medica, including the conduct of clinical trials and independent certification by a designated Notified Body.
About Bentrio
Bentrio is an "over the counter" drug-free nasal spray for personal protection against airborne allergens and, where approved, against airborne viruses. Upon application into the nose, Bentrio forms a protective gel layer on the nasal mucosa. This thin film is designed to prevent the contact of airborne particles with cells; in addition, the composition serves to bind such particles and help with their discharge. The efficacy and safety of Bentrio have been demonstrated in a total of four clinical trials, of which the largest one ("NASAR" study) enrolled 100 patients suffering from seasonal allergic rhinitis. In NASAR, participants self-administered either Bentrio or saline nasal spray for two weeks 3 times per day. The study showed a statistically significant reduction in the mean daily reflective Total Nasal Symptom Score (rTNSS) for Bentrio compared to saline (p = 0.013), as well as a statistically highly significant improvement in health-related quality of life (Rhinoconjunctivitis Quality of Life Questionnaire, p www.bentrio.com
About Altamira Therapeutics
Altamira Therapeutics is developing and supplying peptide-based nanoparticle technologies for efficient nucleic acid delivery (xPhore™ platform). The versatile delivery platform is suited for different nucleic acid modalities, including siRNA, mRNA, circRNA, as well as DNA, and made available to pharma or biotech companies through out-licensing. The Company has two proprietary flagship programs based on xPhore and siRNA payloads: AM-401 for KRAS driven cancer and AM-411 for rheumatoid arthritis, both in preclinical development beyond in vivo proof of concept. In addition, Altamira holds a 49% stake (with additional economic rights) in Altamira Medica AG, which owns its commercial-stage legacy asset Bentrio, an OTC nasal spray for allergic rhinitis. Further, the Company is in the process of partnering / divesting its inner ear legacy assets. Founded in 2003, Altamira is headquartered in Hamilton, Bermuda, with its main operations in Basel, Switzerland. For more information, visit: https://altamiratherapeutics.com/
Forward-Looking Statements
This press release may contain statements that constitute "forward-looking statements" within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Forward-looking statements are statements other than historical facts and may include statements that address future operating, financial or business performance or Altamira's strategies or expectations. In some cases, you can identify these statements by forward-looking words such as "may", "might", "will", "should", "expects", "plans", "anticipates", "believes", "estimates", "predicts", "projects", "potential", "outlook" or "continue", or the negative of these terms or other comparable terminology. Forward-looking statements are based on management's current expectations and beliefs and involve significant risks and uncertainties that could cause actual results, developments and business decisions to differ materially from those contemplated by these statements. These risks and uncertainties include but are not limited to the clinical utility of Altamira's product candidates, the timing or likelihood of regulatory filings and approvals, Altamira's intellectual property position and Altamira's financial position. These risks and uncertainties also include, but are not limited to, those described under the caption "Risk Factors" in Altamira's Annual Report on Form 20-F for the year ended December 31, 2024, and in Altamira's other filings with the Securities Exchange Commission ("SEC"), which are available free of charge on the SEC's website at: www.sec.gov. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those indicated. All forward-looking statements and all subsequent written and oral forward-looking statements attributable to Altamira or to persons acting on behalf of Altamira are expressly qualified in their entirety by reference to these risks and uncertainties. You should not place undue reliance on forward-looking statements. Forward-looking statements speak only as of the date they are made, and Altamira does not undertake any obligation to update them in light of new information, future developments or otherwise, except as may be required under applicable law.
Investor Contact:
SOURCE: Altamira Therapeutics Ltd.
View the original press release on ACCESS Newswire
M.Fischer--AMWN