
-
 El Salvador's missing thousands buried by official indifference
El Salvador's missing thousands buried by official indifference
-
Trump's Fed chair pick to face lawmakers at key confirmation hearing

-
 PGA Tour to scrap Hawaii opening events from 2027
PGA Tour to scrap Hawaii opening events from 2027
-
Amazon invests another $5 bn in Anthropic

-
 Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
-
Wembanyama wins NBA defensive player of the year
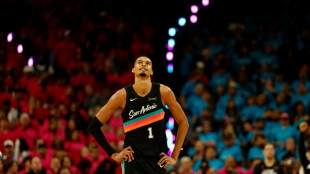
-
 'The Devil Wears Prada 2' stars reunite for glamorous premiere
'The Devil Wears Prada 2' stars reunite for glamorous premiere
-
El Salvador holds mass trial of nearly 500 alleged gang members

-
 Apple's Tim Cook to step down as CEO in September
Apple's Tim Cook to step down as CEO in September
-
West Ham's draw at Palace relegates Wolves, piles pressure on Spurs

-
 Canadian tourist killed in Mexico archaeological site shooting
Canadian tourist killed in Mexico archaeological site shooting
-
Wolves relegated from Premier League

-
 Oil jumps on Hormuz tensions, stocks mostly retreat
Oil jumps on Hormuz tensions, stocks mostly retreat
-
Colombian environmental activist honored amid threats and exile

-
 Gun battle traps more than 200 tourists at Rio viewpoint
Gun battle traps more than 200 tourists at Rio viewpoint
-
Alcaraz may skip French Open rather than rush injury comeback

-
 Top US court to hear case of Catholic schools excluded from state funding
Top US court to hear case of Catholic schools excluded from state funding
-
Trump Fed chair pick to vow interest rate independence at key hearing

-
 EU to host Taliban officials for talks on deporting Afghans
EU to host Taliban officials for talks on deporting Afghans
-
Blue Origin probing rocket's failure to deliver satellite

-
 Pope blasts 'exploitation' as he wraps up tour of Angola
Pope blasts 'exploitation' as he wraps up tour of Angola
-
Wembanyama 'changing the game as we speak', says Nowitzki

-
 Singer D4vd charged with murder after teen's body found in Tesla
Singer D4vd charged with murder after teen's body found in Tesla
-
Swiss football club turn down Kanye West concert approach

-
 Leicester fairytale turns sour as relegation to third tier looms
Leicester fairytale turns sour as relegation to third tier looms
-
Pope Leo blasts 'exploitation' as he wrap up tour of resource-rich Angola

-
 Varma ton revives Mumbai's IPL hopes with win over Gujarat
Varma ton revives Mumbai's IPL hopes with win over Gujarat
-
Formula One makes rule changes after drivers' criticism

-
 Singer D4vd charged with murder over teen's body found in Tesla
Singer D4vd charged with murder over teen's body found in Tesla
-
UK PM denies misleading MPs, says officials hid Mandelson info

-
 Tit-for-tat blockades once again cripple traffic in Hormuz
Tit-for-tat blockades once again cripple traffic in Hormuz
-
Cafu says 2026 World Cup is perfect time for Brazil to win again

-
 Erdogan vows new measures after deadly Turkey school shootings
Erdogan vows new measures after deadly Turkey school shootings
-
Rose to take charge at Bournemouth after Iraola exit

-
 Olympic status a massive 'boost' for squash says European champion Crouin
Olympic status a massive 'boost' for squash says European champion Crouin
-
Kenyan double-double as Korir, Lokedi defend Boston Marathon crowns

-
 Whale stranded on German coast swims off, gets stuck again
Whale stranded on German coast swims off, gets stuck again
-
Iran pulling Hormuz 'lever' to maximum in US standoff

-
 Argentine film and theater great Luis Brandoni dies at 86
Argentine film and theater great Luis Brandoni dies at 86
-
French Open sensation Boisson returns to action after 'most difficult' spell

-
 Desmond Morris: from 'Naked Ape' to watching 'Big Brother'
Desmond Morris: from 'Naked Ape' to watching 'Big Brother'
-
Rosenior says Chelsea owners supportive despite slump

-
 Oil jumps on Hormuz tensions, stocks retreat
Oil jumps on Hormuz tensions, stocks retreat
-
Romania legend Hagi eyes 'winning every game' on return as coach

-
 Rana stars as Bangladesh down New Zealand to level ODI series at 1-1
Rana stars as Bangladesh down New Zealand to level ODI series at 1-1
-
Real Madrid coach Arbeloa launches stout defence of Mbappe

-
 Pope Leo blasts 'exploitation' on visit to resource-rich Angola
Pope Leo blasts 'exploitation' on visit to resource-rich Angola
-
Amy Winehouse's father loses suit against friends selling her clothes

-
 Japan issues warning after 7.7-magnitude quake hits north
Japan issues warning after 7.7-magnitude quake hits north
-
UniCredit woos Commerzbank shareholders in takeover battle


EDGE Surgical Launches 40mm and 120mm Electronic Depth Gauges
Comprehensive Product Line Now Available for Orthopedic Surgery
BURLINGTON, MA / ACCESS Newswire / March 2, 2026 / EDGE Surgical, Inc., the pioneering force behind the world's only electronic depth gauge for orthopedic surgery, proudly announces the expansion of its Ortho EDG® platform with the introduction of the 40mm and the 120mm single-patient use, universal electronic depth gauges. Eliminating the need for reusable depth gauges, EDGE Surgical boasts a complete depth gauge line to address the mini, small and large fragment markets, including tailored solutions for orthopedic specialties like hand and wrist, foot and ankle, craniomaxillofacial and others. The new devices build on the proven success of the previously launched, universal 65mm depth gauge, setting a new standard in surgical precision.
These groundbreaking products will be on display at AAOS.
It's Time to Take Reusable Depth Gauges out of the Tray
The shortcomings associated with reusable depth gauges-including potential inaccuracy and contamination-are well known and present significant risks that can lead to severe, avoidable complications. These risks no longer have to be accepted. According to a prominent chief of orthopedics in Florida, "We don't reuse drill bits, we don't reuse wires-reusing a depth gauge doesn't make sense."
Contaminated surgical instruments have led to shutdowns and delays at hospitals nationwide. These shutdowns not only impact patients but restrict healthcare access to the broader local community and burden the taxpayers who help fund these institutions.
It's no secret why hospitals across the country are adopting state-of-the-art electronic depth gauges from EDGE. Replacing standard depth gauges can reduce cost of care and improve patient outcomes, while also alleviating burden from the overtaxed, under-resourced SPD team struggling to properly clean and sterilize the complicated, reusable depth gauge. Early adopters of the 65mm EDG, including surgeons and hospitals, have seen this value.
"From eight-figure lawsuits to life-changing infections, the stakes for hospitals and patients couldn't be higher. It's imperative to remove the reusable depth gauge from the operating room immediately," said Leo Carayannopoulos, CEO of EDGE Surgical. "Surgeons have struggled, and patients have been put at risk with this antiquated technology long enough. With a complete solution from small to large fragment, EDG reduces risk and helps improve patient outcomes. EDGE is the obvious choice."
Multiple plate and screw manufacturers have expressed interest in adopting the EDGE product line. From improved surgeon support to offering a competitive advantage in workflow and patient outcomes, EDG presents a strategic opportunity.
Tailored, Surgeon-Designed Solutions
The EDG product family is built with surgeon and industry expert guidance input to address the needs of different surgical specialties. The surgeon-driven design, combined with universal compatibility with all major plate and screw systems, eliminates the need for reusable depth gauges in the surgical tray.
A New, Better Standard of Care
Adopting EDG® just makes sense. EDGE Surgical is not merely the future of depth gauge technology-it is the depth gauge perfected.
Accurate: Featuring a digital display far more precise than traditional gauges, combined with a patented hook, the EDG ensures accurate measurements the first time, every time.
Sterile: With every device sterile and packaged for single-patient use, EDG eradicates the risk of secondary infections linked to depth gauges, helping to ensure patient safety.
Universal: EDG is compatible with all major plating systems, manufacturers, and screw sizes, eliminating the need for multiple depth gauges.
Universal Compatibility
The Universal Depth Gauge can be easily calibrated to any plating system from major manufacturers, including Depuy Synthes, Zimmer Biomet, Smith & Nephew, Stryker, Acumed, Paragon 28, Globus, Skeletal Dynamics, Arthrex, Medartis, Trimed, Conventus Flower Orthopedics, and more.
Benefits Beyond the OR
EDG is a game-changer. Compatible with all major plate and screw systems, EDG seamlessly fits into existing surgical workflows. With features like dynamic calibration, the Electronic Depth Gauge ensures ease of use and accurate measurements, contributing to better surgical outcomes.
EDG delivers benefits that go far beyond the OR.
Reducing Backups in Sterile Processing
EDG® is for single-patient use, ultimately leading to the elimination of many depth gauges from surgical trays and reducing SPD load. Confusing or non-existent instructions, and hidden nooks, crannies and narrow cannulations make it nearly impossible to reliably clean depth gauges.
Driving Down Secondary Infection Risk while Driving Efficiency
EDG eliminates a point of risk exposure for secondary site infections and assures the risk management and infection prevention teams that their depth gauges are clean.
For OR leadership, EDG® helps drive efficiency in OR flow and removes potential delays that could impact OR scheduling.
EDGE continues to innovate, developing variants tailored to specialties such as hand and wrist, foot and ankle, lower extremity and others. EDG gives surgeons a sterile, accurate device, no matter the procedure.
The Human Cost of Inaction
The impact of secondary infections and surgical complications extends beyond statistics; they deeply affect individuals, leading to long-term loss of mobility, years of surgeries, physical therapy, and sometimes only partial recovery. EDGE is committed to highlighting these personal stories, bringing attention to the real faces behind these complications and fostering greater awareness of their profound impact. Look for these stories on edgesurcial.com in the weeks to come.
Available for Order at AAOS this week
EDGE Surgical will be showcasing its innovative devices at booth 1003 during the AAOS conference in New Orleans. Attendees will have the opportunity to experience firsthand how the EDG revolutionizes orthopedic practices. The EDGE team will be available to answer questions and discuss how their solutions can enhance surgical precision.
About EDGE Surgical
EDGE Surgical reduces risks and delays in Open Reduction and Internal Fixation (ORIF) surgeries. Its flagship product, the Electronic Depth Gauge (Ortho EDG®), saves time, saves money, and prevents infections, removing risks associated with traditional reusable depth gauges. The EDG, universally compatible with all major plate and screw systems, uses a digital display and patented hook to deliver consistent and accurate results every time. The single-use device is sterile packaged, eliminating risk of biocontamination.
EDG delivers benefits across the organization by reducing unplanned tray replacement orders for supply chain, relieving SPD of cleaning duties for a difficult instrument, lowering secondary infection liability risk, and helping OR leadership stay on schedule. Visit www.edgesurgical.com for more information.
Media Contact:
Mark Severns
Director of Marketing
EDGE Surgical, Inc.
[email protected]
10 Burlington Mall Rd, Suite 206
Burlington, MA 01803
SOURCE: EDGE Surgical
View the original press release on ACCESS Newswire
P.Silva--AMWN