
-
 El Salvador's missing thousands buried by official indifference
El Salvador's missing thousands buried by official indifference
-
Trump's Fed chair pick to face lawmakers at key confirmation hearing

-
 PGA Tour to scrap Hawaii opening events from 2027
PGA Tour to scrap Hawaii opening events from 2027
-
Amazon invests another $5 bn in Anthropic

-
 Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
-
Wembanyama wins NBA defensive player of the year
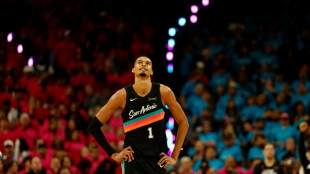
-
 'The Devil Wears Prada 2' stars reunite for glamorous premiere
'The Devil Wears Prada 2' stars reunite for glamorous premiere
-
El Salvador holds mass trial of nearly 500 alleged gang members

-
 Apple's Tim Cook to step down as CEO in September
Apple's Tim Cook to step down as CEO in September
-
West Ham's draw at Palace relegates Wolves, piles pressure on Spurs

-
 Canadian tourist killed in Mexico archaeological site shooting
Canadian tourist killed in Mexico archaeological site shooting
-
Wolves relegated from Premier League

-
 Oil jumps on Hormuz tensions, stocks mostly retreat
Oil jumps on Hormuz tensions, stocks mostly retreat
-
Colombian environmental activist honored amid threats and exile

-
 Gun battle traps more than 200 tourists at Rio viewpoint
Gun battle traps more than 200 tourists at Rio viewpoint
-
Alcaraz may skip French Open rather than rush injury comeback

-
 Top US court to hear case of Catholic schools excluded from state funding
Top US court to hear case of Catholic schools excluded from state funding
-
Trump Fed chair pick to vow interest rate independence at key hearing

-
 EU to host Taliban officials for talks on deporting Afghans
EU to host Taliban officials for talks on deporting Afghans
-
Blue Origin probing rocket's failure to deliver satellite

-
 Pope blasts 'exploitation' as he wraps up tour of Angola
Pope blasts 'exploitation' as he wraps up tour of Angola
-
Wembanyama 'changing the game as we speak', says Nowitzki

-
 Singer D4vd charged with murder after teen's body found in Tesla
Singer D4vd charged with murder after teen's body found in Tesla
-
Swiss football club turn down Kanye West concert approach

-
 Leicester fairytale turns sour as relegation to third tier looms
Leicester fairytale turns sour as relegation to third tier looms
-
Pope Leo blasts 'exploitation' as he wrap up tour of resource-rich Angola

-
 Varma ton revives Mumbai's IPL hopes with win over Gujarat
Varma ton revives Mumbai's IPL hopes with win over Gujarat
-
Formula One makes rule changes after drivers' criticism

-
 Singer D4vd charged with murder over teen's body found in Tesla
Singer D4vd charged with murder over teen's body found in Tesla
-
UK PM denies misleading MPs, says officials hid Mandelson info

-
 Tit-for-tat blockades once again cripple traffic in Hormuz
Tit-for-tat blockades once again cripple traffic in Hormuz
-
Cafu says 2026 World Cup is perfect time for Brazil to win again

-
 Erdogan vows new measures after deadly Turkey school shootings
Erdogan vows new measures after deadly Turkey school shootings
-
Rose to take charge at Bournemouth after Iraola exit

-
 Olympic status a massive 'boost' for squash says European champion Crouin
Olympic status a massive 'boost' for squash says European champion Crouin
-
Kenyan double-double as Korir, Lokedi defend Boston Marathon crowns

-
 Whale stranded on German coast swims off, gets stuck again
Whale stranded on German coast swims off, gets stuck again
-
Iran pulling Hormuz 'lever' to maximum in US standoff

-
 Argentine film and theater great Luis Brandoni dies at 86
Argentine film and theater great Luis Brandoni dies at 86
-
French Open sensation Boisson returns to action after 'most difficult' spell

-
 Desmond Morris: from 'Naked Ape' to watching 'Big Brother'
Desmond Morris: from 'Naked Ape' to watching 'Big Brother'
-
Rosenior says Chelsea owners supportive despite slump

-
 Oil jumps on Hormuz tensions, stocks retreat
Oil jumps on Hormuz tensions, stocks retreat
-
Romania legend Hagi eyes 'winning every game' on return as coach

-
 Rana stars as Bangladesh down New Zealand to level ODI series at 1-1
Rana stars as Bangladesh down New Zealand to level ODI series at 1-1
-
Real Madrid coach Arbeloa launches stout defence of Mbappe

-
 Pope Leo blasts 'exploitation' on visit to resource-rich Angola
Pope Leo blasts 'exploitation' on visit to resource-rich Angola
-
Amy Winehouse's father loses suit against friends selling her clothes

-
 Japan issues warning after 7.7-magnitude quake hits north
Japan issues warning after 7.7-magnitude quake hits north
-
UniCredit woos Commerzbank shareholders in takeover battle


Bioxytran, Inc. Reports Positive Phase 1b/2a Clinical Study Results for ProLectin-M, a Broad-Range Antiviral Drug in Mild to Moderate COVID-19
BOSTON, MA / ACCESS Newswire / March 2, 2026 / Bioxytran, Inc. (OTCQB:BIXT), a clinical-stage biotechnology company developing carbohydrate-based therapeutics, today announced results from a randomized, double-blind, placebo-controlled Phase 1b/2a clinical study evaluating oral ProLectin-M in hospitalized patients with mild to moderate COVID-19 caused by SARS-CoV-2.
The study showed that the highest evaluated dose of ProLectin-M (16,800 mg/day) was associated with statistically significant earlier viral clearance and faster clinical improvement by Day 5 compared with placebo, while demonstrating a favorable safety and tolerability profile. By Day 7, viral clearance was observed across all study arms, consistent with the expected natural resolution of infection in this population, indicating the treatment effect may be related to accelerating viral clearance. No serious adverse events were reported, and no treatment-related discontinuations occurred.
"We believe an oral, well-tolerated antiviral with a differentiated mechanism could address important gaps in current treatment approaches, particularly in early-stage respiratory infections." said Dr. Leslie Ajayi, Bioxytran's Chief Medical Officer. "Our clinical data suggests ProLectin-M demonstrated earlier reductions in viral shedding compared with placebo with a favorable safety profile, and these findings support further evaluation of ProLectin-M in larger, well-controlled studies to assess its potential role as a first-line therapy."
"These findings provide confirmation of an early clinical trials antiviral effect and support further evaluation of ProLectin-M's novel galectin-targeting mechanism," said David Platt, PhD, CEO of Bioxytran. "The clinical trials results are opening a new horizon for a new generation of safe anti-viral drugs. We believe the consistency of the observed activity supports continued clinical development of this oral therapeutic approach."
Study Design
The Phase 1b/2a study enrolled 39 participants in India with RT-PCR-confirmed SARS-CoV-2 infection and mild to moderate disease. Participants were randomized to receive one of three dose levels of ProLectin-M plus standard of care (SOC), or placebo plus SOC, administered over five days.
Dose Arms:
5,600 mg/day ProLectin-M + SOC, 11,200 mg/day ProLectin-M + SOC, 16,800 mg/day ProLectin-M + SOC, Placebo + SOC.
The primary endpoint evaluated absence of detectable viral RNA at Day 7. Secondary endpoints included earlier viral clearance, changes in viral load, clinical status improvement, safety, and pharmacokinetics.
Key Findings
Earlier Viral Clearance (Day 5)
90% of participants receiving 16,800 mg/day achieved non-detectable viral shedding by Day 5. It was compared with 20.0% (placebo), 20.0% (5,600 mg), and 40.0% (11,200 mg). The difference between the 16,800 mg/day cohort and placebo was statistically significant (p=0.001).
Clinical Improvement
90% of participants in the highest-dose cohort achieved at least a 2-point improvement on the WHO Ordinal Scale by Day 5 compared with 20.0%, 40.0%, and 20.0% in the lower-dose and placebo groups. All participants improved clinically by Day 7.
Viral Load Trends
Cycle threshold (Ct) values increased over time across all groups, consistent with declining viral load. Numerically earlier Ct increases were observed in the highest-dose cohort beginning as early as Day 3, supporting the observed Day-5 antiviral signal.
Primary Endpoint Outcome
Because mild-to-moderate COVID-19 in this population typically resolves within 7 days, the primary endpoint at Day 7 did not differentiate treatment arms. However, earlier viral clearance observed at Day 5 suggests a potential acceleration of viral resolution.
Safety and Tolerability
ProLectin-M was well tolerated at all evaluated dose levels with no serious adverse events, no treatment-related discontinuations, no clinically meaningful changes in laboratory values, ECGs, or vital signs. High compliance with the 5-day dosing regimen.
Development Context
ProLectin-M is designed to target galectins, carbohydrate-binding proteins that certain viruses utilize to attach to and enter host cells. By acting on host-virus interactions rather than intracellular viral replication, this approach represents a differentiated antiviral strategy that may have applicability across multiple viral infections. In future studies, ProLectin-M may also be evaluated for its potential as a preventive therapy. The Company believes these results support continued evaluation of ProLectin-M as a potential oral therapeutic and provide a foundation for future clinical studies.
About ProLectin-M
ProLectin-M is an investigational oral antiviral being developed under an active U.S. Investigational New Drug (IND) framework as well as international regulatory oversight. The therapy leverages carbohydrate chemistry to block viral entry mechanisms mediated by galectin interactions.
About Bioxytran, Inc.
Bioxytran, Inc. is a clinical-stage biotechnology company focused on developing novel carbohydrate-based therapeutics to address significant unmet medical needs in infectious and cardiovascular diseases.
Forward-Looking Statements
This press release includes forward-looking statements as defined under federal law, including those related to the performance of technology described in this press release. These forward-looking statements are generally identified by the words "believe," "expect," "anticipate," "estimate," "intend," "plan," and similar expressions, although not all forward-looking statements contain these identifying words. Such statements are subject to significant risks, assumptions and uncertainties. Known material factors that could cause Bioxytran's actual results to differ materially from the results contemplated by such forward-looking statements are described in the forward-looking statements and risk factors in the Company's Annual Report on Form 10-K for the fiscal year ended December 31, 2024, and those risk factors set forth from time-to-time in other filings with the Securities and Exchange Commission. Bioxytran undertakes no obligation to correct or update any forward-looking statement, whether as a result of new information, future events, or otherwise, except to the extent required under federal securities laws.
For more information, please visit:
www.bioxytraninc.com
Investor Contact:
David Platt, PhD
CEO, Bioxytran, Inc.
617-484-1199
[email protected]
SOURCE: BioXyTran, Inc.
View the original press release on ACCESS Newswire
A.Mahlangu--AMWN