
-
 Two women die on migrant boat seeking to reach UK
Two women die on migrant boat seeking to reach UK
-
Mumbai coach Jayawardene backs Suryakumar to find his 'rhythm'

-
 Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach
Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach
-
Bangkok food vendor curbs push city staple from the streets

-
 More Nepalis drive electric, evading global fuel shocks
More Nepalis drive electric, evading global fuel shocks
-
Latecomer Japan eyes slice of rising global defence spending

-
 Messi goal not enough as Miami collapse in 4-3 loss to Orlando
Messi goal not enough as Miami collapse in 4-3 loss to Orlando
-
German fertiliser makers and farmers struggle with Iran war fallout

-
 OPEC+ to make first post-UAE production decision
OPEC+ to make first post-UAE production decision
-
Massive crowds fill Rio's Copacabana beach for Shakira concert

-
 Embiid, Maxey shine as 76ers eliminate Celtics in NBA playoffs
Embiid, Maxey shine as 76ers eliminate Celtics in NBA playoffs
-
Fleeting freedom at festival for India's transgender community
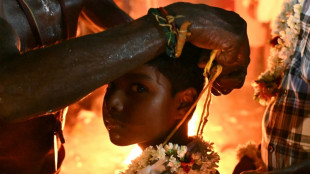
-
 Trump says cutting US troop numbers in Germany 'way down'
Trump says cutting US troop numbers in Germany 'way down'
-
Man charged with murdering Indigenous girl in Australian outback

-
 China's Wu Yize wins last-frame thriller to reach snooker world final
China's Wu Yize wins last-frame thriller to reach snooker world final
-
Serene Korda takes three-shot lead at LPGA Mexico

-
 Golden Tempo wins Kentucky Derby in historic triumph for trainer DeVaux
Golden Tempo wins Kentucky Derby in historic triumph for trainer DeVaux
-
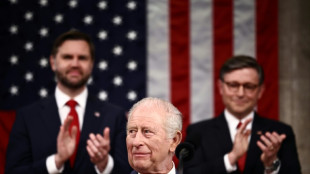
King Charles grasped 'opportunity' on US trip, palace says
-
 China's Wu wins last-frame thriller to reach snooker world final
China's Wu wins last-frame thriller to reach snooker world final
-
Verstappen sees light at the end of tunnel

-
 Young stretches PGA lead to six at Doral
Young stretches PGA lead to six at Doral
-
Rio's Copacabana beach hosts massive crowd for free Shakira concert

-
 Celtics' Tatum ruled out for decisive game seven against Sixers
Celtics' Tatum ruled out for decisive game seven against Sixers
-
Wolff heralds Antonelli speed as teen joins Senna and Schumacher in record books

-
 Senior Iranian officer says fresh conflict with US 'likely'
Senior Iranian officer says fresh conflict with US 'likely'
-
Barcelona on verge of Liga title, Villarreal secure top four

-
 Teen F1 leader Antonelli takes Miami Grand Prix pole
Teen F1 leader Antonelli takes Miami Grand Prix pole
-
Porto edge Alverca to clinch Portuguese league title

-
 US airlines step up as Spirit winds down
US airlines step up as Spirit winds down
-
Barcelona on verge of La Liga title defence with win at Osasuna

-
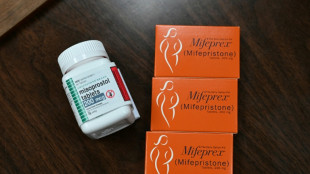 Drugmaker asks US Supreme Court to restore abortion pill access
Drugmaker asks US Supreme Court to restore abortion pill access
-
Schalke return to Bundesliga after three-year absence

-
 NATO, top Republicans question US troop withdrawal from Germany
NATO, top Republicans question US troop withdrawal from Germany
-
Napoli frustrate Como in costly Serie A stalemate

-
 Illegal party at French military site draws up to 40,000 ravers
Illegal party at French military site draws up to 40,000 ravers
-
Arsenal hit stride to go six points clear, West Ham loss offers Spurs hope

-
 Arsenal go six points clear as Gyokeres double sinks Fulham
Arsenal go six points clear as Gyokeres double sinks Fulham
-
Clinical Chennai down Mumbai to keep playoff hopes alive

-
 Napoli and Como play out goalless draw in Serie A
Napoli and Como play out goalless draw in Serie A
-
Murphy into World Snooker Championship final after edging Higgins

-
 PSG held by Lorient with fringe team ahead of Bayern Munich return leg
PSG held by Lorient with fringe team ahead of Bayern Munich return leg
-
Aviation companies step up as Spirit winds down

-
 Champion Norris leads Piastri home in sprint 1-2 triumph for McLaren
Champion Norris leads Piastri home in sprint 1-2 triumph for McLaren
-
UK PM says some pro-Palestinian marches could be banned

-
 The Puma out of Kentucky Derby, leaving 19 starters
The Puma out of Kentucky Derby, leaving 19 starters
-
'Bookless bookstore': audio-only book shop opens in New York

-
 Kostyuk defeats Andreeva to claim first Madrid Open title
Kostyuk defeats Andreeva to claim first Madrid Open title
-
Leinster survive Toulon scare to reach Champions Cup final

-
 Villarreal secure Champions League spot, rotated Atletico win
Villarreal secure Champions League spot, rotated Atletico win
-
'Relieved' Inoue outlasts Nakatani in Tokyo Dome superfight


Is Washington Bypassing Marijuana FDA's Botanical Drug Pathway? Medicare's $500 CBD Pilot Sparking Regulatory Firestorm
WASHINGTON, DC / ACCESS Newswire / March 26, 2026 / The Centers for Medicare & Medicaid Services (CMS) is preparing to launch an unprecedented federal pilot program that could furnish participating care organizations with up to $500 annually per beneficiary for hemp-derived CBD products. While proponents at the White House and CMS hail the initiative as a "Make America Healthy Again" breakthrough for seniors and veterans, a growing chorus of regulatory experts and pharmaceutical leaders are sounding the alarm: the federal government may be rewriting the rules of medicine.

The core of the controversy lies in a fundamental breach of the traditional U.S. medical sequence. For over a century, the standard has been absolute: Science → FDA Approval → Reimbursement. With the new "Substance Access Beneficiary Engagement Incentive" (BEI), critics argue the sequence has been reversed to: Policy → Access → Evidence Later.
The Conflict: CMS Reimbursement vs. FDA Validation
CMS is not a scientific regulatory body; it is a payer. Traditionally, CMS only reimburses therapies that have met the FDA's rigorous standards for safety, efficacy, and manufacturing consistency. However, the new CBD pilot-expected to begin as early as April 1, 2026-identifies no specific validated formulation, no pharmacokinetic standards, and no FDA-approved therapeutic profile for the products it will fund.
"There is already a clearly defined federal pathway for botanical cannabinoid medicines-the FDA created it," stated Duane Boise, CEO of MMJ International Holdings. "Companies that followed the rules spent years on IND submissions, stability testing, and orphan drug research under strict Schedule I oversight. Now, a federal reimbursement program is moving forward before those standards are applied. That is not how medicine is developed in this country."
The "Real-World Data" Paradox
Supporters of the CMS pilot, led by Administrator Dr. Mehmet Oz, argue that the program will generate critical "real-world evidence" to inform future care. However, pharmaceutical developers point out that 20 years of anecdotal state-level use has yet to produce the "regulatory-grade" datasets required for a prescription-grade drug.
The concern is that by funding unvalidated consumer-grade hemp products, the government is incentivizing a "gray market" over companies that have invested millions in the formal FDA process, such as MMJ International Holdings and its work on it's IND for Huntington's disease.
A Contradiction in Federal Policy
The pilot also highlights a bizarre policy paradox within the current administration:
In Congress: Lawmakers are currently tightening the federal definition of hemp, with new statutory limits expected to shrink the ingestible cannabinoid market.
At CMS: The agency is simultaneously expanding federal dollars into that very same market.
The Bottom Line: Redefining the Standard of Care
The CBD pilot may represent a historic shift toward "payer-driven validation," where reimbursement precedes formal botanical drug standardization. If successful, it could change how all future plant-based therapies enter the U.S. clinical ecosystem. If it fails, it risks exposing the nation's most vulnerable populations to inconsistent, unvalidated treatments under a federal banner.
As the April 1 launch date nears, the question remains: Is Washington accelerating innovation, or is it sacrificing the scientific integrity of the drug approval system?
About MMJ International Holdings MMJ International Holdings is a leading biopharmaceutical company dedicated to the development of plant-derived, FDA-approved cannabinoid medicines. Through its subsidiaries, MMJ BioPharma Cultivation and MMJ BioPharma Labs, the company advances therapeutic solutions for rare neurological diseases under strict federal oversight.
Madison Hisey
[email protected]
203-231-85832
SOURCE: MMJ International Holdings
View the original press release on ACCESS Newswire
A.Malone--AMWN