
-
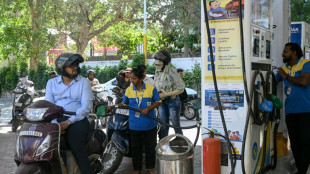 Stocks drop, oil climbs after fresh Trump warning to Iran
Stocks drop, oil climbs after fresh Trump warning to Iran
-
Twins wow Cannes with 'mesmeric' tale of Nigeria's rich

-
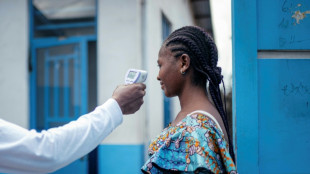 New Ebola outbreak in DR Congo: What we know
New Ebola outbreak in DR Congo: What we know
-
Iran Nobel winner discharged from hospital: supporters

-
 Spanish court orders 55 mn euro tax refund to Shakira
Spanish court orders 55 mn euro tax refund to Shakira
-
Ryanair flags Iran war uncertainty as annual profit jumps

-
 Hearts have bright future despite Scottish title pain: McInnes
Hearts have bright future despite Scottish title pain: McInnes
-
Fernandes 'proud' to match Premier League assists record

-
 Germany set to miss 2030 climate goal: experts
Germany set to miss 2030 climate goal: experts
-
G7 finance chiefs meet to seek common stance on unstable ground

-
 Hantavirus-hit cruise ship docks in Rotterdam at voyage end
Hantavirus-hit cruise ship docks in Rotterdam at voyage end
-
Philippines swears in senators for VP Duterte's impeachment trial

-
 Iran's World Cup football team leaves for Turkey: media
Iran's World Cup football team leaves for Turkey: media
-
Hantavirus-hit cruise ship steams towards Rotterdam at voyage end

-
 Japan arrests Americans over stunt at baby monkey Punch's zoo
Japan arrests Americans over stunt at baby monkey Punch's zoo
-
Trump says 'clock ticking' for Iran as peace negotiations stall

-
 Hong Kong court hears closing arguments in Tiananmen activists' trial
Hong Kong court hears closing arguments in Tiananmen activists' trial
-
World Cup duo Ghana, Cape Verde not among AFCON top seeds

-
 African players in Europe: Daring Semenyo wins final for City
African players in Europe: Daring Semenyo wins final for City
-
Kenya's new poaching problem: smuggling Giant Harvester Ants

-
 WHO kicks off annual assembly amid hantavirus, Ebola crises
WHO kicks off annual assembly amid hantavirus, Ebola crises
-
S. Korean blockbuster 'Hope' underscores growing film ambition

-
 Train driver charged after deadly Bangkok bus collision
Train driver charged after deadly Bangkok bus collision
-
Angry Chinese table tennis fans demand apology for flag gaffe

-
 India's lifeline ferry across strategic archipelago
India's lifeline ferry across strategic archipelago
-
Encroaching world threatens India's last 'uncontacted' tribe

-
 India's strategic $9 bn megaport plan for pristine island
India's strategic $9 bn megaport plan for pristine island
-
In Tierra del Fuego, a hunt for the rodent carrier of hantavirus

-
 Mitchell leads Cavs past top-seeded Detroit into NBA East finals
Mitchell leads Cavs past top-seeded Detroit into NBA East finals
-
China's April consumption, factory output growth slowest in years

-
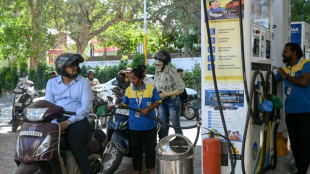 Asian stocks sink, oil rises on US-Iran deadlock
Asian stocks sink, oil rises on US-Iran deadlock
-
Cleveland Cavaliers eliminate top-seeded Detroit from NBA playoffs

-
 Who could be the 2026 World Cup's breakout star?
Who could be the 2026 World Cup's breakout star?
-
Humble PGA champ Rai celebrates English, Indian, Kenyan heritage

-
 Hantavirus-hit cruise ship nears end of voyage, to dock in Rotterdam
Hantavirus-hit cruise ship nears end of voyage, to dock in Rotterdam
-
He said, she said, AI said: Wall Street sex scandal rivets and confounds

-
 UN General Assembly to take up climate change 'obligations' resolution
UN General Assembly to take up climate change 'obligations' resolution
-
Four takeaways from Musk vs OpenAI trial

-
 Jury to decide fate of Musk's blockbuster suit against OpenAI
Jury to decide fate of Musk's blockbuster suit against OpenAI
-
Frustrated McIlroy drops F-bomb in exchange with PGA heckler

-
 Defending champion Palou storms to Indy 500 pole
Defending champion Palou storms to Indy 500 pole
-
Messi shines as Inter Miami finally win at new stadium

-
 Shai Gilgeous-Alexander wins second straight NBA MVP award
Shai Gilgeous-Alexander wins second straight NBA MVP award
-
White House mass prayer event seeks to reclaim US Christian roots

-
 International dive group joins Maldives search for missing Italians
International dive group joins Maldives search for missing Italians
-
'Staggering' Iran toll drives up global executions: Amnesty

-
 Kingsway Publishes Investor Day Presentation to Company Website
Kingsway Publishes Investor Day Presentation to Company Website
-
Helio Ignites Space Infrastructure IP Race with Patent Filing for Deployable Boom Tech, Positioning as Future Leader in Space-Based Solar Power and Orbital Systems

-
 Ryde Appoints Chief Product Officer to Accelerate Platform Innovation and Marketplace Growth
Ryde Appoints Chief Product Officer to Accelerate Platform Innovation and Marketplace Growth
-
XCF Global Highlights Long-Term Growth Drivers for Renewable Fuels as U.S. Biofuel Policy Evolves and SAF Demand Continues to Build


US makes new Alzheimer's drug more widely accessible
The US drug regulator gave full approval to a new Alzheimer's medicine on Thursday, a move that makes it more widely available to the public through government-run health insurance for the elderly.
Leqembi, developed jointly by Japan's Eisai and Biogen of the United States, was shown in a clinical trial to modestly reduce cognitive decline among patients in the early stages of the disease.
But the study also raised concerns about side effects including brain bleeds and swelling.
Leqembi was initially granted "accelerated approval" by the Food and Drug Administration in January, which meant it was not broadly covered by the government-run Medicare program for people aged 65 and older.
Thursday's decision, which follows further study of the drug, means Medicare will now defray a large portion of treatment, initially listed by the makers at $26,500 per year.
"This confirmatory study verified that it is a safe and effective treatment for patients with Alzheimer's disease," senior FDA official Teresa Buracchio said in a statement.
Chiquita Brooks-LaSure, administrator of the agency that runs Medicare, added: "This is welcome news for the millions of people in this country and their families who are affected by this debilitating disease."
But people covered by Medicare will still need to meet 20 percent of the cost, or thousands of dollars, themselves.
Approximately 6.5 million Americans suffer from Alzheimer's, which is characterized by memory loss and declining mental acuity.
Leqembi, also known as lecanemab, is an antibody treatment that is injected into the brain every two weeks and works by reducing amyloid beta, a protein that builds into plaques and causes brain cells to die, as well as brain shrinkage.
The FDA's decision was welcomed by patient groups.
"This treatment, while not a cure, can give people in the early stages of Alzheimer's more time to maintain their independence and do the things they love," said Joanne Pike, Alzheimer's Association president and CEO.
"This gives people more months of recognizing their spouse, children and grandchildren."
Leqembi was the second Alzheimer's drug developed by Eisai and Biogen to receive approval. The first, Aduhelm, was approved in 2021 but the decision was highly controversial as the data about its efficacy was inconsistent.
In May, US drugmaker Eli Lilly announced its drug donanemab also significantly slowed cognitive decline associated with Alzheimer's, and would soon seek worldwide regulatory approval.
Alzheimer's disease accounts for 60 to 80 percent of dementia, according to the Alzheimer's Association. It progressively destroys thinking and memory, eventually robbing people of the ability to carry out the simplest of tasks.
D.Kaufman--AMWN