-
 Man pleads guilty in Austria to plotting attack on Taylor Swift concert
Man pleads guilty in Austria to plotting attack on Taylor Swift concert
-
Climbers open Everest route past dangerous ice block

-
 Indian billionaire's son offers home for Escobar's hippos
Indian billionaire's son offers home for Escobar's hippos
-
Iranian Vafaei capable of great things, says beaten rival Trump

-
 Comedian Kimmel hits back at criticism over Melania Trump joke
Comedian Kimmel hits back at criticism over Melania Trump joke
-
Man goes on trial in Austria over Taylor Swift concert attack plan

-
 South Korean court increases ex-first lady's graft sentence
South Korean court increases ex-first lady's graft sentence
-
Bullying claims 'nonsense', actress Rebel Wilson tells Sydney court

-
 BP reports huge profit rise in first quarter
BP reports huge profit rise in first quarter
-
Crude extends gains, stocks drop as Trump considers latest Iran proposal

-
 How China block of AI deal could stop 'Singapore-washing'
How China block of AI deal could stop 'Singapore-washing'
-
North Korean executions rose dramatically during Covid: report

-
 Budget airlines first to cut flights as jet fuel prices soar
Budget airlines first to cut flights as jet fuel prices soar
-
Simeone, Atletico chasing redemption against Arsenal

-
 'Bring it on', says Rice as Arsenal chase Champions League history
'Bring it on', says Rice as Arsenal chase Champions League history
-
US says examining latest Iran proposal

-
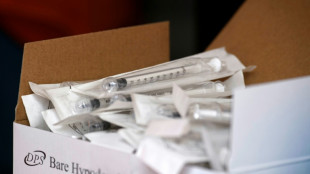 S. Korea probes syringe hoarding as war hits plastic makers
S. Korea probes syringe hoarding as war hits plastic makers
-
Australia aims to tax tech giants unless they pay news outlets

-
 Bangladesh's tigers stalk uncertain future in Sundarbans
Bangladesh's tigers stalk uncertain future in Sundarbans
-
Horses unlikely saviours for those who serve in uniform

-
 Crude extends gains as Trump considers latest Iran proposal
Crude extends gains as Trump considers latest Iran proposal
-
Nations to kick off world-first fossil fuel exit talks

-
 Philippine museum brings deadly, lucrative galleon trade to life
Philippine museum brings deadly, lucrative galleon trade to life
-
Opening remarks Tuesday in Elon Musk versus OpenAI

-
 New York restaurant's $40 half chicken fuels cost of dining debate
New York restaurant's $40 half chicken fuels cost of dining debate
-
Trump shooting scare renews 'staged' conspiracy theory
-
 LIV Golf postpones June event set for New Orleans: reports
LIV Golf postpones June event set for New Orleans: reports
-
XCF Global Highlights Strategic Relevance of Modular SAF Model in Australia as Asia Pacific Jet Fuel Prices Surge from ~$90 to ~$230 per Barrel, a ~155% Increase in Late Feb-Early March 2026

-
 Who Does the Best Tummy Tuck in Kirkland?
Who Does the Best Tummy Tuck in Kirkland?
-
Dynamite Deploys AI-Powered Identity Technology to Deliver Institutional-Grade Security at Consumer-Level Simplicity

-
 Which Customer Review Management Software Leads in 2026?
Which Customer Review Management Software Leads in 2026?
-
Kingfisher Metals Confirms Early Jurassic Porphyry Mineralization Across Hank-Mary District, a Fertile Window Between Mitchell Cu-Au and Brucejack Au-Ag Deposits

-
 Star Copper Completes Copperline Project Inaugural 2025 Field Program at Omineca Mining Division, BC
Star Copper Completes Copperline Project Inaugural 2025 Field Program at Omineca Mining Division, BC
-
BioNxt Applies Advanced Drug Delivery Strategy and Psychedelic Compound Library to Emerging Therapeutics Market Amid Accelerating Global Momentum

-
 SphingoTec Introduces ELISA sphingotest(R) penKid(R) to Enable Broad Kidney Biomarker Testing for Research Applications
SphingoTec Introduces ELISA sphingotest(R) penKid(R) to Enable Broad Kidney Biomarker Testing for Research Applications
-
Apex Drills 4.02% REO over 23.7 m, within Broader 124 m Zone at 1.94% REO, Extending Mineralization Significantly to the South

-
 Hemogenyx Pharmaceuticals PLC Announces Institutional Fundraise
Hemogenyx Pharmaceuticals PLC Announces Institutional Fundraise
-
Caledonia Mining Corporation Plc: Notice of Q1 2026 Results and Investor Presentation

-
 Genflow Biosciences PLC Announces Expansion of Confidentiality Agreements
Genflow Biosciences PLC Announces Expansion of Confidentiality Agreements
-
InterContinental Hotels Group PLC Announces Transaction in Own Shares - April 28

-
 Colombian peace accord failed to protect nature: ex-leader Santos
Colombian peace accord failed to protect nature: ex-leader Santos
-
Nations have chance to break 'fossil fuel mindset': Mary Robinson

-
 Colombia in mourning after deadliest attack in decades
Colombia in mourning after deadliest attack in decades
-
Jury in place for Elon Musk's legal battle with OpenAI

-
 Weinstein rape accuser gives emotional testimony at US retrial
Weinstein rape accuser gives emotional testimony at US retrial
-
Rybakina crashes out of Madrid Open, Sabalenka reaches quarters

-
 Trump and team renew attacks on adversaries after gala shooting
Trump and team renew attacks on adversaries after gala shooting
-
Carrick hails Casemiro and Fernandes after vital Man Utd win

-
 Felix, 40, says she plans comeback for LA Olympics
Felix, 40, says she plans comeback for LA Olympics
-
French FM says Iran must make 'major concessions' to end crisis


enVVeno Medical Successfully Completes Final Wave of Implants in Pre-Clinical GLP Study for enVVe
Successful Completion of All Planned Implants in GLP Study
enVVe Delivery System Demonstrates Consistent Performance
Company Maintains Timeline for IDE Application Submission by Mid-2025, Pending GLP Study Results
enVVeno Medical Corporation (Nasdaq:NVNO) ("enVVeno" or the "Company"), a company setting new standards of care for the treatment of venous disease, today announced the successful completion of the final wave of implants for shorter-term subjects in its six-month pre-clinical GLP study for enVVe, its transcatheter-delivered replacement venous valve.
The successful completion of all planned implants in the GLP study, including both long-term and short-term subjects, completes a critical phase of the study. The follow-up period, which began with the first wave of implants, is ongoing as scheduled. Pending successful completion of the GLP study, the Company anticipates submitting its IDE application to the FDA in mid-2025. The submission, if approved, would allow the Company to initiate the pivotal clinical trial for enVVe.
"With the successful completion of all planned implants in the enVVe GLP study, we have achieved the last of our milestones for 2024," said Robert Berman, enVVeno Medical's Chief Executive Officer. "Our enhanced enVVe crimping and delivery system has performed very well throughout the study and is ready for the pivotal trial. We will continue to monitor the performance of the enVVe valves throughout the remainder of the study and with successful data and pathology, should be in a position to file the IDE on schedule in mid 2025. We remain focused on our goal of becoming the established leader in both the surgical and non-surgical replacement venous valve markets for patients with severe deep venous CVI."
Severe, deep venous Chronic Venous Insufficiency (CVI) is a debilitating disease that is most often caused by blood clots (deep vein thromboses or DVTs) in the deep veins of the leg. When valves inside of the veins of the leg fail, blood flows in the wrong direction and pools in the lower leg, causing pressure within the veins of the leg to increase (venous hypertension). Symptoms of severe CVI include leg swelling, pain, edema, and in the most severe cases, recurrent open sores known as venous ulcers. The disease can severely impact everyday functions such as sleeping, bathing, dressing, and walking, and is known to result in high rates of depression and anxiety. There are currently no effective treatments for severe CVI of the deep vein system caused by valvular incompetence. Estimates indicate that CVI costs the U.S. healthcare system in excess of $4 billion each year.
The Company's lead product is the VenoValve,® a potential first-in-class, surgical replacement venous valve for patients with severe deep venous CVI. In November, the Company submitted a PMA application with the U.S. Food and Drug Administration seeking approval to market and sell the VenoValve in the U.S. The Company estimates that there are approximately 2.5 million potential new patients each year in the U.S. that could be candidates for the VenoValve. The Company is also developing enVVe®, a next-generation, transcatheter based replacement venous valve, that could appeal to an even larger market in terms of both patients and physicians.
Beginning early next year, the Company will begin to implement its strategy to transition from a development stage to a commercial entity for the VenoValve, while completing the necessary non-clinical and GLP testing for enVVe in preparation for its IDE application.
About enVVeno Medical Corporation
enVVeno Medical (NASDAQ:NVNO) is an Irvine, California-based, late clinical-stage medical device Company focused on the advancement of innovative bioprosthetic (tissue-based) solutions to improve the standard of care for the treatment of venous disease. The Company's lead product, the VenoValve®, is a first-in-class surgical replacement venous valve being developed for the treatment of deep venous Chronic Venous Insufficiency (CVI). The Company is also developing a non-surgical, transcatheter based replacement venous valve for the treatment of deep venous CVI called enVVe®. CVI occurs when valves inside of the veins of the leg become damaged, resulting in the backwards flow of blood (reflux), blood pooling in the lower leg, increased pressure in the veins of the leg (venous hypertension) and in severe cases, venous ulcers that are difficult to heal and become chronic. Both the VenoValve and enVVe are designed to act as one-way valves, to help assist in propelling blood up the leg, and back to the heart and lungs. The VenoValve is currently being evaluated in the SAVVE U.S. pivotal study and the Company is currently performing the final testing necessary to seek approval for the pivotal trial for enVVe.
Cautionary Note on Forward-Looking Statements
This press release and any statements of stockholders, directors, employees, representatives and partners of enVVeno Medical Corporation (the "Company") related thereto contain, or may contain, among other things, certain "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements involve significant risks and uncertainties. Such statements may include, without limitation, statements identified by words such as "projects," "may," "will," "could," "would," "should," "believes," "expects," "anticipates," "estimates," "intends," "plans," "potential" or similar expressions. These statements are based upon the current beliefs and expectations of the Company's management and are subject to significant risks and uncertainties, including those detailed in the Company's filings with the Securities and Exchange Commission. Actual results and timing (may differ significantly from those set forth or implied in the forward-looking statements. Forward-looking statements involve certain risks and uncertainties that are subject to change based on various factors (many of which are beyond the Company's control). The Company undertakes no obligation to publicly update any forward-looking statements, whether as a result of new information, future presentations or otherwise, except as required by applicable law.
###
INVESTOR CONTACT:
Jenene Thomas, JTC Team, LLC
[email protected]
(908) 824-0775
SOURCE: enVVeno Medical Corporation
P.Stevenson--AMWN