
-
 Cut off from the West, Muscovites rediscover Russian 'roots'
Cut off from the West, Muscovites rediscover Russian 'roots'
-
'Joint venture in reverse': foreign carmakers seek edge with China partners

-
 Nations backing fossil fuel exit 'a new power': conference host Colombia
Nations backing fossil fuel exit 'a new power': conference host Colombia
-
Rockets thrash Lakers, Wembanyama triumphant on Spurs return

-
 ECB set to hold rates steady with eye on Iran crisis
ECB set to hold rates steady with eye on Iran crisis
-
Team-first Kane propelling Bayern to glory as PSG showdown looms

-
 Pogacar vows to keep going until Seixas 'destroys' him
Pogacar vows to keep going until Seixas 'destroys' him
-
From Adele to Raye, the UK school nurturing future stars

-
 Final talks begin on missing piece for pandemic treaty
Final talks begin on missing piece for pandemic treaty
-
Oil rises, stocks swing as peace talk hopes wobble

-
 'Heartbroken' Xavi Simons out of World Cup and Spurs relegation fight
'Heartbroken' Xavi Simons out of World Cup and Spurs relegation fight
-
North Korea's Kim reaffirms support for Russia's 'sacred' Ukraine war

-
 Spurs win in Wembanyama return to take 3-1 lead over Trail Blazers
Spurs win in Wembanyama return to take 3-1 lead over Trail Blazers
-
As some hijabs come off in Iran, restrictions still in place

-
 Orangutan uses Indonesia canopy bridge in 'world first': NGO
Orangutan uses Indonesia canopy bridge in 'world first': NGO
-
Dealing with the dead in the ruins of Sudan's war

-
 North Korea strengthens nuclear push as US flails in Middle East
North Korea strengthens nuclear push as US flails in Middle East
-
Stage set for Elon Musk's court battle with OpenAI

-
 Caught between wars, US Afghan allies trapped in Qatar without safe exit
Caught between wars, US Afghan allies trapped in Qatar without safe exit
-
British royals begin four-day US visit despite shooting

-
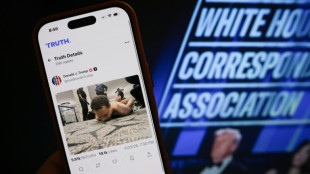 Suspect in shooting at Trump press dinner to appear in court
Suspect in shooting at Trump press dinner to appear in court
-
Fitzpatrick brothers capture PGA Tour's Zurich Classic pairs crown

-
 Spurs win in Wembanyama return to take 3-1 lead on Trail Blazers
Spurs win in Wembanyama return to take 3-1 lead on Trail Blazers
-
Toulouse fall to first home defeat for a year

-
 Global military spending surges on insecurity: report
Global military spending surges on insecurity: report
-
Marseille see Champions League chance slip further away

-
 Nelly Korda wins LPGA Chevron Championship
Nelly Korda wins LPGA Chevron Championship
-
Syrian court begins proceedings against Assad and allies

-
 Colombia road bombing death toll rises to 20
Colombia road bombing death toll rises to 20
-
Raptors top Cavs to pull level in NBA playoff series

-
 Iran minister heads to Russia as talks remain stalled
Iran minister heads to Russia as talks remain stalled
-
Rinku stars as Kolkata edge Lucknow in Super Over

-
 T'Wolves Edwards to miss several weeks - report
T'Wolves Edwards to miss several weeks - report
-
Michael Jackson biopic debuts atop N. America box office

-
 King Charles state visit to US to go on as planned after shooting
King Charles state visit to US to go on as planned after shooting
-
Inter pegged back by Torino as Serie A title charge hits bump in road

-
 Mali junta in crisis after minister killed, key city 'captured'
Mali junta in crisis after minister killed, key city 'captured'
-
Dortmund down Freiburg to seal Champions League spot

-
 McFarlane hails Chelsea 'character' after FA Cup semi-final win
McFarlane hails Chelsea 'character' after FA Cup semi-final win
-
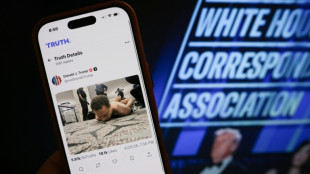
Gunman sought to kill Trump, cabinet at gala dinner
-
 Arsenal punish Lyon errors in Champions League semi
Arsenal punish Lyon errors in Champions League semi
-
Suspect in US press gala shooting - what we know

-
 Key US senator lifts block on Fed chair nominee
Key US senator lifts block on Fed chair nominee
-
Attacks in Mali: What we know

-
 Vollering wins women's Lige-Bastogne-Liege for 3rd time
Vollering wins women's Lige-Bastogne-Liege for 3rd time
-
Sinner motors on in Madrid as Gauff overcomes stomach bug

-
 Fernandez sends Chelsea into FA Cup final to lift gloom after Rosenior sacking
Fernandez sends Chelsea into FA Cup final to lift gloom after Rosenior sacking
-
Colombia road bombing death toll rises to 19

-
 Stuttgart stumble against Bremen in top-four race
Stuttgart stumble against Bremen in top-four race
-
Two former Israel PMs unite to challenge Netanyahu in elections


One-Year Data from the VenoValve(R) U.S. Pivotal Trial Highlighting Impact on Patients' Quality of Life (QOL) to be Presented Today at the 37th Annual Meeting of the American Venous Forum
One-year follow-up data demonstrates sustained improvements across venous specific quality-of-life (QoL) indicators.
PMA application for the VenoValve submitted in November 2024, with an FDA decision expected in the second half of 2025.
IRVINE, CA / ACCESS Newswire / February 19, 2025 / enVVeno Medical Corporation (NASDAQ:NVNO) ("enVVeno" or the "Company"), a company setting new standards of care for the treatment of venous disease, today announced that one-year follow-up data on 75 subjects from the VenoValve U.S. pivotal trial will be presented by Dr. Cassius Iyad Ochoa Chaar, Associate Professor Surgery, Yale School of Medicine, Division of Vascular and Endovascular Surgery, Principal Investigator, and lead enroller for the trial, at the 37th Annual Meeting of the American Venous Forum (VENOUS2025) being held February 16-19, 2025 in Atlanta, GA. The presentation is scheduled for today at 9:47 AM ET.

The data show that trial subjects who had the SAVVE procedure experienced statistically significant improvements in QoL metrics related to venous disease at 12 months compared to baseline as demonstrated by the VEINS-SYM/QOL scores. To view results from the trial, please visit www.venovalve.com. The Company has submitted a pre-market authorization (PMA) application for the VenoValve to the U.S. Food and Drug Administration (FDA), with a decision anticipated in the second half of 2025.
"We continue to be asked to present our data at leading vascular conferences throughout the world as news of the potential for the VenoValve spreads among vascular surgeons whose practices include the treatment of venous diseases," said Robert Berman, enVVeno Medical's Chief Executive Officer. "The VenoValve has the potential to be the first FDA approved treatment for deep venous CVI, and we are attempting to reach the top of a mountain that nobody has successfully climbed and with many failed attempts over the past several decades. With the summit of the mountain finally in sight, this is an exciting time for our company and for the millions of patients with severe deep venous CVI who have no effective treatment options."
Severe, deep venous CVI is a debilitating disease that is most often caused by blood clots (deep vein thromboses or DVTs) in the deep veins of the leg. When valves inside of the veins of the leg fail, blood flows in the wrong direction and pools in the lower leg, causing pressure within the veins of the leg to increase (venous hypertension). Symptoms of severe CVI include leg swelling, pain, edema, and in the most severe cases, recurrent open sores known as venous ulcers. The disease can severely impact everyday functions such as sleeping, bathing, dressing, and walking, and is known to result in high rates of depression and anxiety. There are currently no effective treatments for severe CVI of the deep vein system caused by valvular incompetence. Estimates indicate that CVI costs the U.S. healthcare system in excess of $4 billion each year.
The Venous Insufficiency Epidemiological and Economic Study Quality of Life/Symptoms (VEINES-QoL/Sym) questionnaire is a validated, disease-specific instrument to measure patient-reported quality of life and symptom severity in individuals with chronic venous disease. By assessing factors such as pain, functional limitations, and psychosocial impacts, VEINES-QoL/Sym provides a standardized framework for evaluating treatment outcomes and guiding clinical decision-making.
About enVVeno Medical Corporation
enVVeno Medical (NASDAQ:NVNO) is an Irvine, California-based, late clinical-stage medical device Company focused on the advancement of innovative bioprosthetic (tissue-based) solutions to improve the standard of care for the treatment of venous disease. The Company's lead product, the VenoValve®, is a first-in-class surgical replacement venous valve being developed for the treatment of deep venous Chronic Venous Insufficiency (CVI). The Company is also developing a non-surgical, transcatheter based replacement venous valve for the treatment of deep venous CVI called enVVe®. CVI occurs when valves inside of the veins of the leg become damaged, resulting in the backwards flow of blood (reflux), blood pooling in the lower leg, increased pressure in the veins of the leg (venous hypertension) and in severe cases, venous ulcers that are difficult to heal and become chronic. Both the VenoValve and enVVe are designed to act as one-way valves, to help assist in propelling blood up the leg, and back to the heart and lungs. The VenoValve is currently being evaluated in the SAVVE U.S. pivotal study and the Company is currently performing the final testing necessary to seek approval for the pivotal trial for enVVe.
Cautionary Note on Forward-Looking Statements
This press release and any statements of stockholders, directors, employees, representatives and partners of enVVeno Medical Corporation (the "Company") related thereto contain, or may contain, among other things, certain "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements involve significant risks and uncertainties. Such statements may include, without limitation, statements identified by words such as "projects," "may," "will," "could," "would," "should," "believes," "expects," "anticipates," "estimates," "intends," "plans," "potential" or similar expressions. These statements are based upon the current beliefs and expectations of the Company's management and are subject to significant risks and uncertainties, including those detailed in the Company's filings with the Securities and Exchange Commission. Actual results and timing (may differ significantly from those set forth or implied in the forward-looking statements. Forward-looking statements involve certain risks and uncertainties that are subject to change based on various factors (many of which are beyond the Company's control). The Company undertakes no obligation to publicly update any forward-looking statements, whether as a result of new information, future presentations or otherwise, except as required by applicable law.
###
INVESTOR CONTACT:
Jenene Thomas, JTC Team, LLC
[email protected]
(908) 824-0775
SOURCE: enVVeno Medical Corporation
View the original press release on ACCESS Newswire
J.Oliveira--AMWN

