
-
 No Iran delegation sent to US talks yet as truce expiry nears
No Iran delegation sent to US talks yet as truce expiry nears
-
Rover discovers more building blocks of life on Mars

-
 Russia, North Korea connect road bridge ahead of summer opening
Russia, North Korea connect road bridge ahead of summer opening
-
'Strangled': Pakistan faces economic imperative in Iran war peace push

-
 Apple's Tim Cook to step down as CEO after 15-year run
Apple's Tim Cook to step down as CEO after 15-year run
-
Michael Jackson fans pack Hollywood for biopic premiere

-
 Turkey arrests 110 coal miners on hunger strike
Turkey arrests 110 coal miners on hunger strike
-
Oil prices dip, stocks rise on lingering Iran peace hopes

-
 Associated British Foods to spin off Primark clothes brand
Associated British Foods to spin off Primark clothes brand
-
Pope visits Eq. Guinea on last stop of Africa tour

-
 Hello Kitty's parent company to make own video games
Hello Kitty's parent company to make own video games
-
Di Matteo says 'vital' for faltering Chelsea to add experience

-
 Ex-Spurs star Davids condemns 'lack of quality, lack of management'
Ex-Spurs star Davids condemns 'lack of quality, lack of management'
-
Turkmenistan, the gas giant increasingly dependent on China

-
 Romanian AI music sensation Lolita sparks racism debate
Romanian AI music sensation Lolita sparks racism debate
-
Timberwolves battle back to stun Nuggets in NBA playoffs

-
 Eta appointment 'no surprise' for Union Berlin's ascendant women
Eta appointment 'no surprise' for Union Berlin's ascendant women
-
Democrats eye Virginia gains in war with Trump over US voting map

-
 Tourists trickle back to Kashmir, one year after deadly attack
Tourists trickle back to Kashmir, one year after deadly attack
-
Inside the world of ultra-luxury wedding cakes

-
 Chinese AI circuit board maker soars on Hong Kong debut
Chinese AI circuit board maker soars on Hong Kong debut
-
Oil prices dip, most stocks rise on lingering Iran peace hopes

-
 Tim Cook's time as Apple chief marked by profit absent awe
Tim Cook's time as Apple chief marked by profit absent awe
-
Mitchell, Harden shine as Cavs down Raptors for 2-0 series lead

-
 El Salvador's missing thousands buried by official indifference
El Salvador's missing thousands buried by official indifference
-
Trump's Fed chair pick to face lawmakers at key confirmation hearing

-
 PGA Tour to scrap Hawaii opening events from 2027
PGA Tour to scrap Hawaii opening events from 2027
-
Amazon invests another $5 bn in Anthropic

-
 Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
-
Lobe Sciences Ltd. Engages Massive Intelligence Pty Ltd for Investor Relations and Corporate Communications Services

-
 Genoil Inc. Offers 30-60 Day Upstream Solution To Combat Hormuz Crisis As Physical Oil Tops $250, Threatening $700 Barrel Based On A Global Derivative Collapse
Genoil Inc. Offers 30-60 Day Upstream Solution To Combat Hormuz Crisis As Physical Oil Tops $250, Threatening $700 Barrel Based On A Global Derivative Collapse
-
Silver Range Advances the Drum Project in Utah

-
 Battery X Metals Files International PCT Patent Application for Lithium-Ion Battery Rebalancing Technology, Providing a Pathway to Pursue Patent Protection in 150+ Countries for Technology Validated by a Leading Scientific Institution to Recover ~99% Capacity Loss and Extend Battery Lifespan
Battery X Metals Files International PCT Patent Application for Lithium-Ion Battery Rebalancing Technology, Providing a Pathway to Pursue Patent Protection in 150+ Countries for Technology Validated by a Leading Scientific Institution to Recover ~99% Capacity Loss and Extend Battery Lifespan
-
InterContinental Hotels Group PLC Announces Transaction in Own Shares - April 21

-
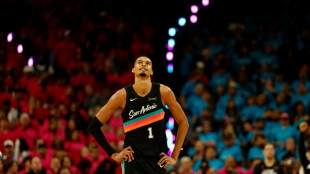 Wembanyama wins NBA defensive player of the year
Wembanyama wins NBA defensive player of the year
-
'The Devil Wears Prada 2' stars reunite for glamorous premiere

-
 El Salvador holds mass trial of nearly 500 alleged gang members
El Salvador holds mass trial of nearly 500 alleged gang members
-
Apple's Tim Cook to step down as CEO in September

-
 West Ham's draw at Palace relegates Wolves, piles pressure on Spurs
West Ham's draw at Palace relegates Wolves, piles pressure on Spurs
-
Canadian tourist killed in Mexico archaeological site shooting

-
 Wolves relegated from Premier League
Wolves relegated from Premier League
-
Oil jumps on Hormuz tensions, stocks mostly retreat

-
 Colombian environmental activist honored amid threats and exile
Colombian environmental activist honored amid threats and exile
-
Gun battle traps more than 200 tourists at Rio viewpoint

-
 Alcaraz may skip French Open rather than rush injury comeback
Alcaraz may skip French Open rather than rush injury comeback
-
Top US court to hear case of Catholic schools excluded from state funding

-
 Trump Fed chair pick to vow interest rate independence at key hearing
Trump Fed chair pick to vow interest rate independence at key hearing
-
EU to host Taliban officials for talks on deporting Afghans

-
 Blue Origin probing rocket's failure to deliver satellite
Blue Origin probing rocket's failure to deliver satellite
-
Pope blasts 'exploitation' as he wraps up tour of Angola


Dr. Roy Levitt, Executive Chairman of Adolore BioTherapeutics, Presented at the Next Generation Gene Therapy Vectors Summit
DELRAY BEACH, FL / ACCESS Newswire / August 12, 2025 / Adolore BioTherapeutics, Inc., ("Company" or "Adolore") announced that Roy Clifford Levitt, MD, Clinical Professor at the University of Miami, Principal Investigator and Program Director of the NIH, NINDS, HEAL UH3 Award supporting ADB-102 development for the treatment of chronic knee pain due to osteoarthritis, ("OA"), and founder and Executive Chairman of Adolore presented the Company's breakthrough non-opioid gene therapy programs for chronic pain at the Next Generation Gene Therapy Vectors Summit on July 31, 2025 in Boston.
Dr. Levitt presented the latest safety and efficacy data on Adolore's replication defective, disease-free, HSV viral vectors during his talk entitled: "Rethinking Vector Choice: Utilizing Optimized HSV to Enhance Safety & Efficacy". He presented evidence of long-lasting (>7 months), profound analgesia (equivalent to high doses of opioids) for their HSV gene therapy with regional administration (single intra-articular knee joint injection) in model systems. Additional data demonstrated excellent cellular tropism (neuronal specificity), biodistribution, and shedding characteristics. Dr. Levitt also highlighted how regional administration minimizes off-target effects, improving safety, efficacy, and minimizes immunogenicity.
Adolore is advancing two preclinical development programs: a lead program for knee pain due to OA and a program for erythromelalgia, ("EM"), an orphan neuropathic pain indication for which there are no FDA-approved treatments. EM is a rare, heritable, chronic and debilitating pain disease.
In model systems, replication-defective, disease-free, herpes simplex virus (rdHSV) gene therapy expressing an analgesic carbonic anhydrase-8 (CA8*) peptide variant corrects somatosensory hyperexcitability by activating Kv7 voltage-gated potassium channels, thereby producing profound, long-lasting analgesia. Adolore has achieved proof-of-concept in animal models, validating the mechanism of action in knee pain from OA and EM.
About Adolore BioTherapeutics, Inc.
Adolore BioTherapeutics, Inc., is a biotechnology company focused on developing novel therapies for treating chronic pain using a revolutionary intra-cellular replication-defective HSV (rdHSV) drug delivery platform that is disease-free, non-toxic, and permits localized peripheral nervous system delivery of proprietary biotherapeutics. This rdHSV gene therapy technology incorporates an established re-dosing strategy and an excellent safety profile. HSV vectors are known for their stability and prolonged gene expression, providing an excellent basis for the long-term treatment of chronic pain conditions and neurological disorders. Our best-in-class CA8* programs are long-acting, locally acting gene therapies that are opioid-free Disease-Modifying Anti-Pain therapies (DMAPs) designed to treat many forms of chronic pain as well as therapies for epilepsy and hearing loss.
Leveraging its innovative gene therapy vectors expressing CA8* analgesic peptides (ADLR-1001), Adolore is currently advancing two preclinical development programs: ADB-101 for the treatment of patients' chronic pain caused by erythromelalgia, an orphan disease, and ADB-102, their lead program for the treatment of patients with chronic pain caused by knee OA. Based on substantial compelling preclinical data generated to date, the Company is progressing these programs toward IND filings and first-in-human clinical studies. Adolore has two additional programs available for partnering: ADB-104 for Drug-Resistant Refractory Focal Epilepsy, ADB-105 for Acute Severe Hearing Loss and ADB-106 for Chronic Eye Pain.
For more information, visit adolore.com.
Forward-Looking Statements
To the extent this announcement contains information and statements that are not historical, they are forward-looking statements within the meaning of the federal securities laws. You can identify forward-looking statements by the use of the words "believe," "expect," "anticipate," "intend," "estimate," "project," "will," "should," "may," "plan," "intend," "assume" and other expressions which predict or indicate future events and trends and which do not relate to historical matters. You should not rely on forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, some of which are beyond the control of the Company. These risks and uncertainties include, but are not limited to, those associated with drug development. These risks, uncertainties, and other factors may cause the actual results, performance, or achievements of the Company to be materially different from the anticipated future results, performance, or achievements expressed or implied by the forward-looking statements.
Investor Relations Contact
Paul Barone (215) 622-4542
[email protected]
SOURCE: Adolore Biotherapeutics, Inc.
View the original press release on ACCESS Newswire
O.Karlsson--AMWN