
-
 Oil prices bounce back on Iran war escalation
Oil prices bounce back on Iran war escalation
-
Residents return to ravaged homes months after Hong Kong fire

-
 Australia's Green wins playoff for third LPGA LA Championship title
Australia's Green wins playoff for third LPGA LA Championship title
-
Pakistan's military chief takes lead on US-Iran talks in diplomatic blitz

-
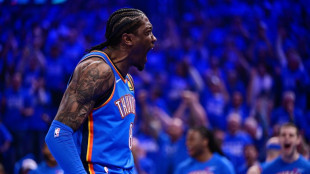 Thunder, Celtics open NBA playoffs with big wins, Magic shock Pistons
Thunder, Celtics open NBA playoffs with big wins, Magic shock Pistons
-
US begins Philippines war games in thick of Middle East conflict

-
 Who's Bad? Not Michael Jackson in new big-budget biopic
Who's Bad? Not Michael Jackson in new big-budget biopic
-
Nations gather for first-ever conference on fossil fuel exit

-
 Money, lobbyists, inertia: why fossil fuels are so hard to quit
Money, lobbyists, inertia: why fossil fuels are so hard to quit
-
France summons Elon Musk over X probe

-
 'Save humanity': Four figures battling it out to lead embattled UN
'Save humanity': Four figures battling it out to lead embattled UN
-
Gilgeous-Alexander, Wemby, Jokic finalists for NBA MVP

-
 Israel vows to level homes in Lebanon, counter threats with 'full force'
Israel vows to level homes in Lebanon, counter threats with 'full force'
-
Rahm coasts to LIV Golf win in Mexico City

-
 Fitzpatrick survives Scheffler playoff to win RBC Heritage
Fitzpatrick survives Scheffler playoff to win RBC Heritage
-
Thunder thrash Suns, Celtics crush Sixers in NBA playoff openers
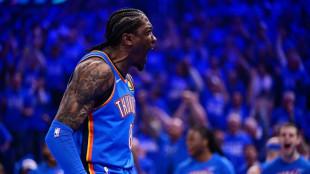
-
 Bulgaria's former president tops parliamentary vote
Bulgaria's former president tops parliamentary vote
-
Kenyans Korir, Lokedi seek to repeat at Boston Marathon

-
 AC Milan, Juventus close in on Champions League qualification
AC Milan, Juventus close in on Champions League qualification
-
Spring double keeps Racing 92 in Top 14 play-off hunt with Paris derby win

-
 Endrick stars as Lyon dent PSG's Ligue 1 title hopes
Endrick stars as Lyon dent PSG's Ligue 1 title hopes
-
History haunts Arsenal as Man City take control of title race

-
 AC Milan and Juventus close in on Champions League qualification
AC Milan and Juventus close in on Champions League qualification
-
Celtics crush Sixers as Tatum and Brown shine in playoff opener

-
 Guardiola warns title not won yet as Man City hunt down Arsenal
Guardiola warns title not won yet as Man City hunt down Arsenal
-
Arteta tells Arsenal to 'go again' in pursuit of Premier League title

-
 Treble-chasing Bayern put beer showers on ice despite title win
Treble-chasing Bayern put beer showers on ice despite title win
-
Eight children dead in US domestic violence shooting

-
 Arya, Connolly help Punjab hammer Lucknow in IPL
Arya, Connolly help Punjab hammer Lucknow in IPL
-
Man City beat Arsenal to seize control of title race, Liverpool win

-
 Kane scores as Bayern sink Stuttgart to claim Bundesliga title
Kane scores as Bayern sink Stuttgart to claim Bundesliga title
-
Balogun continues Monaco scoring streak, Rennes boost Champions League hopes

-
 Trump orders negotiators to Pakistan, but Iran on the fence over talks
Trump orders negotiators to Pakistan, but Iran on the fence over talks
-
Haaland gives Man City edge over Arsenal in Premier League title showdown

-
 Slot hails Liverpool mentality after last-gasp derby winner
Slot hails Liverpool mentality after last-gasp derby winner
-
Top boss vows 'no sitting still' as rugby bids to conquer US

-
 Fils wins on Barcelona clay with French Open looming
Fils wins on Barcelona clay with French Open looming
-
'Super Mario Galaxy' rules N. America box office for third week

-
 Liverpool snatch derby win ahead of City-Arsenal showdown
Liverpool snatch derby win ahead of City-Arsenal showdown
-
Evenepoel outsprints Skjelmose to win Amstel Gold Race

-
 Liverpool beat Everton ahead of City-Arsenal showdown
Liverpool beat Everton ahead of City-Arsenal showdown
-
Rabiot fires AC Milan past Verona to verge of Champions League return

-
 UK PM vows to find arsonists of London Jewish sites
UK PM vows to find arsonists of London Jewish sites
-
Rinku blitz leads Kolkata to first win of IPL season

-
 Shelton wins fifth ATP title with victory in Munich
Shelton wins fifth ATP title with victory in Munich
-
UK's Starmer to face grilling from MPs over Mandelson scandal

-
 Trump again threatens Iran infrastructure as he orders negotiators to Pakistan
Trump again threatens Iran infrastructure as he orders negotiators to Pakistan
-
Rybakina outclasses Muchova to win Stuttgart WTA title

-
 Blasi stuns field with victory in women's Amstel Gold Race
Blasi stuns field with victory in women's Amstel Gold Race
-
Pakistan tightens security in Islamabad ahead of US-Iran talks


Form Bio and Cure Rare Disease Partner to Accelerate Genetic Medicine Development, Advancing CRD's Duchenne Muscular Dystrophy Program
Partnership leverages AI-powered genome design to optimize AAV constructs, accelerate lead candidate selection, and improve drug substance manufacturability for rare and ultra-rare neuromuscular diseases
AUSTIN, TX AND WOODBRIDGE, CT, TX / ACCESS Newswire / October 6, 2025 / Form Bio, a leader in AI-powered genetic medicine development and genome engineering, today announced a strategic partnership with Cure Rare Disease (CRD), a nonprofit biotechnology company developing genetic therapies for the treatment of rare and ultra-rare neuromuscular and neurodegenerative diseases. Together, the organizations will accelerate programs to identify and optimize multiple lead indications, beginning with Duchenne muscular dystrophy (DMD).
Through this collaboration, Form Bio will conduct AI research using its proprietary in silico platform to support CRD in developing gene therapy drug candidates optimized for packaging, expression, safety, and manufacturability, critical factors in ensuring clinical success and cost reduction. The program is designed to streamline lead candidate selection, accelerating development timelines and advancing outcomes for patients with devastating neuromuscular diseases that currently lack treatment options.
CRD initially partnered with Form Bio to improve a DMD therapeutic candidate that was not yet optimized for manufacturing or reduced immunogenicity. Using long-read sequencing, genome integrity analysis, rational design, and AI/ML models, Form Bio identified packaging inefficiencies that could have inhibited clinical success. By modifying the promoter and optimizing the nuclease, Form Bio improved the percentage of AAV full genome from 13% to 59%, a 4.4-fold increase in therapeutic yield achieved within just one month.
"For patients and families facing rare neuromuscular and neurodegenerative diseases, time is critical," said Richard Horgan, Founder and CEO of Cure Rare Disease. "Form Bio's expertise and technology have already delivered a measurable improvement in our DMD program, advancing it to in vivo preclinical studies with significantly greater manufacturability and efficiency. This partnership represents a powerful example of how nonprofits and technology leaders can join forces to accelerate the development of life-changing therapies for overlooked populations."
"Our mission at Form Bio is to empower partners like Cure Rare Disease with our AI-driven insights that transform biology into engineered, programmable systems," said Michelle Chen, Ph.D., President and CEO of Form Bio. "By optimizing AAV vector design upfront, we can reduce time, cost, and risk across the entire gene therapy development pipeline. We are proud to enable CRD in bringing much-needed therapies closer to patients who currently have no options."
Form Bio's AI-augmented CDS optimization and smart vector genome design create measurable improvements that compound across every stage of development, from cell-based research to commercial manufacturing. By maximizing the proportion of capsids containing a full vector genome, Form Bio de-risks manufacturing for genome integrity, a critical quality attribute for regulatory approval, and supports the development of successful therapeutics. In addition, FORMsightAI technology simplifies genome complexity and enables balanced optimization for both expression and CpG reduction.
Together, Form Bio and CRD are demonstrating how advanced computational tools and patient-focused biotech innovation can accelerate breakthroughs, reduce costs, and bring safer, more effective therapies to patients with urgent unmet needs.
About Form Bio
Form Bio is a leader in developing AI-powered solutions for genetic medicine and genome engineering. Since spinning out from Colossal Biosciences in 2022, the company has become a trusted innovation partner to cell and gene therapy developers and beyond. Form Bio's advanced in silico technology and computational tools deliver deep insights into genome integrity, optimized payload design, and other critical factors-enabling safer, more efficacious, and more manufacturable therapeutics. By reducing risk, shortening timelines, and optimizing outcomes, Form Bio helps teams accelerate the path from discovery to the clinic. For more information, visit www.formbio.com or follow us on LinkedIn.
About Cure Rare Disease
Cure Rare Disease, a 501(c)(3) nonprofit biotechnology company based in Woodbridge, CT, is dedicated to developing life-saving genetic therapies for ultra-rare diseases, transforming the lives of patients previously deemed untreatable. By fostering strategic collaborations among top researchers, clinicians, policy experts, and donors, we bring hope and advanced solutions to those awaiting a cure.
Contact Information
Angela Anderson
Form Bio
[email protected]
Ahna Gavrelos
Cure Rare Disease
[email protected]
SOURCE: Form Bio
View the original press release on ACCESS Newswire
S.Gregor--AMWN


