
-
 El Salvador's missing thousands buried by official indifference
El Salvador's missing thousands buried by official indifference
-
Trump's Fed chair pick to face lawmakers at key confirmation hearing

-
 PGA Tour to scrap Hawaii opening events from 2027
PGA Tour to scrap Hawaii opening events from 2027
-
Amazon invests another $5 bn in Anthropic

-
 Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
Israel PM vows 'harsh action' against soldier vandalising Jesus statue in Lebanon
-
Wembanyama wins NBA defensive player of the year
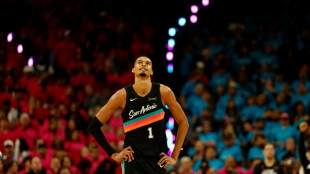
-
 'The Devil Wears Prada 2' stars reunite for glamorous premiere
'The Devil Wears Prada 2' stars reunite for glamorous premiere
-
El Salvador holds mass trial of nearly 500 alleged gang members

-
 Apple's Tim Cook to step down as CEO in September
Apple's Tim Cook to step down as CEO in September
-
West Ham's draw at Palace relegates Wolves, piles pressure on Spurs

-
 Canadian tourist killed in Mexico archaeological site shooting
Canadian tourist killed in Mexico archaeological site shooting
-
Wolves relegated from Premier League

-
 Oil jumps on Hormuz tensions, stocks mostly retreat
Oil jumps on Hormuz tensions, stocks mostly retreat
-
Colombian environmental activist honored amid threats and exile

-
 Gun battle traps more than 200 tourists at Rio viewpoint
Gun battle traps more than 200 tourists at Rio viewpoint
-
Alcaraz may skip French Open rather than rush injury comeback

-
 Top US court to hear case of Catholic schools excluded from state funding
Top US court to hear case of Catholic schools excluded from state funding
-
Trump Fed chair pick to vow interest rate independence at key hearing

-
 EU to host Taliban officials for talks on deporting Afghans
EU to host Taliban officials for talks on deporting Afghans
-
Blue Origin probing rocket's failure to deliver satellite

-
 Pope blasts 'exploitation' as he wraps up tour of Angola
Pope blasts 'exploitation' as he wraps up tour of Angola
-
Wembanyama 'changing the game as we speak', says Nowitzki

-
 Singer D4vd charged with murder after teen's body found in Tesla
Singer D4vd charged with murder after teen's body found in Tesla
-
Swiss football club turn down Kanye West concert approach

-
 Leicester fairytale turns sour as relegation to third tier looms
Leicester fairytale turns sour as relegation to third tier looms
-
Pope Leo blasts 'exploitation' as he wrap up tour of resource-rich Angola

-
 Varma ton revives Mumbai's IPL hopes with win over Gujarat
Varma ton revives Mumbai's IPL hopes with win over Gujarat
-
Formula One makes rule changes after drivers' criticism

-
 Singer D4vd charged with murder over teen's body found in Tesla
Singer D4vd charged with murder over teen's body found in Tesla
-
UK PM denies misleading MPs, says officials hid Mandelson info

-
 Tit-for-tat blockades once again cripple traffic in Hormuz
Tit-for-tat blockades once again cripple traffic in Hormuz
-
Cafu says 2026 World Cup is perfect time for Brazil to win again

-
 Erdogan vows new measures after deadly Turkey school shootings
Erdogan vows new measures after deadly Turkey school shootings
-
Rose to take charge at Bournemouth after Iraola exit

-
 Olympic status a massive 'boost' for squash says European champion Crouin
Olympic status a massive 'boost' for squash says European champion Crouin
-
Kenyan double-double as Korir, Lokedi defend Boston Marathon crowns

-
 Whale stranded on German coast swims off, gets stuck again
Whale stranded on German coast swims off, gets stuck again
-
Iran pulling Hormuz 'lever' to maximum in US standoff

-
 Argentine film and theater great Luis Brandoni dies at 86
Argentine film and theater great Luis Brandoni dies at 86
-
French Open sensation Boisson returns to action after 'most difficult' spell

-
 Desmond Morris: from 'Naked Ape' to watching 'Big Brother'
Desmond Morris: from 'Naked Ape' to watching 'Big Brother'
-
Rosenior says Chelsea owners supportive despite slump

-
 Oil jumps on Hormuz tensions, stocks retreat
Oil jumps on Hormuz tensions, stocks retreat
-
Romania legend Hagi eyes 'winning every game' on return as coach

-
 Rana stars as Bangladesh down New Zealand to level ODI series at 1-1
Rana stars as Bangladesh down New Zealand to level ODI series at 1-1
-
Real Madrid coach Arbeloa launches stout defence of Mbappe

-
 Pope Leo blasts 'exploitation' on visit to resource-rich Angola
Pope Leo blasts 'exploitation' on visit to resource-rich Angola
-
Amy Winehouse's father loses suit against friends selling her clothes

-
 Japan issues warning after 7.7-magnitude quake hits north
Japan issues warning after 7.7-magnitude quake hits north
-
UniCredit woos Commerzbank shareholders in takeover battle


Prolocor(R) Research Named a "Top 10 Article of 2025" by the Journal of Invasive Cardiology
PHILADELPHIA, PENNSYLVANIA / ACCESS Newswire / March 2, 2026 / Prolocor, Inc., a healthcare company developing the innovative diagnostic Prolocor pFCG™ test to identify patients at risk of thrombotic events, today announced that its recent publication, entitled "Prognostic Implications Over Time of Platelet FcγRIIa Expression in Patients with Myocardial Infarction," has been selected by the Journal of Invasive Cardiology (JIC) as one of the Editors' Top 10 Articles of 2025.
The recognized research highlights the powerful prognostic value of the pFCG™ test in the critical months following a heart attack. The study found that patients with high platelet FcγRIIa expression faced a significantly higher risk of the composite endpoint of death, MI, or stroke, particularly within the first 30 days.
Expert Insight: Dr. Deepak L. Bhatt Interviews Dr. David Schneider
In conjunction with this recognition, Deepak L. Bhatt, MD, MPH, Editor-in-Chief of the Journal of Invasive Cardiology, hosted an in-depth interview with the study's lead author and Prolocor Co-Founder, David Schneider, MD, FACC, FAHA.
During the discussion, Dr. Schneider elaborated on how the FcγRIIa receptors on a patient's platelets acts as a biological "accelerant" for thrombosis, and how a greater number of these receptors is associated with a higher risk. identifying this high-risk phenotype can guide clinicians in individualizing dual antiplatelet therapy (DAPT).
Key Study Findings:
Early Risk Stratification: The pFCG™ test demonstrated its greatest prognostic power early after a myocardial infarction (MI), with a Hazard Ratio (HR) of 3.84 (P=.0009) in the first month.
Sustained Predictive Value: The HR remained high at 2.90 (P=.00005) through the first six months, effectively identifying patients at risk of recurrent events.
Clinical Utility: The test provides a precise tool for clinicians to balance ischemic and bleeding risks, potentially shifting the paradigm toward precision cardiovascular care.
"This recognition from the Journal of Invasive Cardiology validates the clinical importance of science behind our platform," said Peter DiBattiste, MD, CEO of Prolocor. "Our mission is to support physicians in optimizing antiplatelet therapy. By providing a clear biological marker of risk, we are giving cardiologists the data they need to tailor treatments specifically to the individual patient's biology, improving safety and efficacy across the continuum of care."
"We are honored that the editors have recognized the clinical significance of this work," added Dr. Schneider. "By identifying patients at the highest risk during the most vulnerable period following an MI, the pFCG™ test empowers physicians to make more informed, personalized treatment decisions."
About Prolocor, Inc.
Founded by a team that deeply understands thrombosis and cardiovascular disease, Prolocor is building its strategy around platelet FcγRIIa and embarking on a journey to commercialize an innovative precision diagnostic test that quantifies FcγRIIa on the surface of platelets. For more information on Prolocor, please visit the company's website at www.prolocor.com and follow us on LinkedIn.
Forward-Looking Statements
This press release contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts. These statements include projections and estimates and their underlying assumptions, statements regarding plans, objectives, intentions and expectations with respect to future financial results, events, operations, services, product development and potential, and statements regarding future performance. Forward-looking statements are generally identified by the words "expects," "anticipates," "believes," "intends," "estimates," "plans" and similar expressions. Although Prolocor's management believes that the expectations reflected in such forward-looking statements are reasonable, forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Prolocor, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements. These risks and uncertainties include among other things, Prolocor's planned level of revenues and capital expenditures, Prolocor's available cash and its ability to obtain additional funding, the uncertainties inherent in research and development, future clinical data and analysis, including post marketing, decisions by regulatory authorities regarding whether and when to approve any device or application that may be filed for any such product candidates and other matters that could affect the availability or commercial potential of such product candidates, the fact that product candidates if approved may not be commercially successful, the future approval and commercial success of therapeutic alternatives, Prolocor's ability to benefit from external growth opportunities, to complete related transactions and/or obtain regulatory clearances, and risks associated with intellectual property. Other than as required by applicable law, Prolocor does not undertake any obligation to update or revise any forward-looking information or statements.
Contact
SOURCE: Prolocor Inc
View the original press release on ACCESS Newswire
C.Garcia--AMWN

