-
 Rahm coasts to LIV Golf win in Mexico City
Rahm coasts to LIV Golf win in Mexico City
-
Fitzpatrick survives Scheffler playoff to win RBC Heritage

-
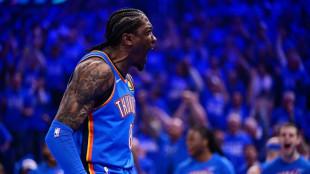 Thunder thrash Suns, Celtics crush Sixers in NBA playoff openers
Thunder thrash Suns, Celtics crush Sixers in NBA playoff openers
-
Bulgaria's former president tops parliamentary vote

-
 Kenyans Korir, Lokedi seek to repeat at Boston Marathon
Kenyans Korir, Lokedi seek to repeat at Boston Marathon
-
AC Milan, Juventus close in on Champions League qualification

-
 Spring double keeps Racing 92 in Top 14 play-off hunt with Paris derby win
Spring double keeps Racing 92 in Top 14 play-off hunt with Paris derby win
-
Endrick stars as Lyon dent PSG's Ligue 1 title hopes

-
 History haunts Arsenal as Man City take control of title race
History haunts Arsenal as Man City take control of title race
-
AC Milan and Juventus close in on Champions League qualification

-
 Celtics crush Sixers as Tatum and Brown shine in playoff opener
Celtics crush Sixers as Tatum and Brown shine in playoff opener
-
Guardiola warns title not won yet as Man City hunt down Arsenal

-
 Arteta tells Arsenal to 'go again' in pursuit of Premier League title
Arteta tells Arsenal to 'go again' in pursuit of Premier League title
-
Treble-chasing Bayern put beer showers on ice despite title win

-
 Eight children dead in US domestic violence shooting
Eight children dead in US domestic violence shooting
-
Arya, Connolly help Punjab hammer Lucknow in IPL

-
 Man City beat Arsenal to seize control of title race, Liverpool win
Man City beat Arsenal to seize control of title race, Liverpool win
-
Kane scores as Bayern sink Stuttgart to claim Bundesliga title

-
 Balogun continues Monaco scoring streak, Rennes boost Champions League hopes
Balogun continues Monaco scoring streak, Rennes boost Champions League hopes
-
Trump orders negotiators to Pakistan, but Iran on the fence over talks

-
 Haaland gives Man City edge over Arsenal in Premier League title showdown
Haaland gives Man City edge over Arsenal in Premier League title showdown
-
Slot hails Liverpool mentality after last-gasp derby winner

-
 Top boss vows 'no sitting still' as rugby bids to conquer US
Top boss vows 'no sitting still' as rugby bids to conquer US
-
Fils wins on Barcelona clay with French Open looming

-
 'Super Mario Galaxy' rules N. America box office for third week
'Super Mario Galaxy' rules N. America box office for third week
-
Liverpool snatch derby win ahead of City-Arsenal showdown

-
 Evenepoel outsprints Skjelmose to win Amstel Gold Race
Evenepoel outsprints Skjelmose to win Amstel Gold Race
-
Liverpool beat Everton ahead of City-Arsenal showdown

-
 Rabiot fires AC Milan past Verona to verge of Champions League return
Rabiot fires AC Milan past Verona to verge of Champions League return
-
UK PM vows to find arsonists of London Jewish sites

-
 Rinku blitz leads Kolkata to first win of IPL season
Rinku blitz leads Kolkata to first win of IPL season
-
Shelton wins fifth ATP title with victory in Munich

-
 UK's Starmer to face grilling from MPs over Mandelson scandal
UK's Starmer to face grilling from MPs over Mandelson scandal
-
Trump again threatens Iran infrastructure as he orders negotiators to Pakistan

-
 Rybakina outclasses Muchova to win Stuttgart WTA title
Rybakina outclasses Muchova to win Stuttgart WTA title
-
Blasi stuns field with victory in women's Amstel Gold Race

-
 Pakistan tightens security in Islamabad ahead of US-Iran talks
Pakistan tightens security in Islamabad ahead of US-Iran talks
-
Nagelsmann backs injured Gnabry as World Cup doubts grow

-
 Rampant South Africa tame Argentina to win Hong Kong Sevens at last
Rampant South Africa tame Argentina to win Hong Kong Sevens at last
-
Turkey 'optimistic' Middle East ceasefire will be extended

-
 Iran entrepreneurs angered by months-long internet blackout
Iran entrepreneurs angered by months-long internet blackout
-
UK PM says 'appalled' by arson attacks against Jewish sites in London

-
 Pope Leo XIV calls for 'hope' before 100,000 faithful in Angola
Pope Leo XIV calls for 'hope' before 100,000 faithful in Angola
-
Champions League or bust for Atletico after Copa del Rey agony

-
 Rat poison found in baby food jar in Austria as products recalled
Rat poison found in baby food jar in Austria as products recalled
-
Humans far behind as robot breaks record at Beijing half marathon

-
 Zelensky slams oil sanctions relief for Russia
Zelensky slams oil sanctions relief for Russia
-
Thousands gather for Pope Leo's first mass in Angola

-
 French billionaire shrugs off mass exodus at hallowed French publisher
French billionaire shrugs off mass exodus at hallowed French publisher
-
'DJ Priest' mixes religion and rave in Buenos Aires tribute to Pope Francis


BrYet US, Inc. Files IND Application for ML-016, Lead Oncology Therapeutic for Advanced Solid Tumors with Lung and/or Liver Metastases
HOUSTON, TX / ACCESS Newswire / March 4, 2026 / BrYet US, Inc. ("BrYet") - a biotechnology innovator focused on developing curative therapies for advanced cancers - announced today that it has filed an Investigational New Drug (IND) application with the U.S. Food and Drug Administration (FDA) for its lead drug, ML-016. Upon approval, the IND will support the expansion of BrYet's ML-016 clinical program with additional trials in the United States.
"Submitting our first IND application is a major step forward for BrYet, bringing us closer to delivering new therapeutic options for advanced cancer patients worldwide," said Dr. Mauro Ferrari, President and CEO of BrYet. "This milestone reflects decades of scientific research behind our unique approach to treating cancer, based on our proprietary platform for targeting phenotypes."
Last year, BrYet received Australian approval to begin its first-in-human clinical study of ML-016. The broadly scoped Phase I/II trial will enroll patients with any primary or metastatic cancer involving the lungs or liver, with the first patient dose expected to be administered in the second quarter of 2026. The U.S. IND will expand the clinical program with a focus on specific indications.
ML-016 has shown strong therapeutic efficacy in preclinical models, achieving long-term survival in about 50% of test animals in multiple metastatic tumor models. GLP preclinical data reconfirmed the strong safety profile of ML-016 and BrYet's platform for targeting phenotypes.
About ML-016
ML-016 is comprised of an amino acid polymer conjugated to doxorubicin with a formulation that includes BrYet's proprietary Si-PlateloidTM technology, designed to target the vascular endothelium of blood vessels in the tumor microenvironment. The amino acid-drug conjugate is released into the tumor, where it forms exosome-like vesicles.
These "exosomoids" are designed to be preferentially taken up by cancer cells including those that have previously been resistant to therapy. Through the mechanisms of cellular trafficking, the exosomoid vesicles are intended to be transported to late-stage endosomes and lysosomes, where the increased acidity releases the cytotoxic agent into the cell nucleus.
About BrYet US, Inc.
BrYet is a privately held biotechnology company developing potentially first-in-class therapies for patients suffering from cancers for which there is no current curative treatment. BrYet's lead asset, ML-016, is being developed for cancers of the lungs and liver, including advanced primary malignancies and metastatic spread from primary cancer that originates in other organs or tissue of the body. The company's fundamental belief is that upon localization in the lungs and liver, these cancers acquire molecular transport phenotypes that are conserved regardless of site of origin and are largely independent of molecular mutations and their continued evolution. BrYet designs multi-component new chemical entities and formulations, which are directed against the fundamental aspects of these cancer-associated, organ-specific transport phenotypes. The company's proprietary platforms include the Si-PlateloidTM and the mathematical formalism for designing the multi-component drugs, termed Transport Oncophysics. BrYet believes that similar approaches may provide advances against other forms of presently incurable cancers, as well as other pathologies of the lungs and liver. For more information about BrYet, please visit the company's website: https://bryetpharma.com/
Safe-Harbor Statement
This press release contains forward-looking statements concerning BrYet and its business. These statements are based on the beliefs of, assumptions made by, and information currently available to the company's management. When used in this document, the words "expects," "anticipates," "estimates," "intends," "believes," "plans," "predicts," "should," "could," "will," and similar expressions are intended to identify forward-looking statements.
Forward-looking statements are subject to risks and uncertainties that could cause actual results to differ from expectations. Factors that could contribute to these differences include the results of studies and clinical trials, regulatory approvals, challenges in clinical trials, the ability to retain employees, research and development expenses, reliance on third parties, intellectual property issues, competition, future funding needs, economic conditions, and other industry-specific risks. You should not place undue reliance on these statements, which are current as of the date of this press release. BrYet does not plan to update these statements unless legally required.
Media & Investor Contact
BrYet US, Inc.
2450 Holcombe Blvd., Suite 1520, Houston, TX 77021, USA
[email protected]
###
SOURCE: Bryet US Inc
View the original press release on ACCESS Newswire
M.A.Colin--AMWN