
-
 Central banks meet as Mideast war fuels inflation fears
Central banks meet as Mideast war fuels inflation fears
-
European bank battle heats up as UniCredit swoops for Commerzbank

-
 Oil eases on hopes for Strait of Hormuz passage
Oil eases on hopes for Strait of Hormuz passage
-
Race for Paris mayor on knife's edge after first round

-
 Denmark's election candidates bare all in sauna campaigning
Denmark's election candidates bare all in sauna campaigning
-
Russia targets Kyiv at rush hour, kills 3 across Ukraine

-
 Iran defiant as strikes hit Gulf transport, energy hubs
Iran defiant as strikes hit Gulf transport, energy hubs
-
Frenchman jailed in Azerbaijan for 10 years for 'spying'

-
 EU wants to tap citizens' savings. Easier said than done
EU wants to tap citizens' savings. Easier said than done
-
Record breaker Duplantis ready for return to 'special' Torun

-
 Italian bank UniCredit makes bid for Germany's Commerzbank
Italian bank UniCredit makes bid for Germany's Commerzbank
-
China says 'maintaining communication' with US over Trump visit

-
 Oil rises further above $100, most Asian stocks fall as Iran war rages
Oil rises further above $100, most Asian stocks fall as Iran war rages
-
Blasts rock Kyiv in rare daytime Russian attack: AFP

-
 Israel announces Lebanon ground assault against Hezbollah
Israel announces Lebanon ground assault against Hezbollah
-
AI to drive growth despite geopolitics, Taiwan's Foxconn says

-
 Indonesia police seek four suspects for acid attack on activist
Indonesia police seek four suspects for acid attack on activist
-
Iranian protesters fled 'hell' at home, watch war from exile

-
 'Rein in your heart': I.Coast women trapped by divorce taboo
'Rein in your heart': I.Coast women trapped by divorce taboo
-
Five top moments from the Oscars

-
 Filipinas seek abortions online in largely Catholic nation
Filipinas seek abortions online in largely Catholic nation
-
With Oscar race locked, actresses celebrate backstage anyway

-
 Chinese firms seek to loosen West's grip on lucrative snow business
Chinese firms seek to loosen West's grip on lucrative snow business
-
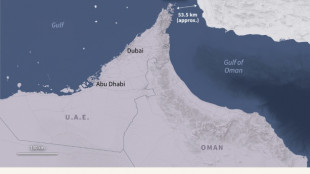
Japan not currently mulling maritime ops despite Trump pressure over Hormuz
-
 'One Battle After Another' wins best picture Oscar
'One Battle After Another' wins best picture Oscar
-
South Koreans bask in Oscars triumph for 'KPop Demon Hunters'

-
 'One Battle After Another' dominates Oscars
'One Battle After Another' dominates Oscars
-
Oil hovers around $100, stocks mixed as Iran war rages

-
 Future looks bright for Ireland and Farrell, says ex-Irish prop Ross
Future looks bright for Ireland and Farrell, says ex-Irish prop Ross
-
Epic Six Nations raises defensive questions before Rugby World Cup

-
 Ryan Coogler: from indie to blockbuster to Oscar
Ryan Coogler: from indie to blockbuster to Oscar
-
English sides seek redemption from Champions League reality check

-
 Jessie Buckley: From reality TV hopeful to Oscar winner
Jessie Buckley: From reality TV hopeful to Oscar winner
-
Paul Thomas Anderson: eclectic filmmaker, critical darling

-
 Michael B. Jordan battles his way to Oscar for 'Sinners'
Michael B. Jordan battles his way to Oscar for 'Sinners'
-
20 Thai sailors return home after vessel attacked in Gulf

-
 Norway's Oscar winner 'Sentimental Value': a failing father seeks redemption
Norway's Oscar winner 'Sentimental Value': a failing father seeks redemption
-
Races off, Mercedes dominance, McLaren woe: China GP talking points

-
 Indonesia firms in palm oil fraud probe supplied fuel majors
Indonesia firms in palm oil fraud probe supplied fuel majors
-
Sean Penn: Hollywood's rebel with a cause wins third Oscar

-
 It's 'Sinners' vs 'One Battle' as the Oscars begin
It's 'Sinners' vs 'One Battle' as the Oscars begin
-
Magdalena Biosciences, a Joint Venture Between Jaguar Health and Filament Health, Initiates Preclinical Study of Coca for Appetite Suppression

-
 iFabric Corp Provides Strong 2025 Revenue Estimate and 2026 Outlook
iFabric Corp Provides Strong 2025 Revenue Estimate and 2026 Outlook
-
Tactical Resources Signs Agreement to Acquire the Sierra Blanca Quarry and Tailings Access for the Peak Rare Earth Project

-
 CuriosityStream to Participate at the 38th Annual Roth Conference
CuriosityStream to Participate at the 38th Annual Roth Conference
-
GILi Guise(R) Gains Nationwide TV Coverage for Its Innovative 'Grow-With-Me' Kids Denim

-
 Kingsway Names Adam J. Patinkin as Chairman of Its Board of Directors
Kingsway Names Adam J. Patinkin as Chairman of Its Board of Directors
-
Ondas Completes Acquisition of Rotron Aerospace Ltd., Expanding Long-Range Propulsion Technologies for Advanced Unmanned Systems for Defense

-
 Elektros Opens Global Licensing Dialogue for Breakthrough Multi‑Source EV Charging Patent
Elektros Opens Global Licensing Dialogue for Breakthrough Multi‑Source EV Charging Patent
-
SKYLINE Announces Financial Results for the Year Ended December 31, 2025


uBriGene Congratulates Strategic Partner Grit Biotechnologies on FDA IND Clearance for Its Next-Generation GT307 TIL Therapy
GERMANTOWN, MD / ACCESS Newswire / March 16, 2026 / uBriGene Biosciences (uBriGene), a global CDMO leader, is excited to announce that its strategic partner, Grit Biotechnologies, has received FDA clearance for its Investigational New Drug (IND) application on GT307, a next-generation gene-edited Tumor-Infiltrating Lymphocytes (TIL) therapy.
uBriGene played a key role in enabling this milestone, providing tech transfer, process development, GMP manufacturing, and regulatory support to advance GT307 from development to clinical readiness.
"uBriGene leveraged its TIL-TurboTM production platform, a fully closed and automated system that minimizes variability, reduces contamination risk, and ensures reproducible cell expansion," said Xiulian Sun, CTO, uBriGene Biosciences. "Using this platform, we successfully completed process transfer, process verification, and GMP manufacturing at our Maryland facility, delivering high-quality material ready for first-in-human studies."

"GT307 employs high-fidelity CRISPR/AaCas12bMAX gene editing to knock out key immune regulatory genes, designed to address key bottlenecks of conventional TIL therapies, such as TIL exhaustion within the tumor microenvironment and insufficient persistence in vivo," said Dr. Yarong Liu, CEO, Grit Biotechnologies. "This precise gene-editing strategy, combined with the extensive cell and gene therapy manufacturing experience of our partner uBriGene, allows us to advance next-generation TIL therapies efficiently and safely into the clinic."
The platform has successfully completed more than ten GMP TIL manufacturing runs and enables robust, high-yield expansion within approximately three weeks, consistently achieving cell yields that meet clinical-grade requirements.
About Grit Biotechnologies
Founded in 2019, Grit Biotechnologies is an innovative immunotherapy company focused on TIL and off-the-shelf cell therapies. Its GT101 is China's first TIL therapy approved for clinical trials, now in pivotal Phase II studies. The company's GT201, a membrane-bound IL-15 complex-engineered TIL therapy, has completed dual submissions in China and the U.S.
Grit Biotechnologies' core platforms include StemTexp® for TIL expansion, StaViral® viral stable line technology, ImmuT Finder® immune target discovery, and KOReTIL® high-efficiency gene knockout system. Leveraging these capabilities, the company is advancing next-generation gene-edited TIL therapies into clinical studies for patients in difficult-to-treat solid tumors.
About uBriGene Biosciences
Founded in 2015, uBriGene Biosciences is a leading Contract Development and Manufacturing Organization for advanced therapeutic medicinal products (ATMPs). The company provides integrated CDMO and CRO solutions, encompassing services for cell therapy products, viral vectors, and RNA-related products, with in-house QC testing and regulatory IND filing. Our global GMP Centers of Excellence, AAV-Turbo manufacturing platform, LVV Turbo manufacturing platform, TIL-Turbo manufacturing platform, iPSC Reprogramming technologies are propelling many biotech and pharmaceutical companies forward successfully.
Contact
Mingjuan Liu
Director of Marketing
[email protected]
240-551-5140
SOURCE: UBRIGENE BIOSCIENCES INC
View the original press release on ACCESS Newswire
A.Mahlangu--AMWN

