
-
 War in Middle East: latest developments
War in Middle East: latest developments
-
No cadmium please: French want less toxin in their baguettes

-
 Warsh set to take over a divided Fed facing Trump assaults
Warsh set to take over a divided Fed facing Trump assaults
-
Shots heard at Philippine Senate as lawmaker wanted by ICC holds out

-
 France locks down 1,700 on cruise ship after 90-year-old dies
France locks down 1,700 on cruise ship after 90-year-old dies
-
After the hobbits, director Peter Jackson tackles 'Tintin'

-
 Real Madrid win legal battle over Bernabeu concert noise
Real Madrid win legal battle over Bernabeu concert noise
-
EU won't ban LGBTQ 'conversion therapy' but will push states to act

-
 Revived Swiatek cruises past Pegula and into Italian Open semis
Revived Swiatek cruises past Pegula and into Italian Open semis
-
Shots heard at Philippine Senate as lawmaker wanted by ICC holds out: AFP

-
 Vin Diesel drives 'Fast and Furious' tribute in Cannes
Vin Diesel drives 'Fast and Furious' tribute in Cannes
-
Heckler ejected from Eurovision after Israel song disruption

-
 Australia's North savours 'tremendous honour' of England role
Australia's North savours 'tremendous honour' of England role
-
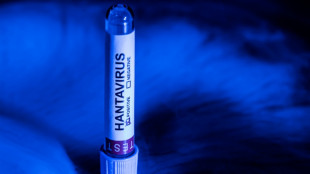
For hantavirus, experts aim to inform without igniting Covid panic
-
 Japan rides box office boom into Cannes
Japan rides box office boom into Cannes
-
Trump arrives in China for superpower summit with Xi

-
 UK's Catherine on first official foreign trip since cancer diagnosis
UK's Catherine on first official foreign trip since cancer diagnosis
-
British scientists among winners of top Spanish award

-
 Mbappe can show 'commitment' to Real Madrid: Arbeloa
Mbappe can show 'commitment' to Real Madrid: Arbeloa
-
Chinese tech giant Alibaba posts profit drop amid AI drive

-
 King Charles lays out Starmer's agenda as PM fights for survival
King Charles lays out Starmer's agenda as PM fights for survival
-
Japan suspend Eddie Jones for verbally abusing officials

-
 England drop Crawley for 1st Test against New Zealand
England drop Crawley for 1st Test against New Zealand
-
Stocks rise ahead of US-China summit as Iran talks stall

-
 One trip, one ticket: New EU rules aim to ease train travel
One trip, one ticket: New EU rules aim to ease train travel
-
SoftBank profit quadruples to $32 bn on AI investments

-
 Africa must drop 'victim mentality': mogul Tony Elumelu
Africa must drop 'victim mentality': mogul Tony Elumelu
-
'Ungovernable' Britain? Once-stable politics in freefall

-
 China tech giant Tencent sees Q1 profit jump after AI bets
China tech giant Tencent sees Q1 profit jump after AI bets
-
Nissan expects return to profit after huge loss

-
 World Cup broadcast deadlock ends up in Indian court
World Cup broadcast deadlock ends up in Indian court
-
Asian stocks mixed on US-Iran impasse, AI setbacks

-
 Besieged Starmer seeks to heal Labour divisions in King's Speech
Besieged Starmer seeks to heal Labour divisions in King's Speech
-
After winter storms, fires now threaten Portugal's forests

-
 Philippine senator seeks military support to block ICC drug war arrest
Philippine senator seeks military support to block ICC drug war arrest
-
UK's Catherine on first official foreign trip since cancer revelation

-
 'Short of blue-collar workers': Ukraine's battle for labour
'Short of blue-collar workers': Ukraine's battle for labour
-
'Don't understand it, but it looks fun': cricket bowls Japan over

-
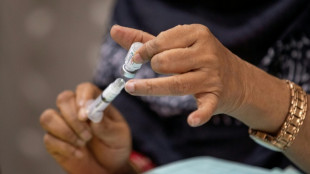 Poor planning fuels Bangladesh contraceptive crisis
Poor planning fuels Bangladesh contraceptive crisis
-
Fugitive financier sought in Malaysian fund scandal seeks Trump's pardon

-
 World Cup comes to 'Soccer Town USA,' but locals priced out
World Cup comes to 'Soccer Town USA,' but locals priced out
-
Don't mention the war: Tucson prepares to welcome Team Iran for World Cup

-
 Hosting World Cup evokes powerful memories for Mexico, and raises expectations
Hosting World Cup evokes powerful memories for Mexico, and raises expectations
-
AI rivalry overshadows push for guardrails at Xi-Trump talks: experts

-
 Asian stocks fall on US-Iran impasse, AI setbacks
Asian stocks fall on US-Iran impasse, AI setbacks
-
Wembanyama leads Spurs to brink as Timberwolves routed

-
 Ronaldo left waiting for Saudi title after goalkeeping gaffe
Ronaldo left waiting for Saudi title after goalkeeping gaffe
-
'Not my son's fault': The women bearing the children of Sudan's war rapes

-
 'I applied to be pope': Losing grip on reality while using ChatGPT
'I applied to be pope': Losing grip on reality while using ChatGPT
-
EU to ease train travel with one journey, one ticket rules


Xenetic Biosciences Reports First Quarter 2026 Financial Results and Highlights Continued Advancement of DNase Oncology Platform
Advancements across investigator-initiated studies, translational research and manufacturing activities supporting future clinical development
Strengthened financial profile with continued royalty revenue growth
Ended first quarter 2026 with approximately $7.3 million in cash while continuing evaluation of strategic alternatives to maximize shareholder value
FRAMINGHAM, MA / ACCESS Newswire / May 13, 2026 / Xenetic Biosciences, Inc. (NASDAQ:XBIO) ("Xenetic" or the "Company"), a biopharmaceutical company focused on advancing innovative immuno-oncology technologies addressing difficult to treat cancers, today reported its financial results for the quarter ended March 31, 2026 and provided a corporate update on the advancement of its innovative DNase oncology platform.
Recent Highlights
Generated robust translational data supporting the DNase I platform across multiple oncology settings
Completed process improvement and analytical development activities supporting future clinical manufacturing readiness
Established translational and biomarker insights intended to help inform and de-risk future clinical trial design
Collaboration partner received approval from Israeli Ministry of Health to conduct investigator-initiated exploratory study of DNase I in combination with anti-CD19 CAR T cells in large B-cell lymphoma
"We entered 2026 with a clear focus on advancing the translational, manufacturing and clinical foundation of our DNase I platform while maintaining financial discipline," said James Parslow, Interim Chief Executive Officer and Chief Financial Officer of Xenetic. "During the quarter, we achieved important progress across investigator-initiated studies, translational research and manufacturing readiness activities. We believe the growing body of translational evidence supporting NET-targeting approaches, combined with the expanding understanding of NETs as drivers of tumor progression, immune suppression and therapy resistance, continues to strengthen the potential opportunity for DNase I as a differentiated adjunctive immuno-oncology therapy across multiple cancer settings."
Xenetic's proprietary DNase technology is being developed as a potential adjunctive therapeutic approach designed to improve the effectiveness of existing cancer treatments, including immunotherapies, through the targeting of NETs, which are increasingly recognized as key drivers of immune suppression and therapeutic resistance within the tumor microenvironment.
Summary of Financial Results for First Quarter 2026
Royalty revenue for the three months ended March 31, 2026 increased approximately 36% to approximately $0.8 million, compared to approximately $0.6 million for the comparable prior year period, primarily driven by increased royalty payments recognized under the Company's sublicense agreement with Takeda Pharmaceuticals Co. Ltd.
Research and development expenses for the three months ended March 31, 2026 decreased approximately 25% to approximately $0.7 million from approximately $0.9 million for the comparable prior year period, primarily due to lower preclinical and exploratory study costs, partially offset by increased manufacturing development efforts supporting the Company's DNase I program.
General and administrative expenses for the three months ended March 31, 2026 decreased by approximately 1.4%, to approximately $0.6 million from approximately $0.7 million in the comparable quarter in 2025. The decrease was primarily due to a decrease in personnel costs and share-based expenses related to our interim Chief Executive Officer substantially offset by an increase in legal expenses related to our strategic review process during the first quarter of 2026 compared to the same period in 2025.
Net loss for the quarter ended March 31, 2026 decreased approximately 49% to approximately $0.5 million, compared to approximately $0.9 million for the same period in 2025.
The Company ended the first quarter of 2026 with approximately $7.3 million in cash and cash equivalents, compared to approximately $7.9 million as of December 31, 2025.
About Xenetic Biosciences
Xenetic Biosciences, Inc. is a biopharmaceutical company focused on advancing innovative immuno-oncology technologies addressing difficult to treat cancers. The Company's proprietary DNase technology is designed to improve outcomes of existing treatments, including immunotherapies, by targeting neutrophil extracellular traps (NETs), which are involved in cancer progression. Xenetic is currently focused on advancing its systemic DNase program into the clinic as an adjunctive therapy for pancreatic carcinoma and locally advanced or metastatic solid tumors.
For more information, please visit the Company's website at www.xeneticbio.com and connect on X, LinkedIn, and Facebook.
Forward-Looking Statements
This press release contains forward-looking statements that we intend to be subject to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. All statements contained in this press release other than statements of historical facts may constitute forward-looking statements within the meaning of the federal securities laws. These statements can be identified by words such as "expects," "plans," "projects," "will," "may," "anticipates," "believes," "should," "intends," "estimates," "remain," "focus", "confidence in", "potential", "continues", "warrants", and other words of similar meaning, including, but not limited to, all statements regarding future clinical development, manufacturing readiness and trial design, our focus on advancing innovative immuno-oncology technologies addressing difficult to treat cancers, the DNase platform improving outcomes of existing treatments, including immunotherapies, by targeting neutrophil extracellular traps (NETs), which are involved in cancer progression, our belief regarding strengthening the potential opportunity for DNase I as an adjunctive therapy across multiple cancer settings and our focus on advancing DNase into the clinic as an adjunctive therapy for pancreatic carcinoma and locally advanced or metastatic solid tumors. Any forward-looking statements contained herein are based on current expectations and are subject to a number of risks and uncertainties. Many factors could cause our actual activities, performance, achievements, or results to differ materially from the activities and results anticipated in forward-looking statements. Important factors that could cause actual activities, performance, achievements, or results to differ materially from such plans, estimates or expectations include, among others, (1) unexpected costs, charges or expenses resulting from our manufacturing and collaboration agreements; (2) unexpected costs, charges or expenses resulting from the licensing of the DNase platform; (3) uncertainty of the expected financial performance of the Company following the licensing of the DNase platform; (4) failure to realize the anticipated potential of the DNase or PolyXen technologies; (5) the ability of the Company to obtain funding and implement its business strategy; (6) risks and uncertainties as to the outcome and timing of the strategic review process being conducted by the Board and a special independent committee thereof, including the possibility that the Board may decide not to undertake a strategic alternative following the evaluation process, the Company's inability to consummate any proposed strategic alternative resulting from the review due to, among other things, market, regulatory and other factors, the potential for disruption to our business resulting from the review process, and potential adverse effects on the Company's stock price from the announcement, suspension or consummation of the evaluation process and the results thereof, as well as risks and uncertainties related to the potential impacts of consummation of a strategic transaction on the Company's current business operations, anticipated business strategy and product development plans; and (7) other risk factors as detailed from time to time in the Company's reports filed with the SEC, including its annual report on Form 10-K, periodic quarterly reports on Form 10-Q, current reports on Form 8-K and other documents filed with the SEC. The foregoing list of important factors is not exclusive. In addition, forward-looking statements may also be adversely affected by general market factors, general economic and business conditions, including potential adverse effects of public health issues, and geopolitical events, such as the conflicts in Ukraine and in the Middle East, on economic activity, competitive product development, product availability, federal and state regulations and legislation, the regulatory process for new product candidates and indications, manufacturing issues that may arise, patent positions, litigation, and shareholder activism, among other factors. The forward-looking statements contained in this press release speak only as of the date the statements were made, and the Company does not undertake any obligation to update forward-looking statements, except as required by law.
Contact:
JTC Team, LLC
Jenene Thomas
(908) 824-0775
[email protected]
SOURCE: Xenetic Biosciences, Inc.
View the original press release on ACCESS Newswire
Ch.Kahalev--AMWN