
-
 Sinner, defending champ Mensik advance to third round at Miami Open
Sinner, defending champ Mensik advance to third round at Miami Open
-
Iran missile strikes wound over 100 in two south Israel towns

-
 Shai hits 40 as Thunder win despite NBA melee with four ejected
Shai hits 40 as Thunder win despite NBA melee with four ejected
-
Records shattered as US heatwave moves eastward

-
 Iran missiles hit southern Israel, injuring more than 100
Iran missiles hit southern Israel, injuring more than 100
-
LeBron James breaks record for most NBA games played

-
 'Perfect' PSG sweep past Nice to reclaim top spot in Ligue 1
'Perfect' PSG sweep past Nice to reclaim top spot in Ligue 1
-
Japan coach says Asian Cup crown 'well-deserved' for inspirational team

-
 PSG sweep past Nice to reclaim top spot in Ligue 1
PSG sweep past Nice to reclaim top spot in Ligue 1
-
Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81

-
 Milan move to within five points of Serie A leaders Inter
Milan move to within five points of Serie A leaders Inter
-
Duplantis masterclass as Kerr and record-setter Ehammer shine

-
 Rosenior urges Chelsea to 'forget the noise' after damaging loss
Rosenior urges Chelsea to 'forget the noise' after damaging loss
-
Marquez ambushed Di Giannantonio to win Brazil sprint

-
 Sweden's Duplantis wins fourth world indoor pole vault title
Sweden's Duplantis wins fourth world indoor pole vault title
-
Liverpool, Chelsea slip up in Champions League race

-
 WHO sends first overland convoy from emergencies hub to Beirut
WHO sends first overland convoy from emergencies hub to Beirut
-
Everton rub salt in Chelsea wounds as Champions League race tightens

-
 Coach Mignoni returns but Toulon crash to Stade Francais
Coach Mignoni returns but Toulon crash to Stade Francais
-
Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81

-
 Sinner and Pegula advance to third round at Miami Open
Sinner and Pegula advance to third round at Miami Open
-
Britain's Kerr outsprints Hocker for world indoor 3,000m gold

-
 Kane backs Tuchel's call to rest him from England friendly
Kane backs Tuchel's call to rest him from England friendly
-
NBA fines 76ers' Drummond, Magic's Suggs $25,000 each

-
 Switzerland's Ehammer sets indoor heptathlon world record
Switzerland's Ehammer sets indoor heptathlon world record
-
Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set

-
 World Athletics decision to hand Asia two world indoors 'strategic' - Coe
World Athletics decision to hand Asia two world indoors 'strategic' - Coe
-
Trump threatens to use ICE agents for airport security control

-
 Kane moves closer to goals record as Bayern sink Union
Kane moves closer to goals record as Bayern sink Union
-
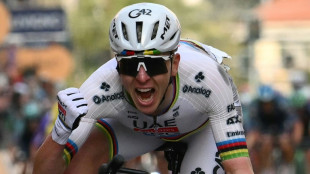
Pogacar ends long wait for Milan-San Remo glory after edging epic
-
 US says 'took out' Iran base threatening blocked Hormuz oil route
US says 'took out' Iran base threatening blocked Hormuz oil route
-
Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez

-
 Welbeck scores twice to dent Liverpool's top-five hopes
Welbeck scores twice to dent Liverpool's top-five hopes
-
US strikes Iran bases threatening blocked Hormuz oil route

-
 Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin
Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin
-
Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast

-
 K-pop kings BTS stun Seoul in '2.0' comeback concert
K-pop kings BTS stun Seoul in '2.0' comeback concert
-
French prosecutors suspect Musk encouraged deepfakes row to inflate X value

-
 Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa
Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa
-
Iranians mark Eid as Tehran reports strike on nuclear plant

-
 Kenya, Uganda open rail extension burdened by Chinese debt
Kenya, Uganda open rail extension burdened by Chinese debt
-
K-pop kings BTS rock Seoul in comeback concert

-
 Invincible Japan edge Australia to win Women's Asian Cup
Invincible Japan edge Australia to win Women's Asian Cup
-
Italy's Paris claims first win of season in World Cup downhill finale

-
 In Finland, divers learn to explore icy polar waters
In Finland, divers learn to explore icy polar waters
-
Dortmund extend injured captain Can's contract

-
 Iranians mark Eid as Trump mulls winding down war
Iranians mark Eid as Trump mulls winding down war
-
Matisse's last years cut out -- but not pasted -- at Paris expo

-
 BTS fans take over central Seoul for K-pop kings' comeback
BTS fans take over central Seoul for K-pop kings' comeback
-
Star jockey McDonald becomes horse racing's most prolific Group 1 winner


Protagenic Therapeutics Announces Completion of Enrollment and Dosing in Phase 1 MAD Study
NEW YORK CITY, NY / ACCESS Newswire / November 13, 2025 / Protagenic Therapeutics Inc. (NASDAQ:PTIX), a biopharmaceutical company developing novel therapeutics for stress-related neuropsychiatric and neurodegenerative disorders, today announced the successful completion of enrollment and dosing in its Multiple Ascending Dose (MAD) Phase 1 study evaluating its lead compound, known as PT00114. The study, conducted in healthy volunteers, was designed to assess the safety and tolerability profile of PT00114 following multiple doses over time. With all healthy volunteers now dosed, the company has achieved a milestone in the clinical development of its proprietary peptide-based therapeutic.
"We've completed all dosing for all healthy volunteers in the Phase 1 Study," said Dr. Garo Armen, Executive Chairman of Protagenic Therapeutics. "We plan to deliver a full summation of our analysis of the safety results within by the end of the month."
The company expects to finalize and release top-line results from the MAD study by November 30th.
"This milestone underscores our commitment to advancing PT00114 as a novel, first-in-class therapeutic aimed at restoring resilience and balance in individuals suffering from chronic stress and its neurological consequences," added Dr. Armen. "The company had previously completed a single ascending dose study in early 2025. We are encouraged by the progress to date and are excited to move toward the next phase of development."
About Protagenic Therapeutics, Inc.
Protagenic Therapeutics, Inc. (NASDAQ:PTIX) is a biopharmaceutical company focused on developing innovative peptide-based therapeutics targeting the biological pathways underlying stress-related neurological and mood disorders. The company's lead compound, PT00114, is a synthetic analog of a naturally occurring brain peptide that helps regulate the body's stress response and emotional equilibrium. For more information, visit www.protagenic.com.
Forward-Looking Statements
Statements in this press release contain "forward-looking statements," within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, that are subject to substantial risks and uncertainties. All statements, other than statements of historical fact, contained in this press release are forward-looking statements. Forward-looking statements contained in this press release may be identified by the use of words such as "anticipate," "believe," "contemplate," "could," "estimate," "expect," "intend," "seek," "may," "might," "plan," "potential," "predict," "project," "suggest," "target," "aim," "should," "will," "would," or the negative of these words or other similar expressions, although not all forward-looking statements contain these words. Forward-looking statements are based on the Company's current expectations and are subject to inherent uncertainties, risks and assumptions that are difficult to predict, including risks related to the completion, timing and results of current and future clinical studies relating to the Company's product candidates. Further, certain forward-looking statements are based on assumptions as to future events that may not prove to be accurate. These and other risks and uncertainties are described more fully in the section titled "Risk Factors" in the Company's Annual Report on Form 10-K for the year ended December 31, 2024, filed with the Securities and Exchange Commission. Forward-looking statements contained in this announcement are made as of this date, and the Company undertakes no duty to update such information except as required under applicable law.
Company Contact:
Alexander K. Arrow, MD, CFA
Chief Financial Officer
Protagenic Therapeutics, Inc.
149 Fifth Ave, Suite 500, New York, NY 10010
Tel: 213-260-4342
Email: [email protected]
SOURCE: Protagenic Therapeutics, Inc.
View the original press release on ACCESS Newswire
A.Rodriguezv--AMWN
