
-
 Coach Valverde to leave Bilbao at end of season
Coach Valverde to leave Bilbao at end of season
-
'Decimated'? The Iranian leaders killed in Israeli-US war

-
 Mistral chief calls for European AI levy to pay creatives
Mistral chief calls for European AI levy to pay creatives
-
Liverpool suffer Salah blow in chase for Champions League

-
 Mahuchikh soars to world indoor high jump gold, Hodgkinson cruises
Mahuchikh soars to world indoor high jump gold, Hodgkinson cruises
-
Spain include Joan Garcia as one of four new call-ups

-
 Salah ruled out of Liverpool's Brighton clash
Salah ruled out of Liverpool's Brighton clash
-
Ship crews ration food in Iran blockade: seafarers

-
 Kuwait refinery hit as Iran marks New Year under shadow of war
Kuwait refinery hit as Iran marks New Year under shadow of war
-
England recall Mainoo, Maguire for pre-World Cup matches

-
 Jerusalem's Muslims despair as war shuts Al-Aqsa Mosque for Eid
Jerusalem's Muslims despair as war shuts Al-Aqsa Mosque for Eid
-
'War has aged us': Lebanon's kids aren't alright

-
 Snooker great O'Sullivan makes history with highest-ever break
Snooker great O'Sullivan makes history with highest-ever break
-
Kuwait refinery hit as Iran says missile production 'no concern'

-
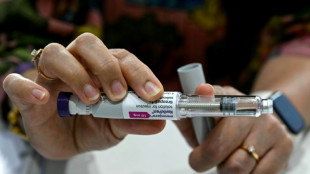 India to tackle global obesity with cheap fat-loss jabs
India to tackle global obesity with cheap fat-loss jabs
-
Somaliland centre saves cheetahs from trafficking to Gulf palaces

-
 China swim sensation Yu, 13, beats multiple Olympic medallist
China swim sensation Yu, 13, beats multiple Olympic medallist
-
North Korean leader, daughter try out new tank

-
 Israel strikes 'decimated' Iran as war roils markets
Israel strikes 'decimated' Iran as war roils markets
-
James ties NBA record for most regular-season games in latest milestone

-
 Trump's Mideast muddle could play into Xi's hands at planned summit
Trump's Mideast muddle could play into Xi's hands at planned summit
-
Wembanyama lifts playoff-bound Spurs, Doncic and James fuel Lakers

-
 Japan ski paradise faces strains of global acclaim
Japan ski paradise faces strains of global acclaim
-
Vinicius, Real Madrid must prove consistency in Atletico derby

-
 Kane credits Kompany's Bayern 'evolution' as treble beckons
Kane credits Kompany's Bayern 'evolution' as treble beckons
-
PSG look back to their best, but not yet out of sight in Ligue 1

-
 Weakened WTO set for high-level meet under cloud of Mideast war
Weakened WTO set for high-level meet under cloud of Mideast war
-
New BTS album to drop ahead of comeback mega-gig

-
 Troubled Spurs face Forest showdown, Chelsea need top-four surge
Troubled Spurs face Forest showdown, Chelsea need top-four surge
-
Australia must be 'smart and adapt' to beat Japan in Asian Cup final: coach

-
 From bats to bonds: Uganda's 'cricket grannies'
From bats to bonds: Uganda's 'cricket grannies'
-
Turkey in cultural diplomacy push to bring history home

-
 'The Bachelorette' canned after star's violent video emerges
'The Bachelorette' canned after star's violent video emerges
-
Trump gets approval for gold coin in his likeness

-
 Behind the BTS comeback, the dark side of K-pop
Behind the BTS comeback, the dark side of K-pop
-
Crude sinks after Netanyahu tries to reassure on Iran war

-
 Three charged with sneaking Nvidia AI chips from US into China
Three charged with sneaking Nvidia AI chips from US into China
-
Swiatek stunned at Miami Open by 50th-ranked Linette

-
 Italy, Germany and France offer help with Hormuz only after ceasefire
Italy, Germany and France offer help with Hormuz only after ceasefire
-
US-backed airstrikes leave Ecuador border communities in fear

-
 'Blackmail': EU leaders round on Orban for stalling Ukraine loan
'Blackmail': EU leaders round on Orban for stalling Ukraine loan
-
Displacement, bombs and air raid sirens weigh on Mideast Eid celebrations

-
 James ties NBA record for most regular-season games played
James ties NBA record for most regular-season games played
-
BTS to drop new album ahead of comeback mega-gig

-
 Elektros Energy Advisory Division Eyes Enterprise Market with AI-Powered Efficiency Solutions
Elektros Energy Advisory Division Eyes Enterprise Market with AI-Powered Efficiency Solutions
-
TRNR Authorizes First Stock Repurchase Program; $500,000 Program Represents 20% Of Shares Outstanding at Last Closing Price

-
 Ondas Reports Revised Preliminary Fourth Quarter and Full Year 2025 Financial Results and will Report Final Results on March 25, 2026 at 8:30 a.m. ET
Ondas Reports Revised Preliminary Fourth Quarter and Full Year 2025 Financial Results and will Report Final Results on March 25, 2026 at 8:30 a.m. ET
-
Unusual Machines Announces Pricing of Approximately $150 Million Public Offering of Common Stock

-
 IRS Debt Can Block Some Professional Licenses - Clear Start Tax Explains the Rules Workers Rarely Hear About
IRS Debt Can Block Some Professional Licenses - Clear Start Tax Explains the Rules Workers Rarely Hear About
-
Applied Energetics to Participate in Two Industry Panels at the 38th Annual ROTH Conference


Altamira Therapeutics Announces Market Approval of Bentrio Nasal Spray in China
Regulatory clearance of Bentrio in allergic rhinitis for Mainland China
Nearly 200 million Chinese estimated to suffer from allergic rhinitis
Bentrio to be launched by partner Nuance Pharma
HAMILTON, BERMUDA / ACCESS Newswire / December 19, 2025 / Altamira Therapeutics Ltd. ("Altamira" or the "Company") (OTCID:CYTOF), a company dedicated to developing and supplying nucleic acid delivery technology for partners in biotech and pharma, today announced that its associate company Altamira Medica ("Medica") has obtained marketing approval from the National Medical Products Administration (NMPA) of China for its Bentrio® nasal spray in allergic rhinitis. The clearance will allow Nuance Pharma, Medica's licensee and distribution partner, to start marketing and distributing Bentrio in Mainland China. According to epidemiological studies, nearly 200 million people in China are estimated to be affected by allergic rhinitis, and prevalence keeps increasing. [1]
"We are very excited by the regulatory clearance of Bentrio for China, which is one of the world's largest markets for 'over the counter' consumer health products," commented Thomas Meyer, Medica's Chairman and CEO. "Thanks to its triple mode of action, which does not require any drug ingredient for protecting against airborne allergens, and its preservative-free formulation, Bentrio has demonstrated strong efficacy and good tolerability across several allergic rhinitis trials. We look forward to supporting the launch of the product in China by our esteemed partner Nuance Pharma, which has built a strong and growing franchise in consumer health and in respiratory diseases."
"The regulatory approval of Bentrio represents an important milestone for Nuance Pharma's effort in the allergy space" commented Mark G. Lotter, CEO and Founder of Nuance Pharma. "Building upon Nuance's existing efforts on Bentrio in the region and commercial capabilities in China, we are excited to broaden the access of Bentrio to benefit the allergic rhinitis patients in China."
About Bentrio
Bentrio is an "over the counter" drug-free nasal spray for personal protection against airborne allergens and, where approved, against airborne viruses. Upon application into the nose, Bentrio forms a protective gel layer on the nasal mucosa. This thin film is designed to prevent the contact of airborne particles with cells; in addition, the composition serves to bind such particles and help with their discharge. The efficacy and safety of Bentrio have been demonstrated in a total of four clinical trials, of which the largest one ("NASAR" study) enrolled 100 patients suffering from seasonal allergic rhinitis. In NASAR, participants self-administered either Bentrio or saline nasal spray for two weeks 3 times per day. The study showed a statistically significant reduction in the mean daily reflective Total Nasal Symptom Score (rTNSS) for Bentrio compared to saline (p = 0.013), as well as a statistically highly significant improvement in health-related quality of life (Rhinoconjunctivitis Quality of Life Questionnaire, p www.bentrio.com
About Altamira Therapeutics
Altamira Therapeutics is developing and supplying peptide-based nanoparticle technologies for efficient nucleic acid delivery (xPhore™ platform). The versatile delivery platform is suited for different nucleic acid modalities, including siRNA, mRNA, circRNA, as well as DNA, and made available to pharma or biotech companies through out-licensing. The Company has two proprietary flagship programs based on xPhore and siRNA payloads: AM-401 for KRAS driven cancer and AM-411 for rheumatoid arthritis, both in preclinical development beyond in vivo proof of concept. In addition, Altamira holds a 49% stake (with additional economic rights) in Altamira Medica AG, which owns its commercial-stage legacy asset Bentrio, an OTC nasal spray for allergic rhinitis. Further, the Company is in the process of partnering / divesting its inner ear legacy assets. Founded in 2003, Altamira is headquartered in Hamilton, Bermuda, with its main operations in Basel, Switzerland. For more information, visit: https://altamiratherapeutics.com/
About Nuance Pharma
Nuance Pharma is an innovation focused biopharmaceutical company, with both late-stage clinical pipeline and commercial stage asset portfolio. Focusing on specialty care, Nuance has established a differentiated combination of commercialized assets and innovative pipeline across respiratory, allergy, emergency care and iron deficiency anemia. With the mission to address critical unmet medical needs in Asia Pacific, Nuance deploys the Dual Wheel model that develops a global leading innovative pipeline, while maintaining a self-sustainable commercial operation in both China and Asia as a region. For more information, visit: https://www.nuancepharma.com/
Forward-Looking Statements
This press release may contain statements that constitute "forward-looking statements" within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Forward-looking statements are statements other than historical facts and may include statements that address future operating, financial or business performance or Altamira's strategies or expectations. In some cases, you can identify these statements by forward-looking words such as "may", "might", "will", "should", "expects", "plans", "anticipates", "believes", "estimates", "predicts", "projects", "potential", "outlook" or "continue", or the negative of these terms or other comparable terminology. Forward-looking statements are based on management's current expectations and beliefs and involve significant risks and uncertainties that could cause actual results, developments and business decisions to differ materially from those contemplated by these statements. These risks and uncertainties include but are not limited to the clinical utility of Altamira's product candidates, the timing or likelihood of regulatory filings and approvals, Altamira's intellectual property position and Altamira's financial position. These risks and uncertainties also include, but are not limited to, those described under the caption "Risk Factors" in Altamira's Annual Report on Form 20-F for the year ended December 31, 2024, and in Altamira's other filings with the Securities Exchange Commission ("SEC"), which are available free of charge on the SEC's website at: www.sec.gov. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those indicated. All forward-looking statements and all subsequent written and oral forward-looking statements attributable to Altamira or to persons acting on behalf of Altamira are expressly qualified in their entirety by reference to these risks and uncertainties. You should not place undue reliance on forward-looking statements. Forward-looking statements speak only as of the date they are made, and Altamira does not undertake any obligation to update them in light of new information, future developments or otherwise, except as may be required under applicable law.
Investor Contact:
[1] Zhang Y, Zhang L (2014), Prevalence of Allergic Rhinitis in China, Allergy Asthma Immunol Res. 6(2):105-13, https://e-aair.org/DOIx.php?id=10.4168/aair.2014.6.2.105
SOURCE: Altamira Therapeutics Ltd.
View the original press release on ACCESS Newswire
A.Mahlangu--AMWN
