
-
 Snooker great O'Sullivan makes history with highest-ever break
Snooker great O'Sullivan makes history with highest-ever break
-
Kuwait refinery hit as Iran says missile production 'no concern'

-
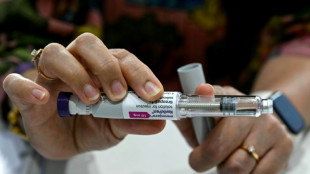 India to tackle global obesity with cheap fat-loss jabs
India to tackle global obesity with cheap fat-loss jabs
-
Somaliland centre saves cheetahs from trafficking to Gulf palaces

-
 China swim sensation Yu, 13, beats multiple Olympic medallist
China swim sensation Yu, 13, beats multiple Olympic medallist
-
North Korean leader, daughter try out new tank

-
 Israel strikes 'decimated' Iran as war roils markets
Israel strikes 'decimated' Iran as war roils markets
-
James ties NBA record for most regular-season games in latest milestone

-
 Trump's Mideast muddle could play into Xi's hands at planned summit
Trump's Mideast muddle could play into Xi's hands at planned summit
-
Wembanyama lifts playoff-bound Spurs, Doncic and James fuel Lakers

-
 Japan ski paradise faces strains of global acclaim
Japan ski paradise faces strains of global acclaim
-
Vinicius, Real Madrid must prove consistency in Atletico derby

-
 Kane credits Kompany's Bayern 'evolution' as treble beckons
Kane credits Kompany's Bayern 'evolution' as treble beckons
-
PSG look back to their best, but not yet out of sight in Ligue 1

-
 Weakened WTO set for high-level meet under cloud of Mideast war
Weakened WTO set for high-level meet under cloud of Mideast war
-
New BTS album to drop ahead of comeback mega-gig

-
 Troubled Spurs face Forest showdown, Chelsea need top-four surge
Troubled Spurs face Forest showdown, Chelsea need top-four surge
-
Australia must be 'smart and adapt' to beat Japan in Asian Cup final: coach

-
 From bats to bonds: Uganda's 'cricket grannies'
From bats to bonds: Uganda's 'cricket grannies'
-
Turkey in cultural diplomacy push to bring history home

-
 'The Bachelorette' canned after star's violent video emerges
'The Bachelorette' canned after star's violent video emerges
-
Trump gets approval for gold coin in his likeness

-
 Behind the BTS comeback, the dark side of K-pop
Behind the BTS comeback, the dark side of K-pop
-
Crude sinks after Netanyahu tries to reassure on Iran war

-
 Three charged with sneaking Nvidia AI chips from US into China
Three charged with sneaking Nvidia AI chips from US into China
-
Swiatek stunned at Miami Open by 50th-ranked Linette

-
 Italy, Germany and France offer help with Hormuz only after ceasefire
Italy, Germany and France offer help with Hormuz only after ceasefire
-
US-backed airstrikes leave Ecuador border communities in fear

-
 'Blackmail': EU leaders round on Orban for stalling Ukraine loan
'Blackmail': EU leaders round on Orban for stalling Ukraine loan
-
Displacement, bombs and air raid sirens weigh on Mideast Eid celebrations

-
 James ties NBA record for most regular-season games played
James ties NBA record for most regular-season games played
-
BTS to drop new album ahead of comeback mega-gig

-
 InterContinental Hotels Group PLC Announces Transaction in Own Shares - March 20
InterContinental Hotels Group PLC Announces Transaction in Own Shares - March 20
-
Netanyahu says Iran 'decimated,' Tehran targets Gulf petro-facilities

-
 Carrick uncertain if Man Utd defender De Ligt will return this season
Carrick uncertain if Man Utd defender De Ligt will return this season
-
US, Israel tactics diverge on Iran as Trump's goals still 'fuzzy'

-
 Japan PM placates Trump on Iran, but faces Pearl Harbor surprise
Japan PM placates Trump on Iran, but faces Pearl Harbor surprise
-
Brazil presidential hopeful Flavio Bolsonaro praises Bukele

-
 The Iran war and the cost of killing 'bad guys'
The Iran war and the cost of killing 'bad guys'
-
US stocks cut losses on Netanyahu war comments as energy prices soar again

-
 Forest beat Midtjylland on penalties to reach Europa League quarters
Forest beat Midtjylland on penalties to reach Europa League quarters
-
Netanyahu says Iran decimated as Tehran warns of 'zero restraint' in energy attacks

-
 Salvadoran anti-corruption lawyer jailed to 'silence her', husband says
Salvadoran anti-corruption lawyer jailed to 'silence her', husband says
-
California to rename Cesar Chavez Day after sex abuse claims

-
 Yazidi woman tells French court of rape, slavery and escape from IS
Yazidi woman tells French court of rape, slavery and escape from IS
-
New FIFA ruling boosts prospects for women coaches

-
 Megan Jones to captain England in Women's Six Nations
Megan Jones to captain England in Women's Six Nations
-
Trump says told Netanyahu not to attack Iran gas fields

-
 MLS reveals shortened 2027 campaign details
MLS reveals shortened 2027 campaign details
-
FIFA planning for World Cup to 'go ahead as scheduled' amid Iran uncertainty


Trump’s Marijuana Schedule III Order Launches a Federally Regulated Healthcare Sector
MMJ's position is further fortified by its FDA Orphan Drug Designations for the treatment of Huntington's Disease. These designations are awarded to drugs intended for the safe and effective treatment of rare diseases and provide MMJ with critical incentives, including: Seven Years of Market Exclusivity: Protecting MMJ's innovations upon FDA approval. Tax Credits: Applied to qualified clinical testing expenses. Waived FDA Fees: Significantly reducing the capital barriers to drug commercialization. Accelerated Review: Streamlining the path to provide relief to patients with unmet medical needs.
WASHINGTON, D.C. / ACCESS Newswire / December 22, 2025 / MMJ International Holdings, MMJ BioPharma Cultivation and MMJ BioPharma Labs ("MMJ"), a pioneer in pharmaceutical-grade cannabinoid drug development, today issued a statement following President Donald Trump's historic executive order directing the reclassification of cannabis from Schedule I to Schedule III under the Controlled Substances Act. The order marks a decisive shift in federal policy-formally recognizing cannabis' credible therapeutic value and validating a science-first, FDA-regulated pharmaceutical pathway that MMJ has pursued for years.

For decades, Schedule I classification functioned as a structural barrier to legitimate clinical research, limiting FDA oversight and leaving millions of patients- including nearly one in four U.S. adults living with chronic pain-without access to standardized, federally regulated treatment options. President Trump's action dismantles that barrier and aligns federal drug policy with established medical and clinical reality.
A Strategic Advantage in a Schedule III Landscape
The transition to Schedule III draws a clear line between recreational commerce and pharmaceutical medicine. MMJ enters this new landscape with a substantial strategic advantage, having already completed much of the foundational work required for pharmaceutical legitimacy, including:
Advanced Formulation - Development of a proprietary, pharmaceutical-grade soft-gel capsule
Scientific Validation - Completion of comprehensive stability testing and chromatography
Clinical Readiness - Advancement of FDA-authorized clinical programs targeting chronic pain and neurological disorders
This infrastructure positions MMJ to move immediately as regulatory friction eases.
Focus Shifts to DOJ Implementation
With federal policy now clarified, attention turns to the U.S. Department of Justice and Attorney General Pamela Bondi, whose actions will determine the speed at which lawful, FDA-authorized cannabinoid research proceeds. MMJ's prior constitutional challenges to the DEA's administrative process-challenges that resulted in DOJ acknowledgments of fundamental defects in the former framework-underscore the importance of prompt, science-driven implementation.
MMJ urges the Department of Justice to ensure that FDA-authorized programs advance without further delay so patients can access safe, consistent, and reproducible medicines under federal oversight.
As institutional capital evaluates the implications of Schedule III, the distinction between speculative cannabis retail and regulated cannabinoid medicine has never been clearer. MMJ's early and sustained investment in pharmaceutical manufacturing, clinical validation, and regulatory compliance offers investors exposure to what is now unmistakably a healthcare sector.
"The federal government has caught up to the strategy MMJ has pursued since its inception," the company added. "The question is no longer whether cannabis has medical value, but how quickly pharmaceutical-grade solutions can reach patients."
About MMJ International Holdings
MMJ International Holdings is a biopharmaceutical company focused on the FDA-authorized development of cannabinoid-based medicines. Through its subsidiaries, the company advances pharmaceutical-grade, reproducible final dosage forms supported by clinical trials and high-standard manufacturing for the treatment of chronic pain and neurological disorders.
MMJ is represented by attorney Megan Sheehan.
CONTACT:
Madison Hisey
[email protected]
203-231-8583
SOURCE: MMJ International Holdings
View the original press release on ACCESS Newswire
F.Pedersen--AMWN