
-
 No.1 Korda cruises to LPGA Mexico crown
No.1 Korda cruises to LPGA Mexico crown
-
Thompson-Herah shines at world relays, Tebogo helps Botswana to win

-
 Three die on Atlantic cruise ship from suspected hantavirus: WHO
Three die on Atlantic cruise ship from suspected hantavirus: WHO
-
Germany's Merz says not 'giving up on working with Donald Trump'

-
 Mercedes' Kimi Antonelli wins Miami Grand Prix
Mercedes' Kimi Antonelli wins Miami Grand Prix
-
Man Utd job feels 'natural' to Carrick

-
 Ferguson taken to hospital before Man Utd win against Liverpool
Ferguson taken to hospital before Man Utd win against Liverpool
-
'Devil Wears Prada 2' takes top spot in N. America box office

-
 Iran weighs US response to peace plan after warning against military action
Iran weighs US response to peace plan after warning against military action
-
Gladbach sink Dortmund, St Pauli edge closer to drop

-
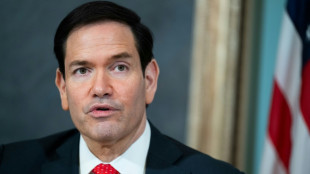 Rubio to visit Rome, meet Pope Leo after Trump row
Rubio to visit Rome, meet Pope Leo after Trump row
-
Kyiv hits Russian oil sites as eight killed in both countries

-
 Iran says US military operation 'impossible' as Trump mulls peace proposal
Iran says US military operation 'impossible' as Trump mulls peace proposal
-
Man Utd beat Liverpool to secure Champions League place

-
 Two die in 'respiratory illness' outbreak on Atlantic cruise ship
Two die in 'respiratory illness' outbreak on Atlantic cruise ship
-
Barcelona sink Bayern to reach women's Champions League final

-
 True Love lands eighth English 1000 Guineas for O'Brien
True Love lands eighth English 1000 Guineas for O'Brien
-
Sinner dismantles Zverev to win Madrid Open, set record

-
 Brilliant Bordeaux clean out Bath to reach Champions Cup final
Brilliant Bordeaux clean out Bath to reach Champions Cup final
-
Second unexploded shell found at illegal French rave: minister

-
 Bournemouth eye European place after crushing Palace
Bournemouth eye European place after crushing Palace
-
Pogacar ends dominant Tour of Romandie with fourth win

-
 Chakravarthy, Narine help Kolkata stay alive in IPL
Chakravarthy, Narine help Kolkata stay alive in IPL
-
Daughter says Maradona died after carers' plan 'went out of control'

-
 Two women suffocate on migrant boat seeking to reach UK
Two women suffocate on migrant boat seeking to reach UK
-
How Schalke returned to the Bundesliga after their 'worst season ever'

-
 Two women die on migrant boat seeking to reach UK
Two women die on migrant boat seeking to reach UK
-
Mumbai coach Jayawardene backs Suryakumar to find his 'rhythm'

-
 Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach
Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach
-
Bangkok food vendor curbs push city staple from the streets

-
 More Nepalis drive electric, evading global fuel shocks
More Nepalis drive electric, evading global fuel shocks
-
Latecomer Japan eyes slice of rising global defence spending

-
 Messi goal not enough as Miami collapse in 4-3 loss to Orlando
Messi goal not enough as Miami collapse in 4-3 loss to Orlando
-
German fertiliser makers and farmers struggle with Iran war fallout

-
 OPEC+ to make first post-UAE production decision
OPEC+ to make first post-UAE production decision
-
Massive crowds fill Rio's Copacabana beach for Shakira concert

-
 Embiid, Maxey shine as 76ers eliminate Celtics in NBA playoffs
Embiid, Maxey shine as 76ers eliminate Celtics in NBA playoffs
-
Fleeting freedom at festival for India's transgender community
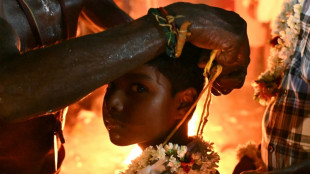
-
 Trump says cutting US troop numbers in Germany 'way down'
Trump says cutting US troop numbers in Germany 'way down'
-
Man charged with murdering Indigenous girl in Australian outback

-
 SCEMFIS-Supported Menhaden Research Advances Work Toward a Scientifically Based Chesapeake Bay Harvest Cap
SCEMFIS-Supported Menhaden Research Advances Work Toward a Scientifically Based Chesapeake Bay Harvest Cap
-
CMS EXPOSED: The "Workaround Doctrine" - How Matt Zorn's Legal CMS Hemp Strategy Undermines the FDA

-
 Prometheus Signals Near-Term Development of Next-Generation Noninvasive Test for MASH Patients at DDW 2026
Prometheus Signals Near-Term Development of Next-Generation Noninvasive Test for MASH Patients at DDW 2026
-
China's Wu Yize wins last-frame thriller to reach snooker world final

-
 Serene Korda takes three-shot lead at LPGA Mexico
Serene Korda takes three-shot lead at LPGA Mexico
-
Golden Tempo wins Kentucky Derby in historic triumph for trainer DeVaux

-
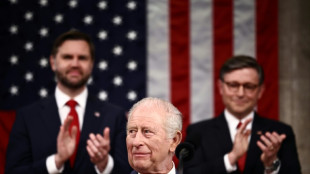 King Charles grasped 'opportunity' on US trip, palace says
King Charles grasped 'opportunity' on US trip, palace says
-
China's Wu wins last-frame thriller to reach snooker world final

-
 Verstappen sees light at the end of tunnel
Verstappen sees light at the end of tunnel
-
Young stretches PGA lead to six at Doral


BioNxt Signs Strategic Agreement for Eurasian Commercialization Following Cladribine ODF Patent Grant in Eurasia and Europe
VANCOUVER, BC / ACCESS Newswire / March 17, 2026 / BioNxt Solutions Inc. ("BioNxt" or the "Company") (CSE:BNXT)(OTCQB:BNXTF)(FSE:BXT), a bioscience innovator specializing in advanced drug delivery systems, is pleased to announce that it has signed a non-binding letter of intent (LOI) establishing an exclusive negotiation framework for the potential commercialization of its sublingual cladribine oral thin film (ODF) product within one or more member states of the Eurasian Patent Organization (EAPO).
The LOI outlines a structured pathway toward a potential regional licensing arrangement and follows recent patent grants and decisions covering BioNxt's proprietary cladribine ODF technology in both the Eurasian region and Europe, significantly strengthening the Company's global intellectual property position.
Structured Commercialization Pathway
The signed LOI represents a significant step toward regional market entry and outlines a defined pathway toward a potential definitive licensing agreement.
The contemplated collaboration framework includes potential regional licensing rights, defined exclusivity during the negotiation period, and financial terms structured around upfront payments, royalties, and revenue sharing. The framework also anticipates collaboration across regulatory strategy, manufacturing coordination, quality control, and commercialization planning within the defined territory.
Under the LOI, the parties have agreed to negotiate exclusively for a period of 60 days while working toward finalizing a definitive commercial agreement. While the LOI is non-binding with respect to the final transaction, it reflects the parties' mutual intent to advance commercialization discussions in the region supported by BioNxt's expanding intellectual property footprint and differentiated drug delivery platform.
"We believe this strategic agreement marks an important milestone in the commercialization of our cladribine ODF program," said Hugh Rogers, CEO and Director of BioNxt Solutions Inc.
"Securing long-term patent protection in both the Eurasian region and Europe strengthens our global IP position and underscores the commercial relevance of our platform. We look forward to engaging with potential partners in Europe and Eurasia to pursue licensing and commercialization opportunities that bring patient-centric therapies to market."
Strengthened Patent Protection in Eurasia and Europe
BioNxt has received formal patent protection for its sublingual cladribine ODF formulation from the Eurasian Patent Organization (EAPO) and the European Patent Office (EPO).
Patent No. 051510, issued on November 14, 2025, by the EAPO, covers sublingual delivery of therapeutic compounds including BioNxt's lead product candidate BNT23001. The patent provides protection in all eight EAPO member states (Armenia, Azerbaijan, Belarus, Kazakhstan, Kyrgyzstan, Russia, Tajikistan, and Turkmenistan), representing a combined population of more than 200 million people, and is valid through at least June 14, 2043.
In addition, effective March 11, 2026, by way of publication in the European Patent Bulletin, the EPO has granted European Patent No. 4539857 covering BioNxt's proprietary sublingual cladribine ODF technology. Upon national validation, the patent is expected to provide protection in up to 39 European Patent Convention (EPC) Contracting States, through to at least June 14, 2043, subject to national procedures and maintenance requirements.
These IP achievements significantly advance BioNxt's global intellectual property strategy and support its ongoing pursuit of strategic licensing and partnership opportunities across Europe, Eurasia, and other international markets.
Access to a More Than 200 Million Population Market and Broader European Opportunity
The Eurasian region's eight member states represent a combined population of over 200 million people, offering meaningful demographic reach for potential commercialization of innovative therapies.
In Europe, once nationally validated, the European patent is expected to provide protection across a broad set of jurisdictions covering many of the world's largest pharmaceutical markets. Combined with the Eurasian grant, this positions BioNxt to pursue licensing partnerships spanning major patient populations across Eurasia and Europe.
The Company is actively evaluating potential licensing partners and commercialization collaborations in European markets, where neurological and autoimmune diseases such as Multiple Sclerosis (MS) represent a substantial therapeutic and commercial opportunity.
Multiple Sclerosis affects approximately 2.9 million people globally, with significant patient populations across Europe, Eastern Europe, and Central Asia. Patient populations with other autoimmune neuromuscular disorders, such as Myasthenia Gravis (MG), further expand the addressable market where patient-friendly, swallow-free delivery options may offer meaningful clinical and commercial differentiation.
Cladribine is an approved therapy for relapsing forms of MS in major markets, with jurisdiction-specific indications. BioNxt's sublingual ODF formulation is designed as a needle-free and swallow-free alternative to conventional tablet administration, with the goal of improving patient convenience, adherence, and real-world usability.
Growing Global Demand for Patient-Centric Drug Delivery
The global needle-free drug delivery market was valued at approximately USD 14.39 billion in 2024 and is projected to reach approximately USD 30.73 billion by 2032.
The global oral thin films market is projected to grow at an approximate 8.6% compound annual growth rate through 2032, reflecting increasing demand for patient-centric dosage forms that enhance compliance, convenience, and real-world usability.
By combining an approved active ingredient with a differentiated, IP-protected delivery system, BioNxt aims to reduce development risk while enabling an accelerated, partnership-driven commercialization strategy supported by long-term territorial exclusivity.
Accelerated Development Pathway
Because cladribine is already an approved active pharmaceutical ingredient, BioNxt's development pathway is primarily focused on demonstrating bioequivalence of its sublingual formulation relative to existing tablet products.
Preparations for a human bioequivalence study are underway following successful preclinical pharmacokinetic studies in animal models, which evaluated systemic exposure following administration of the Company's ODF formulation.
The signed LOI, together with secured Eurasian and European patent protection valid through 2043, positions BioNxt to advance regional commercialization discussions supported by defined territorial exclusivity and long-term intellectual property rights.
About BioNxt Solutions Inc.
BioNxt Solutions Inc. is a bioscience innovator focused on next-generation drug delivery platforms, diagnostic screening systems, and active pharmaceutical ingredient development. Its proprietary platforms include sublingual thin films, transdermal patches, oral tablets, and a new targeted chemotherapy platform designed to deliver cancer drugs directly to tumors while reducing side effects.
With research and development operations in North America and Europe, BioNxt is advancing regulatory approvals and commercialization efforts, primarily focused on European markets. BioNxt is committed to improving healthcare by delivering precise, patient-centric solutions that enhance treatment outcomes worldwide.
BioNxt is listed on the Canadian Securities Exchange: BNXT, OTC Markets: BNXTF and trades in Germany under WKN: A3D1K3. To learn more about BioNxt, please visit www.bionxt.com.
Investor Relations & Media Contact
Hugh Rogers, Co-Founder, CEO and Director
Email: [email protected]
Phone: +1 780-818-6422
Web: www.bionxt.com
LinkedIn: https://www.linkedin.com/company/bionxt-solutions
Instagram: https://www.instagram.com/bionxt
Cautionary Statement Regarding "Forward-Looking" Information
This news release contains forward-looking statements within the meaning of applicable securities laws. Forward-looking statements are based on management's current expectations, estimates, projections, beliefs, and assumptions as of the date of this release.
Forward-looking statements in this release include, but are not limited to, statements regarding: the negotiation and potential execution of a definitive licensing agreement pursuant to the non-binding letter of intent; the scope, territorial coverage, validity, term, and enforceability of the Company's Eurasian patent; the Company's intellectual property strategy; the continued development of its sublingual cladribine oral thin film (ODF) program; the anticipated timing, design, and conduct of planned human bioequivalence studies; potential regulatory pathways; expansion into additional indications; and potential commercialization, licensing, or partnering activities.
Forward-looking statements are subject to known and unknown risks, uncertainties, and other factors that may cause actual results, performance, or achievements to differ materially from those expressed or implied by such forward-looking statements. These risks and uncertainties include, but are not limited to: the possibility that a definitive agreement may not be executed within the exclusivity period or at all; risks associated with intellectual property protection and enforcement; potential patent opposition or validity challenges; uncertainties relating to regulatory review and approval; scientific, formulation, and development risks; the possibility that preclinical or pharmacokinetic results may not be predictive of human clinical outcomes; risks related to the timing, cost, and results of clinical studies; manufacturing and scale-up risks; reliance on third-party service providers; competitive developments; geopolitical risks in the Eurasian region; and general economic, market, and capital market conditions.
Readers are cautioned not to place undue reliance on forward-looking statements. Except as required by applicable securities laws, BioNxt undertakes no obligation to update or revise any forward-looking statements to reflect new information, future events, or otherwise.
SOURCE: BioNxt Solutions Inc.
View the original press release on ACCESS Newswire
O.Johnson--AMWN