
-
 Daughter says Maradona died after carers' plan 'went out of control'
Daughter says Maradona died after carers' plan 'went out of control'
-
Two women suffocate on migrant boat seeking to reach UK

-
 How Schalke returned to the Bundesliga after their 'worst season ever'
How Schalke returned to the Bundesliga after their 'worst season ever'
-
Two women die on migrant boat seeking to reach UK

-
 Mumbai coach Jayawardene backs Suryakumar to find his 'rhythm'
Mumbai coach Jayawardene backs Suryakumar to find his 'rhythm'
-
Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach

-
 Bangkok food vendor curbs push city staple from the streets
Bangkok food vendor curbs push city staple from the streets
-
More Nepalis drive electric, evading global fuel shocks

-
 Latecomer Japan eyes slice of rising global defence spending
Latecomer Japan eyes slice of rising global defence spending
-
Messi goal not enough as Miami collapse in 4-3 loss to Orlando

-
 German fertiliser makers and farmers struggle with Iran war fallout
German fertiliser makers and farmers struggle with Iran war fallout
-
OPEC+ to make first post-UAE production decision

-
 Massive crowds fill Rio's Copacabana beach for Shakira concert
Massive crowds fill Rio's Copacabana beach for Shakira concert
-
Embiid, Maxey shine as 76ers eliminate Celtics in NBA playoffs

-
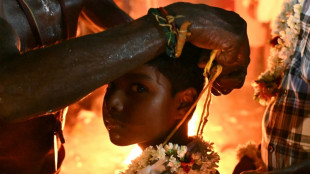 Fleeting freedom at festival for India's transgender community
Fleeting freedom at festival for India's transgender community
-
Trump says cutting US troop numbers in Germany 'way down'

-
 Man charged with murdering Indigenous girl in Australian outback
Man charged with murdering Indigenous girl in Australian outback
-
Prometheus Signals Near-Term Development of Next-Generation Noninvasive Test for MASH Patients at DDW 2026

-
 China's Wu Yize wins last-frame thriller to reach snooker world final
China's Wu Yize wins last-frame thriller to reach snooker world final
-
Serene Korda takes three-shot lead at LPGA Mexico

-
 Golden Tempo wins Kentucky Derby in historic triumph for trainer DeVaux
Golden Tempo wins Kentucky Derby in historic triumph for trainer DeVaux
-
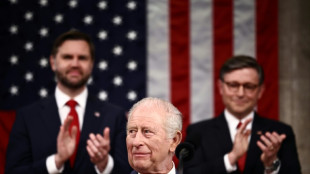
King Charles grasped 'opportunity' on US trip, palace says
-
 China's Wu wins last-frame thriller to reach snooker world final
China's Wu wins last-frame thriller to reach snooker world final
-
Verstappen sees light at the end of tunnel

-
 Young stretches PGA lead to six at Doral
Young stretches PGA lead to six at Doral
-
Rio's Copacabana beach hosts massive crowd for free Shakira concert

-
 Celtics' Tatum ruled out for decisive game seven against Sixers
Celtics' Tatum ruled out for decisive game seven against Sixers
-
Wolff heralds Antonelli speed as teen joins Senna and Schumacher in record books

-
 Senior Iranian officer says fresh conflict with US 'likely'
Senior Iranian officer says fresh conflict with US 'likely'
-
Barcelona on verge of Liga title, Villarreal secure top four

-
 Teen F1 leader Antonelli takes Miami Grand Prix pole
Teen F1 leader Antonelli takes Miami Grand Prix pole
-
Porto edge Alverca to clinch Portuguese league title

-
 US airlines step up as Spirit winds down
US airlines step up as Spirit winds down
-
Barcelona on verge of La Liga title defence with win at Osasuna

-
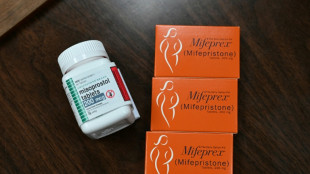 Drugmaker asks US Supreme Court to restore abortion pill access
Drugmaker asks US Supreme Court to restore abortion pill access
-
Schalke return to Bundesliga after three-year absence

-
 NATO, top Republicans question US troop withdrawal from Germany
NATO, top Republicans question US troop withdrawal from Germany
-
Napoli frustrate Como in costly Serie A stalemate

-
 Illegal party at French military site draws up to 40,000 ravers
Illegal party at French military site draws up to 40,000 ravers
-
Arsenal hit stride to go six points clear, West Ham loss offers Spurs hope

-
 Arsenal go six points clear as Gyokeres double sinks Fulham
Arsenal go six points clear as Gyokeres double sinks Fulham
-
Clinical Chennai down Mumbai to keep playoff hopes alive

-
 Napoli and Como play out goalless draw in Serie A
Napoli and Como play out goalless draw in Serie A
-
Murphy into World Snooker Championship final after edging Higgins

-
 PSG held by Lorient with fringe team ahead of Bayern Munich return leg
PSG held by Lorient with fringe team ahead of Bayern Munich return leg
-
Aviation companies step up as Spirit winds down

-
 Champion Norris leads Piastri home in sprint 1-2 triumph for McLaren
Champion Norris leads Piastri home in sprint 1-2 triumph for McLaren
-
UK PM says some pro-Palestinian marches could be banned

-
 The Puma out of Kentucky Derby, leaving 19 starters
The Puma out of Kentucky Derby, leaving 19 starters
-
'Bookless bookstore': audio-only book shop opens in New York


Prometheus Signals Near-Term Development of Next-Generation Noninvasive Test for MASH Patients at DDW 2026
Translational Data Inform a Scalable, Noninvasive Test Designed to Identify Treatment-Eligible Patients and Enable Longitudinal Assessment as MASH Therapies Expand
SAN DIEGO, CA / ACCESS Newswire / May 3, 2026 / Prometheus Laboratories Inc. ("Prometheus"), a leader in precision healthcare, today announced new translational research presented at Digestive Disease Week® (DDW®) 2026 supporting the development of a next‑generation, blood‑based approach designed to address a critical gap in noninvasive testing (NIT) for metabolic dysfunction-associated steatohepatitis (MASH).
Prometheus has been at the forefront of noninvasive liver fibrosis assessment for more than two decades. Its FibroSpect® NASH test was developed at a time when noninvasive alternatives to liver biopsy were limited and disease‑modifying therapies were not yet available. FibroSpect NASH addresses a pressing clinical need by helping identify patients at risk for advanced fibrosis and cirrhosis using an objective, noninvasive and longitudinally trackable score, supporting both risk stratification and ongoing disease assessment.
As the MASH therapeutic landscape has evolved, with treatments now directed toward patients with moderate fibrosis (F2-F3) and additional therapies under development, the diagnostic needs of health systems and clinicians continue to shift. Often asymptomatic, MASH affects up to 20 million people in the United States, with prevalence rising alongside obesity and type 2 diabetes, increasing the importance of timely, scalable and cost‑effective identification of treatment‑eligible patients and ongoing assessment of those receiving therapy with blood‑based testing.
At DDW® 2026, Prometheus is presenting findings from an exploratory, translational research study evaluating how its established FibroSpect biomarker architecture performs when applied to fibrosis staging ranges aligned with today's treatment‑eligible MASH population. The analysis illustrates how interval‑based scoring may help distinguish patients more likely to have moderate fibrosis (F2-F3) while helping rule out cirrhosis, providing clinically relevant insight.
"This work reflects how fibrosis assessment must evolve alongside the therapeutic landscape," said Rohit Loomba, MD, MHSc, lead author of the study, and Chief of the Division of Gastroenterology and Hepatology and Director of the MASLD Research Center at the University of California San Diego. "Interval‑based interpretation can provide more nuanced information than a single cutoff and better aligns noninvasive assessment with clinical decision‑making as therapies increasingly target patients with moderate fibrosis."
These initial findings demonstrate how translational research can inform diagnostic innovation for the MASH treatment era. Building on this work, Prometheus is developing a next‑generation noninvasive blood test planned for launch later this year to support identification, management and risk stratification of treatment‑eligible MASH patients.
Poster Su2029, "Non-Invasive Testing to Rule-In Moderate to Severe Fibrosis While Ruling Out Cirrhosis for Therapeutic Intervention in Metabolic Dysfunction Associated Steatohepatitis," will be presented at DDW® 2026 on Sunday, May 3, at 12:30 p.m. CDT.
Dr. Loomba is a paid consultant and scientific advisor for Prometheus Laboratories.
About Prometheus Laboratories
Prometheus Laboratories has been a leader in diagnostic testing for gastroenterology and hepatology for more than 30 years. The company's portfolio of non‑invasive diagnostic tests supports clinicians in the diagnosis, assessment and management of complex gastrointestinal and liver diseases, including inflammatory bowel disease and liver fibrosis. Prometheus is committed to advancing precision‑guided diagnostics that help inform clinical decision‑making and longitudinal assessment across evolving standards of care. For more information, visit the Prometheus website and follow us on LinkedIn or X (formerly Twitter).
About DDW
Digestive Disease Week® (DDW) is the largest international gathering of physicians, researchers and academics in the fields of gastroenterology, hepatology, endoscopy and gastrointestinal surgery. Jointly sponsored by the American Association for the Study of Liver Diseases (AASLD), the American Gastroenterological Association (AGA), the American Society for Gastrointestinal Endoscopy (ASGE) and the Society for Surgery of the Alimentary Tract (SSAT), DDW is an in-person and online meeting from May 2-5, 2026. The meeting showcases nearly 6,000 abstracts and over 1,000 invited talks on the latest advances in GI research, medicine and technology. More information can be found at www.ddw.org.
Media Contact
Chrystal Johnson
Sr. Marketing & Events Specialist
Prometheus Laboratories Inc.
[email protected]
SOURCE: Prometheus Laboratories Inc.
View the original press release on ACCESS Newswire
L.Harper--AMWN