
-
 'Devil Wears Prada 2' takes top spot in N. America box office
'Devil Wears Prada 2' takes top spot in N. America box office
-
Iran weighs US response to peace plan after warning against military action

-
 Gladbach sink Dortmund, St Pauli edge closer to drop
Gladbach sink Dortmund, St Pauli edge closer to drop
-
Rubio to visit Rome, meet Pope Leo after Trump row
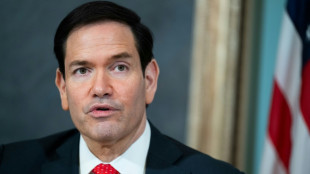
-
 Kyiv hits Russian oil sites as eight killed in both countries
Kyiv hits Russian oil sites as eight killed in both countries
-
Iran says US military operation 'impossible' as Trump mulls peace proposal

-
 Man Utd beat Liverpool to secure Champions League place
Man Utd beat Liverpool to secure Champions League place
-
Two die in 'respiratory illness' outbreak on Atlantic cruise ship

-
 Barcelona sink Bayern to reach women's Champions League final
Barcelona sink Bayern to reach women's Champions League final
-
True Love lands eighth English 1000 Guineas for O'Brien

-
 Sinner dismantles Zverev to win Madrid Open, set record
Sinner dismantles Zverev to win Madrid Open, set record
-
Brilliant Bordeaux clean out Bath to reach Champions Cup final

-
 Second unexploded shell found at illegal French rave: minister
Second unexploded shell found at illegal French rave: minister
-
Bournemouth eye European place after crushing Palace

-
 Pogacar ends dominant Tour of Romandie with fourth win
Pogacar ends dominant Tour of Romandie with fourth win
-
Chakravarthy, Narine help Kolkata stay alive in IPL

-
 Daughter says Maradona died after carers' plan 'went out of control'
Daughter says Maradona died after carers' plan 'went out of control'
-
Two women suffocate on migrant boat seeking to reach UK

-
 How Schalke returned to the Bundesliga after their 'worst season ever'
How Schalke returned to the Bundesliga after their 'worst season ever'
-
Two women die on migrant boat seeking to reach UK

-
 Mumbai coach Jayawardene backs Suryakumar to find his 'rhythm'
Mumbai coach Jayawardene backs Suryakumar to find his 'rhythm'
-
Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach

-
 Bangkok food vendor curbs push city staple from the streets
Bangkok food vendor curbs push city staple from the streets
-
More Nepalis drive electric, evading global fuel shocks

-
 Latecomer Japan eyes slice of rising global defence spending
Latecomer Japan eyes slice of rising global defence spending
-
Messi goal not enough as Miami collapse in 4-3 loss to Orlando

-
 German fertiliser makers and farmers struggle with Iran war fallout
German fertiliser makers and farmers struggle with Iran war fallout
-
OPEC+ to make first post-UAE production decision

-
 Massive crowds fill Rio's Copacabana beach for Shakira concert
Massive crowds fill Rio's Copacabana beach for Shakira concert
-
Embiid, Maxey shine as 76ers eliminate Celtics in NBA playoffs

-
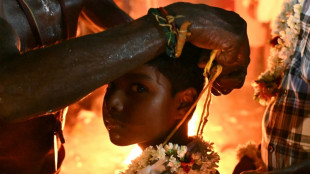 Fleeting freedom at festival for India's transgender community
Fleeting freedom at festival for India's transgender community
-
Trump says cutting US troop numbers in Germany 'way down'

-
 Man charged with murdering Indigenous girl in Australian outback
Man charged with murdering Indigenous girl in Australian outback
-
CMS EXPOSED: The "Workaround Doctrine" - How Matt Zorn's Legal CMS Hemp Strategy Undermines the FDA

-
 Prometheus Signals Near-Term Development of Next-Generation Noninvasive Test for MASH Patients at DDW 2026
Prometheus Signals Near-Term Development of Next-Generation Noninvasive Test for MASH Patients at DDW 2026
-
China's Wu Yize wins last-frame thriller to reach snooker world final

-
 Serene Korda takes three-shot lead at LPGA Mexico
Serene Korda takes three-shot lead at LPGA Mexico
-
Golden Tempo wins Kentucky Derby in historic triumph for trainer DeVaux

-
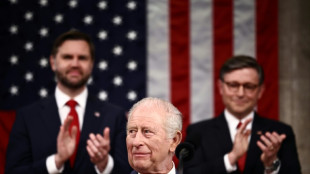 King Charles grasped 'opportunity' on US trip, palace says
King Charles grasped 'opportunity' on US trip, palace says
-
China's Wu wins last-frame thriller to reach snooker world final

-
 Verstappen sees light at the end of tunnel
Verstappen sees light at the end of tunnel
-
Young stretches PGA lead to six at Doral

-
 Rio's Copacabana beach hosts massive crowd for free Shakira concert
Rio's Copacabana beach hosts massive crowd for free Shakira concert
-
Celtics' Tatum ruled out for decisive game seven against Sixers

-
 Wolff heralds Antonelli speed as teen joins Senna and Schumacher in record books
Wolff heralds Antonelli speed as teen joins Senna and Schumacher in record books
-
Senior Iranian officer says fresh conflict with US 'likely'

-
 Barcelona on verge of Liga title, Villarreal secure top four
Barcelona on verge of Liga title, Villarreal secure top four
-
Teen F1 leader Antonelli takes Miami Grand Prix pole

-
 Porto edge Alverca to clinch Portuguese league title
Porto edge Alverca to clinch Portuguese league title
-
US airlines step up as Spirit winds down


FDA Feedback Supports Extension Phase for Jaguar Health's Clinical Trial of Crofelemer for Treatment of Microvillus Inclusion Disease (MVID)
MVID has a lethal natural history requiring life-sustaining parenteral support (PS), which includes total parenteral nutrition (TPN), which is associated with significant toxicities; PS reduction could potentially reduce co-morbidities and improve clinical benefit, as no approved therapies exist for MVID
Jaguar launches news alert text service for investors - click here to sign up for text alerts
SAN FRANCISCO, CA / ACCESS Newswire / March 18, 2026 / Jaguar Health, Inc.(NASDAQ:JAGX) (Jaguar) family company Napo Pharmaceuticals (Napo) today announced that, based on the U.S. Food and Drug Administration (FDA) support for Napo's protocol amendment for its clinical trial to evaluate the safety and efficacy of its novel crofelemer powder-for-oral-solution formulation in pediatric patients with intestinal failure due to MVID, an ultrarare pediatric disorder, Napo plans to initiate a single-blind extension phase for the trial. The extended treatment period has the potential to improve clinical benefit of crofelemer through reduction of parenteral support (PS) requirements, which may result in disease progression-modification of MVID.
"We are pleased to have the opportunity to continue treatment with crofelemer powder for oral solution in our ongoing clinical trial of crofelemer in pediatric MVID patients," said Pravin Chaturvedi, PhD, Jaguar's Chief Scientific Officer and Chair of the Jaguar and Napo Scientific Advisory Board. "This would allow patients who have completed their double-blind treatment period to be eligible for the single-blind phase of continued treatment with crofelemer for further assessment of clinical benefit through reduction of PS for a longer period. PS reduction could potentially reduce co-morbidities associated with the disease and TPN; thus improving clinical benefit and modifying the disease progression to help address the lethal natural history of MVID."
The dose selection for the single-blind extension phase will be determined by the study's independent Data Monitoring Committee (DMC), which will ensure that the study clinicians and Napo personnel will remain blinded. Currently, approximately 30% of the participating MVID patients have completed the study's double-blind treatment phase and the clinical investigators are supporting the patients' participation in the single-blind extended phase.
This trial of crofelemer in MVID patients has clinical sites in the US, Italy and the UAE, and the consent from the FDA, the European Medicines Agency (EMA), The Ministry of Health and Prevention (MOHAP), and the Institutional Review Boards (IRBs) for each site.
"Our development plan for this ultrarare disease, with an expected improved clinical benefit from the longer treatment in the extension phase, may also support the possibility for a Breakthrough Therapy designation from the FDA in the coming months, potentially supporting opportunities for expedited regulatory submission pathways to the FDA for new drug approval for this ultrarare pediatric disorder," said Lisa Conte, Jaguar's founder, president, and CEO. "The extremely low incidence and prevalence of MVID, its lethal natural history and the fact that no therapies are available for MVID other than lifelong PS, underscores the need for new therapies. Together with the support of MVID clinical key opinion leaders (KOLs), we are exploring expedited regulatory pathways for crofelemer."
About the Jaguar Health Family of Companies
Jaguar Health, Inc. (Jaguar) is a commercial stage pharmaceuticals company focused on developing novel proprietary prescription medicines sustainably derived from plants from rainforest areas for people and animals with gastrointestinal distress. Jaguar family companies Napo Pharmaceuticals (Napo) and Napo Therapeutics S.p.A. focus on the development and commercialization of novel crofelemer powder for oral solution for the treatment of rare and orphan gastrointestinal disorders with intestinal failure, including MVID and short bowel syndrome.
For more information about:
Jaguar Health, visit https://jaguar.health
Napo Pharmaceuticals, visit www.napopharma.com
Napo Therapeutics, visit napotherapeutics.com
Forward-Looking Statements
Certain statements in this press release constitute "forward-looking statements." These include statements regarding Jaguar's expectation that Napo will initiate a single-blind extension phase for its MVID trial, the expectation that PS reduction could potentially reduce co-morbidities associated with MVID and TPN, thus improving clinical benefit and modifying the disease progression to help address the lethal natural history of MVID, and Jaguar's expectation that its development plan for MVID may support the possibility for a Breakthrough Therapy designation from the FDA in the coming months, potentially supporting opportunities for expedited regulatory submission pathways to the FDA for new drug approval. In some cases, you can identify forward-looking statements by terms such as "may," "will," "should," "expect," "plan," "aim," "anticipate," "could," "intend," "target," "project," "contemplate," "believe," "estimate," "predict," "potential" or "continue" or the negative of these terms or other similar expressions. The forward-looking statements in this release are only predictions. Jaguar has based these forward-looking statements largely on its current expectations and projections about future events. These forward-looking statements speak only as of the date of this release and are subject to several risks, uncertainties, and assumptions, some of which cannot be predicted or quantified and some of which are beyond Jaguar's control. Except as required by applicable law, Jaguar does not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
Contact:
[email protected]
Jaguar-JAGX
SOURCE: Jaguar Health, Inc.
View the original press release on ACCESS Newswire
A.Rodriguezv--AMWN