
-
 Mali faces advancing rebels in 'difficult' situation
Mali faces advancing rebels in 'difficult' situation
-
Monk ends barefoot Sri Lanka trek with a dog and plea for peace

-
 Macron urges Andorra to 'move forwards' on decriminalising abortion
Macron urges Andorra to 'move forwards' on decriminalising abortion
-
German bid to rescue 'Timmy' the whale passes key hurdle

-
 US Fed expected to keep rates steady as Iran war effects ripple
US Fed expected to keep rates steady as Iran war effects ripple
-
UAE pulls out of OPEC oil cartels citing 'national interests'

-
 Crude back above $110 on Strait stalemate fears
Crude back above $110 on Strait stalemate fears
-
Banking giant JP Morgan becomes Olympics sponsor

-
 Emotional Stones announces Man City exit after golden decade
Emotional Stones announces Man City exit after golden decade
-
Jazz legend John Coltrane's son hits the high notes

-
 John Stones to leave Manchester City after 10 years
John Stones to leave Manchester City after 10 years
-
Croatia, Bosnia sign major gas pipeline deal

-
 Champions League semi-final like a first date: Atletico's Koke
Champions League semi-final like a first date: Atletico's Koke
-
Sinner queries schedule, surges into Madrid Open quarters

-
 ICC orders $8.5mn compensation for victims of Malian war criminal
ICC orders $8.5mn compensation for victims of Malian war criminal
-
EU parliament adopts new rules to protect cats, dogs

-
 EU lawmakers back blockbuster long-term budget
EU lawmakers back blockbuster long-term budget
-
German rescuers launch new bid to free stranded whale

-
 Man pleads guilty in Austria to plotting attack on Taylor Swift concert
Man pleads guilty in Austria to plotting attack on Taylor Swift concert
-
Climbers open Everest route past dangerous ice block

-
 Indian billionaire's son offers home for Escobar's hippos
Indian billionaire's son offers home for Escobar's hippos
-
Iranian Vafaei capable of great things, says beaten rival Trump

-
 Comedian Kimmel hits back at criticism over Melania Trump joke
Comedian Kimmel hits back at criticism over Melania Trump joke
-
Man goes on trial in Austria over Taylor Swift concert attack plan

-
 South Korean court increases ex-first lady's graft sentence
South Korean court increases ex-first lady's graft sentence
-
Bullying claims 'nonsense', actress Rebel Wilson tells Sydney court

-
 BP reports huge profit rise in first quarter
BP reports huge profit rise in first quarter
-
Crude extends gains, stocks drop as Trump considers latest Iran proposal

-
 How China block of AI deal could stop 'Singapore-washing'
How China block of AI deal could stop 'Singapore-washing'
-
North Korean executions rose dramatically during Covid: report

-
 Budget airlines first to cut flights as jet fuel prices soar
Budget airlines first to cut flights as jet fuel prices soar
-
Simeone, Atletico chasing redemption against Arsenal

-
 'Bring it on', says Rice as Arsenal chase Champions League history
'Bring it on', says Rice as Arsenal chase Champions League history
-
US says examining latest Iran proposal

-
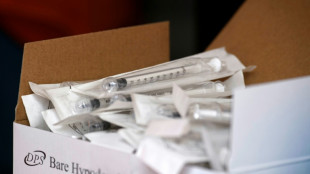 S. Korea probes syringe hoarding as war hits plastic makers
S. Korea probes syringe hoarding as war hits plastic makers
-
Australia aims to tax tech giants unless they pay news outlets

-
 Bangladesh's tigers stalk uncertain future in Sundarbans
Bangladesh's tigers stalk uncertain future in Sundarbans
-
Horses unlikely saviours for those who serve in uniform

-
 Crude extends gains as Trump considers latest Iran proposal
Crude extends gains as Trump considers latest Iran proposal
-
Nations to kick off world-first fossil fuel exit talks

-
 Philippine museum brings deadly, lucrative galleon trade to life
Philippine museum brings deadly, lucrative galleon trade to life
-
Opening remarks Tuesday in Elon Musk versus OpenAI

-
 New York restaurant's $40 half chicken fuels cost of dining debate
New York restaurant's $40 half chicken fuels cost of dining debate
-
Trump shooting scare renews 'staged' conspiracy theory
-
 LIV Golf postpones June event set for New Orleans: reports
LIV Golf postpones June event set for New Orleans: reports
-
As Washington Moves to Reopen Venezuela, Interstice Digital Launches Compliance Infrastructure to Facilitate U.S.-Venezuela Commerce

-
 Zafran Launches Zafran AIR, a Rapid-Response Offering to Help Enterprises Build AI Resilience for the Post-Mythos Eras
Zafran Launches Zafran AIR, a Rapid-Response Offering to Help Enterprises Build AI Resilience for the Post-Mythos Eras
-
BCI Honors Donna Snyder with its 2026 Distinguished Service Award

-
 ONEMETA Integration into Holoscan for Media Announced at NVIDIA GTC 2026 to Support Real-Time Multilingual AI in Live Media Workflows
ONEMETA Integration into Holoscan for Media Announced at NVIDIA GTC 2026 to Support Real-Time Multilingual AI in Live Media Workflows
-
Opti Digital Launches Sixth Edition of Green Challenge to Support Forest Preservation


Protagonist Exercises Rusfertide U.S. Opt-Out Right Under Takeda Collaboration
Opt-out election makes Protagonist eligible for $200 million upon exercise and an additional $200 million upon FDA approval of rusfertide for polycythemia vera
Approval would also trigger a $75 million milestone payment, bringing total potential cash tied to the opt-out election and approval to $475 million
Protagonist becomes eligible for up to $975 million in milestone payments and tiered royalties ranging from 14% to 29% on worldwide net sales
NEWARK, CA / ACCESS Newswire / April 28, 2026 / Protagonist Therapeutics, Inc. (Nasdaq:PTGX) ("Protagonist" or the "Company") today announced that it has exercised its right to opt out of the 50:50 U.S. profit and loss sharing arrangement under its worldwide license and collaboration agreement with Takeda for rusfertide, an investigational first-in-class hepcidin mimetic peptide under U.S. Food and Drug Administration (FDA) Priority Review for the treatment of adults with polycythemia vera (PV).
By exercising its opt-out right during the contractual opt-out period, Protagonist becomes eligible to receive up to $400 million in opt-out payments, consisting of $200 million payable upon the opt-out election and a further $200 million payable upon FDA approval of rusfertide for the treatment of adults with PV. In addition, U.S. approval of rusfertide would trigger a $75 million milestone payment, bringing total potential cash payable in connection with the opt-out election and U.S. approval to $475 million.
As a result of the opt-out election, Takeda now holds exclusive development and commercialization rights to rusfertide in the United States, in addition to its existing exclusive ex-U.S. rights. Under the opt-out structure, Protagonist becomes eligible to receive up to $975 million in milestone payments and tiered royalties on worldwide net sales ranging from 14% to 29%, corresponding to a weighted-average royalty rate of approximately 21% at $1.5 billion in annual net sales, with the top royalty tier of 29% applying to annual net sales above $1.5 billion.
"Exercising our opt-out right reflects our conviction in rusfertide and secures what we believe is the most attractive risk-adjusted value outcome for Protagonist and its shareholders as the hepcidin mimetic peptide approaches a potential approval," said Dinesh V. Patel, Ph.D., President and Chief Executive Officer of Protagonist Therapeutics. "This election provides meaningful near-term non-dilutive cash, materially enhances our long-term economic participation through milestones and worldwide royalties, and further strengthens our ability to invest in our broader pipeline and return value to shareholders. We value our partnership with Takeda and believe they are exceptionally well-positioned to maximize the global opportunity for rusfertide."
Rusfertide is under FDA Priority Review for the treatment of adults with PV, with a Prescription Drug User Fee Act (PDUFA) target action date in the third quarter of 2026. Under the parties' January 2024 collaboration agreement, Protagonist received a $300 million upfront payment and retained the right to elect either a 50:50 U.S. profit and loss share or an opt-out structure featuring enhanced payments, milestones, and worldwide royalties.
About Rusfertide
Rusfertide is a first-in-class investigational subcutaneous treatment that mimics the action of hepcidin, a natural hormone that regulates iron homeostasis and red blood cell production. By targeting the underlying mechanism of iron dysregulation in polycythemia vera, rusfertide aims to reduce excess red blood cell production and help patients achieve sustained hematocrit control. The molecule demonstrated rapid, stable, and durable control of hematocrit in Phase 2 & 3 studies as part of the clinical development program for PV. Rusfertide was discovered and developed using Protagonist Therapeutics' proprietary peptide technology platform. Rusfertide was previously being co-developed in PV. As a result of the opt-out election, Takeda now holds exclusive development and commercialization rights to rusfertide in the United States, in addition to its existing exclusive ex-U.S. rights. The program has received Breakthrough Therapy Designation from the U.S. Food and Drug Administration, underscoring its potential to address significant unmet needs in a chronic hematologic disease with limited modern therapeutic innovation.
About Polycythemia Vera (PV)
Polycythemia vera (PV) is characterized by the overproduction of red blood cells (erythrocytosis), which increases blood viscosity, or thickness, that can result in life-threatening thrombotic events such as stroke, deep vein thrombosis, and pulmonary embolism. Hematocrit (HCT) is the ratio of red blood cells to the total amount of blood in the body. Achieving and maintaining controlled HCT levels of
About Protagonist
Protagonist Therapeutics is a discovery through late-stage development biopharmaceutical company with a proprietary technology platform that enables de novo discovery of peptide therapeutics. Two novel peptides derived from Protagonist's proprietary discovery platform are at or near commercialization. ICOTYDE™ (icotrokinra), licensed to Johnson & Johnson company Janssen Biotech, Inc., is the first and only targeted oral peptide that precisely blocks the Interleukin-23 receptor. ICOTYDE was launched in the U.S. in March 2026 and is approved for the treatment of moderate-to-severe plaque psoriasis in adults and pediatric patients 12 years of age or older and is in Phase 3 development for psoriatic arthritis, ulcerative colitis, and Crohn's disease. ICOTYDE was jointly discovered by Protagonist and Johnson & Johnson scientists, with Protagonist having primary responsibility for the development of ICOTYDE through Phase 1, and Johnson & Johnson assuming responsibility for further development and commercialization. Protagonist also discovered and led development through Phase 3 of rusfertide, a first-in-class hepcidin mimetic peptide licensed to Takeda Pharmaceuticals. An NDA for rusfertide for the treatment of polycythemia vera is under priority review with the FDA. The Company also has a number of clinical and preclinical programs addressing clinically and commercially validated targets, including an oral IL-17 antagonist peptide, obesity dual and triple agonists, an oral hepcidin functional mimetic, and the recently announced IL-4 and amylin programs.
More information on Protagonist, its pipeline drug candidates, and clinical studies can be found on the Company's website at https://www.protagonist-inc.com.
Cautionary Note on Forward-Looking Statements
This press release contains forward-looking statements for purposes of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Forward-looking statements include statements regarding potential payments from its collaboration with Takeda, timing of regulatory actions and capital allocation plans. In some cases, you can identify these statements by forward-looking words such as "anticipate," "believe," "may," "will," "expect," or the negative or plural of these words or similar expressions. Forward-looking statements are not guarantees of future performance and are subject to risks and uncertainties that could cause actual results and events to differ materially from those anticipated, including, but not limited to, our ability to develop and commercialize our product candidates, our ability to earn milestone payments under our collaboration agreements with Janssen and Takeda, our ability to use and expand our programs to build a pipeline of product candidates, our ability to obtain and maintain regulatory approval of our product candidates, our ability to operate in a competitive industry and compete successfully against competitors that have greater resources than we do, and our ability to obtain and adequately protect intellectual property rights for our product candidates. Additional information concerning these and other risk factors affecting our business can be found in our periodic filings with the Securities and Exchange Commission, including under the heading "Risk Factors" contained in our most recently filed periodic reports on Form 10-K and Form 10-Q filed with the Securities and Exchange Commission. Forward-looking statements are not guarantees of future performance, and our actual results of operations, financial condition, and liquidity, and the development of the industry in which we operate, may differ materially from the forward-looking statements contained in this press release. Any forward-looking statements that we make in this press release speak only as of the date of this press release. We assume no obligation to update our forward-looking statements, whether as a result of new information, future events, or otherwise, after the date of this press release.
Investor Relations Contact
Corey Davis, Ph.D.
LifeSci Advisors
[email protected]
+1 212 915 2577
Media Relations Contact
Virginia Amann
ENTENTE Network of Companies
[email protected]
+1 833 500 0061 ext 1
SOURCE: Protagonist Therapeutics
View the original press release on ACCESS Newswire
A.Rodriguezv--AMWN