
-
 'Promised to us': The Israelis dreaming of settling south Lebanon
'Promised to us': The Israelis dreaming of settling south Lebanon
-
'Rare, meaningful': North Korean football team ventures into South

-
 In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
-
Historic Swiss solar-powered plane crashes into sea

-
 A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
-
Indian pharma fuels Africa's 'zombie drug' and opioid crisis

-
 After months of blackout, Iran gives internet to select few
After months of blackout, Iran gives internet to select few
-
Wood urges New Zealand to 'create some history' at World Cup

-
 In Washington, the fight to preserve Black cemeteries
In Washington, the fight to preserve Black cemeteries
-
US children's book author sentenced to life after poisoning husband

-
 Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
-
US renews offer of $100 mn to Cuba if it cooperates

-
 City still 'alive' but need Arsenal slip: Guardiola
City still 'alive' but need Arsenal slip: Guardiola
-
Man City ease past Palace to keep pressure on Arsenal

-
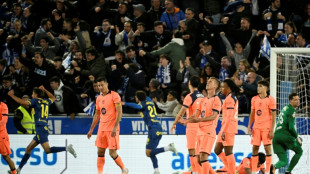 Alaves end champions Barca's bid for 100-point record
Alaves end champions Barca's bid for 100-point record
-
US jury begins deliberations on 737 MAX victim suit against Boeing

-
 PSG clinch fifth straight Ligue 1 title
PSG clinch fifth straight Ligue 1 title
-
Inter Milan win Italian Cup to secure domestic double

-
 Man City see off Palace to keep pressure on Arsenal
Man City see off Palace to keep pressure on Arsenal
-
Trump and Xi set for high-stakes talks in Beijing

-
 S&P 500, Nasdaq end at records as oil prices retreat
S&P 500, Nasdaq end at records as oil prices retreat
-
Iran holds World Cup send-off for national football team

-
 McIlroy's toe 'totally fine' after nine-hole PGA practice
McIlroy's toe 'totally fine' after nine-hole PGA practice
-
Rare 'Ocean Dream' blue-green diamond sells for $17 mn at auction

-
 California says probing possible violations over World Cup ticket sales
California says probing possible violations over World Cup ticket sales
-
US races to secure rare earths to rebuild depleted arsenal

-
 Matthew Perry drug middleman jailed for two years
Matthew Perry drug middleman jailed for two years
-
Warsh confirmed as Fed chair as central bank faces Trump assault

-
 Kohli ton powers Bengaluru past Kolkata, to top of IPL
Kohli ton powers Bengaluru past Kolkata, to top of IPL
-
Ex-Nicaragua guerrilla believes Ortega-Murillo days numbered

-
 Berlin launches scheme to swap trash for treats
Berlin launches scheme to swap trash for treats
-
Sarah Taylor named England men's fielding coach

-
 No plans for PGA outside USA or moving off May date
No plans for PGA outside USA or moving off May date
-
US Senate backs Trump on Iran war despite deadline lapse

-
 Key urges 'world-class' bowler Robinson to make England recall count
Key urges 'world-class' bowler Robinson to make England recall count
-
From Black Death to Covid, ships have long hosted outbreaks

-
 Furyk wants long-term US Ryder blueprint, maybe role for Tiger
Furyk wants long-term US Ryder blueprint, maybe role for Tiger
-
McIlroy back on course on eve of PGA despite blister

-
 Eulalio seizes control of drenched Giro d'Italia
Eulalio seizes control of drenched Giro d'Italia
-
New trial ordered for US lawyer convicted of murdering wife, son

-
 Stocks rise ahead of US-China summit
Stocks rise ahead of US-China summit
-
US wholesale prices jump 6.0% year-on-year in April, highest since 2022

-
 Nations drawing down oil stocks at record pace: IEA
Nations drawing down oil stocks at record pace: IEA
-
Carrick on brink of permanent Man Utd job: reports

-
 Strong US economy's resilience to shocks tested by Iran war
Strong US economy's resilience to shocks tested by Iran war
-
Italy cheers UK's Catherine on first foreign visit since cancer diagnosis

-
 Keys says players will strike over Grand Slam pay if 'necessary'
Keys says players will strike over Grand Slam pay if 'necessary'
-
Eurovision stage inspired by Viennese opera

-
 Gunshots at Philippine Senate as lawmaker wanted by ICC holds out
Gunshots at Philippine Senate as lawmaker wanted by ICC holds out
-
Winning worth the wait for Young no matter the ball


EU Regulators Say Charlotte's Web Hemp CBD Safety "Cannot Be Established" - So Why Is It Entering Medicare?
WASHINGTON, DC / ACCESS Newswire / May 8, 2026 / A growing conflict between international scientific findings and U.S. healthcare policy is raising urgent questions about whether Medicare beneficiaries are being exposed to cannabinoid products that have not been proven safe.
In March 2026, the European Food Safety Authority (EFSA) issued a formal scientific rejection of Charlottes Web hemp product submission concluding that the safety of a carbon dioxide extract derived from Cannabis sativa L. - including products associated with leading U.S. brands - "cannot be established."

That conclusion was not based on politics. It was based on data.
According to EFSA, critical gaps included:
Significant portions of the product remaining uncharacterized
Lack of reliable toxicological studies on the actual material
Absence of human clinical data
Unknown allergenicity and long-term safety profile
The agency's bottom line was clear:
The safety of the product could not be determined.
Meanwhile in the United States
At nearly the same time, the Centers for Medicare & Medicaid Services (CMS) launched the Substance Access Beneficiary Engagement Incentive (BEI) program.
The initiative allows participating healthcare providers to discuss and furnish certain hemp, marijuana derived cannabinoid products to Medicare beneficiaries under Innovation Center authority.
Unlike traditional drug pathways, the BEI program does not require approval from the U.S. Food and Drug Administration.
That distinction is central to a federal lawsuit now pending:
Smart Approaches to Marijuana (SAM), et al. v. Robert F. Kennedy Jr., et al.
Case No. 1:26-cv-01081 (U.S. District Court for the District of Columbia)
The Scientific Gap
Under the FDA framework, products intended for therapeutic use typically undergo:
Controlled clinical trials
Dose standardization
Safety and toxicology evaluation
Manufacturing and stability validation
The BEI program operates outside that structure.
While CMS has described the initiative as an innovation model, critics argue it effectively introduces products into federally funded care environments before those scientific benchmarks are met.
Why This Matters for Seniors
Medicare beneficiaries represent one of the most medically complex populations in the healthcare system.
Many patients:
Take multiple medications (polypharmacy)
Have chronic conditions
Are more vulnerable to drug interactions
Cannabinoid compounds, including THC, are known to interact with metabolic pathways such as CYP450 enzymes, which are involved in processing many common medications.
Without controlled studies in this population, the full safety profile remains uncertain.
Rapid Supply Chain Expansion
At the same time the program launched, several companies publicly announced positioning within the emerging healthcare supply chain:
Charlotte's Web highlighted alignment with CMS guidance
Cornbread Hemp announced institutional distribution through a national group purchasing organization
These developments signal rapid commercialization of a category that has not undergone FDA drug approval review.
A Regulatory Tension
The situation creates a notable contradiction:
International regulators say safety cannot be established
The FDA has not approved these products as medicines
Yet a federal healthcare program is enabling access within Medicare-linked systems
That tension is now at the center of federal litigation.
What the Court Will Decide
In SAM v. Kennedy (1:26-cv-01081), the court is evaluating whether CMS exceeded its statutory authority by introducing cannabinoid furnishing pathways without:
Formal rulemaking
Public notice and comment
FDA validation standards
A ruling is expected to determine whether the program:
Proceeds as implemented
Is narrowed or modified
Or is halted pending further review
The Core Question
This debate ultimately comes down to a fundamental issue in healthcare policy:
Should access to therapeutic products come before scientific validation-
or should science remain the gatekeeper?
Closing Perspective
The EFSA conclusion does not ban CBD products.
But it does highlight a critical reality:
The scientific evidence needed to establish safety is still incomplete.
As federal policy evolves, the question is not whether innovation should occur-
but whether it should occur before the science is fully understood,
particularly for the nation's most vulnerable patients.
CONTACT:
Madison Hisey
[email protected]
203-231-8583
SOURCE: MMJ International Holdings
View the original press release on ACCESS Newswire
A.Rodriguezv--AMWN