
-
 'Promised to us': The Israelis dreaming of settling south Lebanon
'Promised to us': The Israelis dreaming of settling south Lebanon
-
'Rare, meaningful': North Korean football team ventures into South

-
 In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
-
Historic Swiss solar-powered plane crashes into sea

-
 A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
-
Indian pharma fuels Africa's 'zombie drug' and opioid crisis

-
 After months of blackout, Iran gives internet to select few
After months of blackout, Iran gives internet to select few
-
Wood urges New Zealand to 'create some history' at World Cup

-
 In Washington, the fight to preserve Black cemeteries
In Washington, the fight to preserve Black cemeteries
-
US children's book author sentenced to life after poisoning husband

-
 Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
-
US renews offer of $100 mn to Cuba if it cooperates

-
 City still 'alive' but need Arsenal slip: Guardiola
City still 'alive' but need Arsenal slip: Guardiola
-
Man City ease past Palace to keep pressure on Arsenal

-
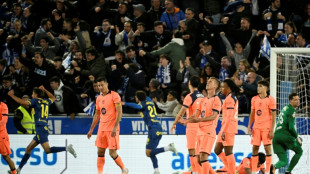 Alaves end champions Barca's bid for 100-point record
Alaves end champions Barca's bid for 100-point record
-
US jury begins deliberations on 737 MAX victim suit against Boeing

-
 PSG clinch fifth straight Ligue 1 title
PSG clinch fifth straight Ligue 1 title
-
Inter Milan win Italian Cup to secure domestic double

-
 Man City see off Palace to keep pressure on Arsenal
Man City see off Palace to keep pressure on Arsenal
-
Trump and Xi set for high-stakes talks in Beijing

-
 S&P 500, Nasdaq end at records as oil prices retreat
S&P 500, Nasdaq end at records as oil prices retreat
-
Iran holds World Cup send-off for national football team

-
 McIlroy's toe 'totally fine' after nine-hole PGA practice
McIlroy's toe 'totally fine' after nine-hole PGA practice
-
Rare 'Ocean Dream' blue-green diamond sells for $17 mn at auction

-
 California says probing possible violations over World Cup ticket sales
California says probing possible violations over World Cup ticket sales
-
US races to secure rare earths to rebuild depleted arsenal

-
 Matthew Perry drug middleman jailed for two years
Matthew Perry drug middleman jailed for two years
-
Warsh confirmed as Fed chair as central bank faces Trump assault

-
 Kohli ton powers Bengaluru past Kolkata, to top of IPL
Kohli ton powers Bengaluru past Kolkata, to top of IPL
-
Ex-Nicaragua guerrilla believes Ortega-Murillo days numbered

-
 Berlin launches scheme to swap trash for treats
Berlin launches scheme to swap trash for treats
-
Sarah Taylor named England men's fielding coach

-
 No plans for PGA outside USA or moving off May date
No plans for PGA outside USA or moving off May date
-
US Senate backs Trump on Iran war despite deadline lapse

-
 Key urges 'world-class' bowler Robinson to make England recall count
Key urges 'world-class' bowler Robinson to make England recall count
-
From Black Death to Covid, ships have long hosted outbreaks

-
 Furyk wants long-term US Ryder blueprint, maybe role for Tiger
Furyk wants long-term US Ryder blueprint, maybe role for Tiger
-
McIlroy back on course on eve of PGA despite blister

-
 Eulalio seizes control of drenched Giro d'Italia
Eulalio seizes control of drenched Giro d'Italia
-
New trial ordered for US lawyer convicted of murdering wife, son

-
 Stocks rise ahead of US-China summit
Stocks rise ahead of US-China summit
-
US wholesale prices jump 6.0% year-on-year in April, highest since 2022

-
 Nations drawing down oil stocks at record pace: IEA
Nations drawing down oil stocks at record pace: IEA
-
Carrick on brink of permanent Man Utd job: reports

-
 Strong US economy's resilience to shocks tested by Iran war
Strong US economy's resilience to shocks tested by Iran war
-
Italy cheers UK's Catherine on first foreign visit since cancer diagnosis

-
 Keys says players will strike over Grand Slam pay if 'necessary'
Keys says players will strike over Grand Slam pay if 'necessary'
-
Eurovision stage inspired by Viennese opera

-
 Gunshots at Philippine Senate as lawmaker wanted by ICC holds out
Gunshots at Philippine Senate as lawmaker wanted by ICC holds out
-
Winning worth the wait for Young no matter the ball


Onco-Innovations Progresses Toward IND Preparation as Nucro-Technics Commences Bioanalysis for PNKP Inhibitor Technology
VANCOUVER, BC / ACCESS Newswire / May 8, 2026 / Onco-Innovations Limited (CBOE CA:ONCO)(OTCQB:ONNVF)(Frankfurt:W1H, WKN:A3EKSZ) ("Onco" or the "Company") announces that Nucro-Technics Inc. ("Nucro-Technics"), a Toronto-based contract research organization collaborating with Onco on preclinical development, has commenced advanced bioanalytical work in support of the Company's ongoing non-GLP1 pharmacokinetic (PK) and biodistribution animal model study involving A83B4C63, the active pharmaceutical ingredient ("API") for Onco's exclusively-licensed PNKP Inhibitor Technology (the "Technology"). The initiation of this work represents a significant step forward in translating the Technology from early-stage discovery into a development-ready therapeutic candidate.
Nucro-Technics has begun developing and executing specialized liquid chromatography-mass spectrometry (LC-MS) analytical methods2 required to measure the API within biological samples generated during the animal model study. These analytical tools will allow researchers to accurately assess how the compound behaves in vivo, supporting a deeper understanding of its exposure, distribution, and potential therapeutic relevance. Although highly technical, this work provides the essential measurement capabilities on which meaningful PK and biodistribution insights depend.
The start of this analytical phase marks a natural progression of Onco's preclinical program, enabling the Company to generate the datasets needed to refine dosing strategies, inform formulation work, and prepare for subsequent GLP-compliant studies. This stage builds upon earlier development efforts and strengthens the foundation of the Company's IND-enabling plan (see Company press release dated September 28, 2025).
"The initiation of this analytical phase gives us the capability to generate the data that truly drives decision-making in early drug development. It allows us to understand how our PNKP inhibitor behaves in vivo and ensures that each subsequent study is grounded in high-quality evidence. This is the type of foundational work that moves a program from concept toward a real clinical candidate," said Thomas O'Shaughnessy, CEO of Onco-Innovations.
Nucro-Technics will continue method development, perform non-GLP analyses, and deliver the corresponding reports in alignment with the study's operational timelines. This coordinated effort supports Onco's broader goal of advancing the Technology toward clinical evaluation and expanding the pipeline of next-generation DNA Damage Response Inhibitor (DDRi) therapies.
About Onco-Innovations Limited
Onco-Innovations is a Canadian-based company dedicated to cancer research and treatment, specializing in oncology. Onco's mission is to pursue the prevention and treatment of cancer through pioneering research and innovative solutions. The company has secured an exclusive worldwide license to patented technology that targets solid tumours.
ON BEHALF OF ONCO-INNOVATIONS LIMITED,
"Thomas O'Shaughnessy"
Chief Executive Officer
For more information, please contact:
Thomas O'Shaughnessy
Chief Executive Officer
Tel: + 1 888 261 8055
[email protected]
Forward-Looking Statements Caution. This news release contains forward-looking statements, including in relation to the Company's business and plans generally, and other statements that are not historical facts. Forward-looking statements are often identified by terms such as "will", "may", "potential", "should", "anticipate", "expects" and similar expressions. All statements other than statements of historical fact, included in this release are forward-looking statements that involve risks and uncertainties. There can be no assurance that such statements will prove to be accurate and actual results and future events could differ materially from those anticipated in such statements. The reader is cautioned that assumptions used in the preparation of any forward-looking information may prove to be incorrect. Events or circumstances may cause actual results to differ materially from those predicted, as a result of numerous known and unknown risks, uncertainties, and other factors, many of which are beyond the control of the Company. The reader is cautioned not to place undue reliance on any forward-looking information. Such information, although considered reasonable by management at the time of preparation, may prove to be incorrect and actual results may differ materially from those anticipated. Forward-looking statements contained in this news release are expressly qualified by this cautionary statement. The forward-looking statements contained in this news release are made as of the date of this news release and the Company will update or revise publicly any of the included forward-looking statements as expressly required by applicable law.
1 Good Laboratory Practice
2 Liquid chromatography coupled to mass spectrometry (LC-MS) is the gold standard in bioanalysis for the development of quantitative assays to support drug development or therapeutic drug monitoring (see doi: 10.1007/s00216-021-03583-2 for more).
SOURCE: Onco-Innovations Limited
View the original press release on ACCESS Newswire
O.Norris--AMWN