-
 Why SpaceX IPO plan is generating so much buzz
Why SpaceX IPO plan is generating so much buzz
-
Thailand continues Cambodia strikes despite Trump truce calls

-
 US envoy to meet Zelensky, Europe leaders in Berlin this weekend
US envoy to meet Zelensky, Europe leaders in Berlin this weekend
-
North Korea acknowledges its troops cleared mines for Russia

-
 US unseals warrant for tanker seized off Venezuelan coast
US unseals warrant for tanker seized off Venezuelan coast
-
Cambodia says Thailand still bombing hours after Trump truce call

-
 Machado urges pressure so Maduro understands 'he has to go'
Machado urges pressure so Maduro understands 'he has to go'
-
Best Gold Investment Companies in USA Announced (Augusta Precious Metals, Lear Capital, Robinhood IRA and More Ranked)

-
 Leinster stutter before beating Leicester in Champions Cup
Leinster stutter before beating Leicester in Champions Cup
-
World stocks mostly slide, consolidating Fed-fuelled gains

-
 Crypto firm Tether bids for Juventus, is quickly rebuffed
Crypto firm Tether bids for Juventus, is quickly rebuffed
-
Union sink second-placed Leipzig to climb in Bundesliga

-
 US Treasury lifts sanctions on Brazil Supreme Court justice
US Treasury lifts sanctions on Brazil Supreme Court justice
-
UK king shares 'good news' that cancer treatment will be reduced in 2026

-
 Wembanyama expected to return for Spurs in NBA Cup clash with Thunder
Wembanyama expected to return for Spurs in NBA Cup clash with Thunder
-
Five takeaways from Luigi Mangione evidence hearings

-
 UK's king shares 'good news' that cancer treatment will be reduced in 2026
UK's king shares 'good news' that cancer treatment will be reduced in 2026
-
Steelers' Watt undergoes surgery to repair collapsed lung

-
 Iran detains Nobel-prize winner in 'brutal' arrest
Iran detains Nobel-prize winner in 'brutal' arrest
-
NBA Cup goes from 'outside the box' idea to smash hit

-
 UK health service battles 'super flu' outbreak
UK health service battles 'super flu' outbreak
-
Can Venezuela survive US targeting its oil tankers?

-
 Democrats release new cache of Epstein photos
Democrats release new cache of Epstein photos
-
Colombia's ELN guerrillas place communities in lockdown citing Trump 'intervention' threats

-
 'Don't use them': Tanning beds triple skin cancer risk, study finds
'Don't use them': Tanning beds triple skin cancer risk, study finds
-
Nancy aims to restore Celtic faith with Scottish League Cup final win

-
 Argentina fly-half Albornoz signs for Toulon until 2030
Argentina fly-half Albornoz signs for Toulon until 2030
-
Trump says Thailand, Cambodia have agreed to stop border clashes

-
 Salah in Liverpool squad for Brighton after Slot talks - reports
Salah in Liverpool squad for Brighton after Slot talks - reports
-
Marseille coach tips Greenwood as 'potential Ballon d'Or'

-
 Draw marks 'starting gun' toward 2026 World Cup, Vancouver says
Draw marks 'starting gun' toward 2026 World Cup, Vancouver says
-
Thai PM says asked Trump to press Cambodia on border truce

-
 Salah admired from afar in his Egypt home village as club tensions swirl
Salah admired from afar in his Egypt home village as club tensions swirl
-
World stocks retrench, consolidating Fed-fuelled gains

-
 Brazil left calls protests over bid to cut Bolsonaro jail time
Brazil left calls protests over bid to cut Bolsonaro jail time
-
Trump attack on Europe migration 'disaster' masks toughening policies

-
 US plan sees Ukraine joining EU in 2027, official tells AFP
US plan sees Ukraine joining EU in 2027, official tells AFP
-
'Chilling effect': Israel reforms raise press freedom fears

-
 Iran frees child bride sentenced to death over husband's killing: activists
Iran frees child bride sentenced to death over husband's killing: activists
-
No doubting Man City boss Guardiola's passion says Toure

-
 Youthful La Rochelle name teen captain for Champions Cup match in South Africa
Youthful La Rochelle name teen captain for Champions Cup match in South Africa
-
World stocks consolidate Fed-fuelled gains

-
 British 'Aga saga' author Joanna Trollope dies aged 82
British 'Aga saga' author Joanna Trollope dies aged 82
-
Man Utd sweat on Africa Cup of Nations trio

-
 EU agrees three-euro small parcel tax to tackle China flood
EU agrees three-euro small parcel tax to tackle China flood
-
Taylor Swift breaks down in Eras documentary over Southport attack

-
 Maresca 'relaxed' about Chelsea's rough patch
Maresca 'relaxed' about Chelsea's rough patch
-
France updates net-zero plan, with fossil fuel phaseout

-
 Nowhere to pray as logs choke flood-hit Indonesian mosque
Nowhere to pray as logs choke flood-hit Indonesian mosque
-
In Pakistan, 'Eternal Love' has no place on YouTube


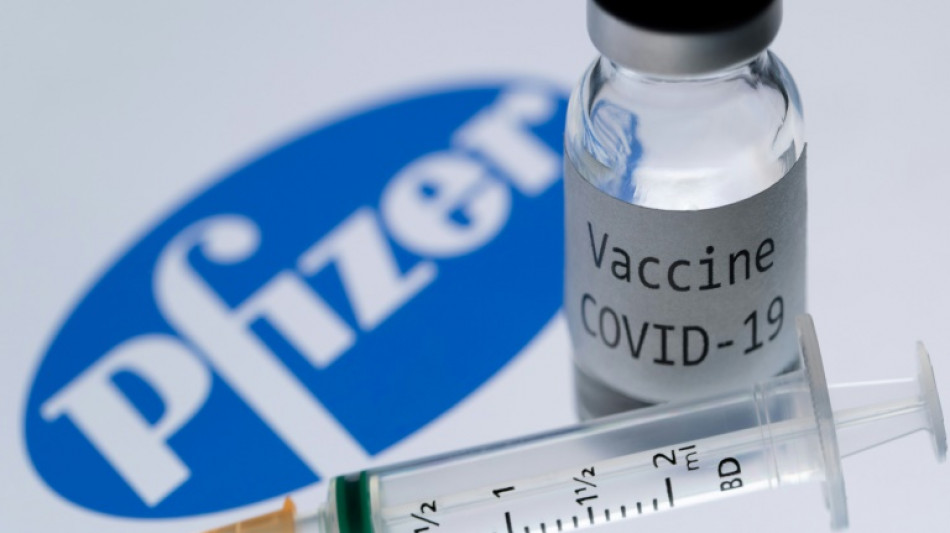
Pfizer seeks US approval of Covid vaccine for children under 5
Pfizer and BioNTech said Tuesday they are seeking emergency authorization from US health regulators for use of their Covid vaccine for children aged over six months and under five years.
If the Food and Drug Administration (FDA) authorizes the two-shot regimen, it will become the first Covid vaccine available to this age group in the United States.
The companies said Tuesday that they started submitting their formal application "following a request" from the FDA, which seemingly wants to get the process moving quickly.
In a tweet soon after the announcement, the FDA said it will hold a meeting in two weeks, on February 15, to consider the request.
"Having a safe and effective vaccine available for children in this age group is a priority for the agency," said interim FDA head Janet Woodcock, who added that the FDA asked for the application "in light of the recent Omicron surge."
The companies are seeking authorization for only two doses of their vaccine, but believe a third will be needed "to achieve high levels of protection against current and potential future variants," Pfizer CEO Albert Bourla said in a statement.
"If two doses are authorized, parents will have the opportunity to begin a Covid-19 vaccination series for their children while awaiting potential authorization of a third dose," he added.
To limit side effects for this young age group, Pfizer chose to significantly decrease the dosage of its vaccine, opting for only three micrograms per jab versus 30 for those over 12 years old, and 10 for ages five to 11.
The company's researchers concluded last fall that low doses of the vaccine provided protection in children up to two years old but not in those aged two to five, and announced in December they would add a third dose to their trials.
Data on the three-dose regimen is "expected in the coming months and will be submitted to the FDA to support a potential expansion" of this initial request, Pfizer and BioNTech said in the statement.
If the FDA authorizes the emergency use of the vaccine, an advisory committee for the Centers for Disease Control and Prevention (CDC) will then meet to decide to recommend its use or not.
- Slower uptake among minors -
There are about 23 million children under the age of five in the United States, and two years after the pandemic began, many parents are waiting impatiently to vaccinate their young children against the coronavirus.
The Pfizer-BioNTech vaccine was approved three months ago for emergency use in children from five to 11, but uptake has been relatively slow.
In that age group, only 30 percent have received at least one dose and around 22 percent are fully vaccinated, according to the Centers for Disease Control and Prevention (CDC).
Young children are less susceptible to falling seriously ill with Covid-19, but some nonetheless end up hospitalized by the virus.
New pediatric Covid hospitalizations hit a record high in the United States in December as the Omicron strain spread rapidly.
Some cases of the life-threatening illness MIS-C (multi-system inflammatory syndrome in children), have also been reported in young children with Covid.
CDC data shows that about 400 children aged four and under have died from Covid-19 in the United States since the pandemic began.
P.Mathewson--AMWN