
-
 Kenya's new poaching problem: smuggling Giant Harvester Ants
Kenya's new poaching problem: smuggling Giant Harvester Ants
-
WHO kicks off annual assembly amid hantavirus, Ebola crises

-
 S. Korean blockbuster 'Hope' underscores growing film ambition
S. Korean blockbuster 'Hope' underscores growing film ambition
-
Train driver charged after deadly Bangkok bus collision

-
 Angry Chinese table tennis fans demand apology for flag gaffe
Angry Chinese table tennis fans demand apology for flag gaffe
-
India's lifeline ferry across strategic archipelago

-
 Encroaching world threatens India's last 'uncontacted' tribe
Encroaching world threatens India's last 'uncontacted' tribe
-
India's strategic $9 bn megaport plan for pristine island

-
 In Tierra del Fuego, a hunt for the rodent carrier of hantavirus
In Tierra del Fuego, a hunt for the rodent carrier of hantavirus
-
Mitchell leads Cavs past top-seeded Detroit into NBA East finals

-
 China's April consumption, factory output growth slowest in years
China's April consumption, factory output growth slowest in years
-
Asian stocks sink, oil rises on US-Iran deadlock
-
 Cleveland Cavaliers eliminate top-seeded Detroit from NBA playoffs
Cleveland Cavaliers eliminate top-seeded Detroit from NBA playoffs
-
Who could be the 2026 World Cup's breakout star?

-
 Humble PGA champ Rai celebrates English, Indian, Kenyan heritage
Humble PGA champ Rai celebrates English, Indian, Kenyan heritage
-
Hantavirus-hit cruise ship nears end of voyage, to dock in Rotterdam

-
 He said, she said, AI said: Wall Street sex scandal rivets and confounds
He said, she said, AI said: Wall Street sex scandal rivets and confounds
-
UN General Assembly to take up climate change 'obligations' resolution

-
 Four takeaways from Musk vs OpenAI trial
Four takeaways from Musk vs OpenAI trial
-
Jury to decide fate of Musk's blockbuster suit against OpenAI

-
 Frustrated McIlroy drops F-bomb in exchange with PGA heckler
Frustrated McIlroy drops F-bomb in exchange with PGA heckler
-
Defending champion Palou storms to Indy 500 pole

-
 Messi shines as Inter Miami finally win at new stadium
Messi shines as Inter Miami finally win at new stadium
-
Shai Gilgeous-Alexander wins second straight NBA MVP award

-
 White House mass prayer event seeks to reclaim US Christian roots
White House mass prayer event seeks to reclaim US Christian roots
-
International dive group joins Maldives search for missing Italians

-
 'Staggering' Iran toll drives up global executions: Amnesty
'Staggering' Iran toll drives up global executions: Amnesty
-
Agronomics Limited Announces Net Asset Value Calculation as at 31 March 2026

-
 Santa Barbara Schools Sexual Assault Complaint by Veen Firm
Santa Barbara Schools Sexual Assault Complaint by Veen Firm
-
InterContinental Hotels Group PLC Announces Transaction in Own Shares - May 18

-
 Rai wins first major at PGA with back-nine birdie blitz
Rai wins first major at PGA with back-nine birdie blitz
-
Woad bags second LPGA title at Queen City Championship

-
 Lebanon says Israeli strikes kill 7 as Hezbollah condemns talks
Lebanon says Israeli strikes kill 7 as Hezbollah condemns talks
-
Revived La Rochelle trounce Top 14 leaders Toulouse

-
 PSG beaten by Paris FC in Ligue 1 as Lille qualify for Champions League
PSG beaten by Paris FC in Ligue 1 as Lille qualify for Champions League
-
Griezmann apologetic on emotional Atletico Madrid farewell

-
 Raging Neymar forced off by refereeing error as Santos lose
Raging Neymar forced off by refereeing error as Santos lose
-
Sinner extends Masters tournament streak on home turf, eyes French Open

-
 Canadian cruise passenger confirmed positive for hantavirus
Canadian cruise passenger confirmed positive for hantavirus
-
England see off gutsy France to clinch another Women's Six Nations

-
 Sevilla safe despite Real Madrid defeat, Mallorca on brink
Sevilla safe despite Real Madrid defeat, Mallorca on brink
-
UK police detail arrests after far-right rally and counter demo

-
 Smalley tees off with PGA lead and stars in hot pursuit
Smalley tees off with PGA lead and stars in hot pursuit
-
Trump issues dire warning to Iran to accept peace deal

-
 West Ham on brink of Premier League relegation, Man Utd seal third
West Ham on brink of Premier League relegation, Man Utd seal third
-
Bulgaria's Eurovision winner flies home to rapturous welcome

-
 Starc takes four to keep Delhi alive in IPL
Starc takes four to keep Delhi alive in IPL
-
Kyiv residents protest 'dangerous' civil code, call for LGBTQ rights
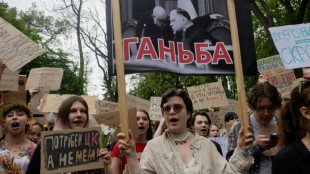
-
 Modiba thunderbolt gives Sundowns victory in African final first leg
Modiba thunderbolt gives Sundowns victory in African final first leg
-
World champions England see off France to clinch another Women's Six Nations


Mixed results for Moderna mRNA flu vaccine trial
US biotech company Moderna said Thursday it had mixed results from a large-scale trial of its mRNA flu shot, based on the same technology used in its successful Covid-19 vaccine.
Moderna's experimental mRNA-1010 flu shot is "quadrivalent," meaning it targets four strains of flu: A/H1N1, A/H3N2, B/Yamagata and B/Victoria -- selected based on recommendations by the World Health Organization (WHO).
The Massachusetts-based company said that its flu shot generated an immune response against influenza A strains that was equal or superior to that of already licensed vaccines.
However, it fell short of the already approved vaccines against strains of the less-common influenza B, Moderna said in a statement.
"Today's results represent an important step forward in the development of mRNA-based influenza vaccines," Moderna president Stephen Hoge said.
"We have already updated the vaccine that we believe could improve immune responses against influenza B and will seek to quickly confirm those improvements in an upcoming clinical study."
The Phase 3 trial of the mRNA shot involved 6,102 adults in Argentina, Australia, Colombia, Panama, and the Philippines during the Southern Hemisphere influenza season.
Participants received a single dose of mRNA-1010 or a single dose of a licensed influenza vaccine.
Moderna said 70 percent of the mRNA-1010 recipients reported adverse reactions such as headaches, swelling and fatigue compared to 48 percent in the other group.
Virus strains have to be selected six to nine months before the vaccines are intended to be used, and their efficacy is approximately 40 to 60 percent.
Moderna is simultaneously conducting an efficacy trial of its vaccine.
Moderna and other vaccine manufacturers, including Sanofi, hope that mRNA technology -- which provokes an immune response by delivering genetic molecules containing the code for key parts of a pathogen into human cells -- can accelerate immunization development and production, and heighten efficacy.
There are some three to five million severe influenza cases annually worldwide and between 290,000 and 650,000 deaths, the WHO says.
Moderna's stock price was down more than six percent in after-hours trading in New York.
L.Durand--AMWN