
-
 Griezmann apologetic on emotional Atletico Madrid farewell
Griezmann apologetic on emotional Atletico Madrid farewell
-
Raging Neymar forced off by refereeing error as Santos lose

-
 Sinner extends Masters tournament streak on home turf, eyes French Open
Sinner extends Masters tournament streak on home turf, eyes French Open
-
Canadian cruise passenger confirmed positive for hantavirus

-
 England see off gutsy France to clinch another Women's Six Nations
England see off gutsy France to clinch another Women's Six Nations
-
Sevilla safe despite Real Madrid defeat, Mallorca on brink

-
 UK police detail arrests after far-right rally and counter demo
UK police detail arrests after far-right rally and counter demo
-
Smalley tees off with PGA lead and stars in hot pursuit

-
 Trump issues dire warning to Iran to accept peace deal
Trump issues dire warning to Iran to accept peace deal
-
West Ham on brink of Premier League relegation, Man Utd seal third

-
 Bulgaria's Eurovision winner flies home to rapturous welcome
Bulgaria's Eurovision winner flies home to rapturous welcome
-
Starc takes four to keep Delhi alive in IPL

-
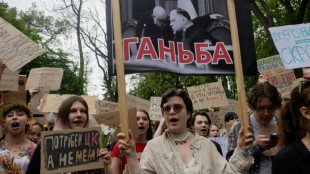 Kyiv residents protest 'dangerous' civil code, call for LGBTQ rights
Kyiv residents protest 'dangerous' civil code, call for LGBTQ rights
-
Modiba thunderbolt gives Sundowns victory in African final first leg

-
 World champions England see off France to clinch another Women's Six Nations
World champions England see off France to clinch another Women's Six Nations
-
Taiwan's leader says island will not be 'traded away'

-
 Sinner wins Italian Open, extends Masters tournament streak
Sinner wins Italian Open, extends Masters tournament streak
-
'Michael' moonwalks back to top of N. America box office

-
 Putter powers sizzling Kitayama to record 63 at PGA
Putter powers sizzling Kitayama to record 63 at PGA
-
Travolta channelled film greats in low-thrust plane movie

-
 Scotland rugby great Scott Hastings dead at 61 - SRU
Scotland rugby great Scott Hastings dead at 61 - SRU
-
Fujimori and Sanchez advance to Peru runoff: official results

-
 Italian PM meets victims of Modena car incident
Italian PM meets victims of Modena car incident
-
'Fight relentlessly': Ukraine commander vows strikes into Russia

-
 Kitayama fires sizzling 63 at PGA as No.1 Scheffler starts
Kitayama fires sizzling 63 at PGA as No.1 Scheffler starts
-
Fernandes equals Premier League assist record in Man Utd win, West Ham brace for Newcastle

-
 Ireland thrash Scotland 54-5 in Women's Six Nations to finish third
Ireland thrash Scotland 54-5 in Women's Six Nations to finish third
-
Vingegaard climbs to victory as Eulalio holds firm in pink

-
 Carrick expects clarity on Man Utd future in 'coming days'
Carrick expects clarity on Man Utd future in 'coming days'
-
Eyewitness says Modena tragedy could have been even worse

-
 Around 10 'new' victims in France's Epstein probe: prosecutor
Around 10 'new' victims in France's Epstein probe: prosecutor
-
Shock threat by billionaire Bollore's Canal+ group rocks French cinema

-
 Kohli, Venkatesh dazzle as Bengaluru qualify for IPL play-offs
Kohli, Venkatesh dazzle as Bengaluru qualify for IPL play-offs
-
Probes ongoing into alleged abuse at 84 Paris preschools: prosecutor

-
 Di Giannantonio wins Catalan MotoGP Grand Prix, Alex Marquez injured in horror crash
Di Giannantonio wins Catalan MotoGP Grand Prix, Alex Marquez injured in horror crash
-
Fernandes equals assist record as Man Utd edge Forest thriller

-
 Earps to leave PSG, in talks with London City Lionesses
Earps to leave PSG, in talks with London City Lionesses
-
Bowlers, Joy put Bangladesh on top in second Pakistan Test

-
 Alex Marquez injured in horrific Catalan MotoGP crash
Alex Marquez injured in horrific Catalan MotoGP crash
-
'Message for friends and foes': Libyan National Army conducts grand exercises

-
 Bayern's Neuer sidelined again with leg issue
Bayern's Neuer sidelined again with leg issue
-
Adam Driver shuts down question about clashes with Lena Dunham

-
 British soprano Felicity Lott dies aged 79
British soprano Felicity Lott dies aged 79
-
Roma near Champions League return with derby triumph, Napoli secure top four

-
 Denmark's Antonsen wins badminton Thailand Open title
Denmark's Antonsen wins badminton Thailand Open title
-
'Toxic' males Trump, Putin, Netanyahu to blame for wars, says star Bardem

-
 Iran have 'constructive' meeting with FIFA over World Cup preparations
Iran have 'constructive' meeting with FIFA over World Cup preparations
-
'Peaky Blinders' creator says he has licence to reinvent James Bond

-
 Xabi Alonso appointed Chelsea manager on four-year deal
Xabi Alonso appointed Chelsea manager on four-year deal
-
Mass Ukraine drone barrage kills 4 in Russia: Moscow


Highly awaited Alzheimer's drug hit by delays
Eli Lilly's highly anticipated Alzheimer's drug has been held back for further review by regulators, the US pharmaceutical giant said Friday, in a blow for patients with the devastating brain disorder.
Donanemab has been found to slow cognitive decline in the early stages of the disease during a clinical trial -- but there was also a high rate of side effects, including deaths.
The Food and Drug Administration (FDA) "has informed Lilly it wants to further understand topics related to evaluating the safety and efficacy of donanemab," the company said in a statement Friday.
The regulator told the Indiana-based company it would convene a new meeting of experts, but hadn't provided a firm date. "As a result, the timing of expected FDA action on donanemab will be delayed beyond the first quarter of 2024."
"We are confident in donanemab's potential to offer very meaningful benefits to people with early symptomatic Alzheimer's disease," said Anne White, the company's executive vice president.
She added the FDA's decision to have a new meeting was "unexpected," but "We will work with the FDA and the stakeholders in the community to make that presentation and answer all questions."
Donanemab is an intravenously injected antibody that targets the build up beta-amyloid, a protein found in the brains of many patients with Alzheimer's.
Another anti-amyloid therapy called Leqembi, which was developed by Eisai of Japan and Biogen of Massachusetts, was granted full approval by the FDA last July and is now accessible through government-run health insurance for the elderly called Medicare.
- Slows decline, but risky -
In a paper published in the Journal of the American Medical Association last year, researchers found donanemab slowed cognitive and functional decline in patients who have early symptoms of the disease.
Forty-seven percent of those who received the drug showed no signs of cognitive decline after one year of treatment, compared to 29 percent who received a placebo.
Serious adverse events, including brain bleeds, occurred in 17.4 percent of those who received donanemab and 15.8 percent of those who received a placebo.
There were also four deaths: three in the donanemab group and one in the placebo group, but all the fatalities were considered a result of the treatment they received.
The trial recruited participants aged 60 to 85 with early symptomatic Alzheimer's, either mild cognitive impairment or Alzheimer's disease with mild dementia.
The news comes after the first Alzheimer's drug to be approved was pulled from the market in January.
The FDA awarded accelerated approval to Aduhelm in June 2021, a decision that was contentious at the time because the agency overruled its own independent advisors, who found there was insufficient evidence of benefit.
Biogen, which co-developed Aduhelm with Eisai, said it was discontinuing Aduhelm to focus its efforts of Leqembi.
Alzheimer's is the most common form of dementia. More than one in nine people over 65 develop the condition, which worsens over time, robbing them of their memories and independence, according to the US Alzheimer's Association.
P.Santos--AMWN


