
-
 Iranian filmmaker Farhadi condemns Middle East war, protest massacres
Iranian filmmaker Farhadi condemns Middle East war, protest massacres
-
'Better than the Oscar': John Travolta gets surprise Cannes prize

-
 Marsh muscle motors Lucknow to victory over Chennai
Marsh muscle motors Lucknow to victory over Chennai
-
Judge declares mistrial in Weinstein case as jury fails to reach verdict

-
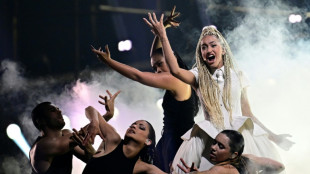 Eurovision finalists tune up as boycotting Spain digs in
Eurovision finalists tune up as boycotting Spain digs in
-
Indonesia's first giant panda is set to charm the public

-
 Cheer and tears as African refugee rap film 'Congo Boy' charms Cannes
Cheer and tears as African refugee rap film 'Congo Boy' charms Cannes
-
Norwegian Ruud rolls into Italian Open final, Sinner set for Medvedev clash

-
 Bolivia government says deal reached with protesting miners
Bolivia government says deal reached with protesting miners
-
Showdowns and spycraft on Trump-Xi summit sidelines

-
 Smalley seizes PGA lead with Matsuyama making a charge
Smalley seizes PGA lead with Matsuyama making a charge
-
Acosta quickest in practice for Catalan MotoGP

-
 Nuno wants VAR 'consistency' as West Ham fight to avoid relegation
Nuno wants VAR 'consistency' as West Ham fight to avoid relegation
-
Vingegaard powers to maiden Giro stage victory

-
 Iran to hold pre-World Cup training camp in Turkey: media
Iran to hold pre-World Cup training camp in Turkey: media
-
US scraps deployment of 4,000 troops to Poland

-
 Ukraine vows more strikes on Russia after attack on Kyiv kills 24
Ukraine vows more strikes on Russia after attack on Kyiv kills 24
-
Bayern veteran Neuer signs one-year contract extension

-
 Ukraine can down Russian drones en masse. But missiles are a problem
Ukraine can down Russian drones en masse. But missiles are a problem
-
Israeli strikes wound dozens in Lebanon as talks in US enter second day

-
 'Everybody wants Hearts to win', says Celtic's O'Neill ahead of title decider
'Everybody wants Hearts to win', says Celtic's O'Neill ahead of title decider
-
Scheffler stumbles from share of lead at windy PGA

-
 New deadly Ebola outbreak hits DR Congo
New deadly Ebola outbreak hits DR Congo
-
Farke calls for Leeds owners to match his ambition

-
 Zverev pulls out of home event in Hamburg with back injury
Zverev pulls out of home event in Hamburg with back injury
-
Xi, Trump eke small wins from talks but no major deals: analysts

-
 De Ligt to miss World Cup after back surgery
De Ligt to miss World Cup after back surgery
-
England's Rice braces for 'hate and love' at World Cup

-
 Milan Fashion Week says will ask brands not to show fur
Milan Fashion Week says will ask brands not to show fur
-
French-German tank maker KNDS to push ahead with IPO

-
 Man City campaign a success regardless of trophies: Guardiola
Man City campaign a success regardless of trophies: Guardiola
-
'World's oldest dog' contender dies in France aged 30

-
 No.1 Scheffler opens with bogey to fall from share of PGA lead
No.1 Scheffler opens with bogey to fall from share of PGA lead
-
Carrick says Man Utd future to be decided 'pretty soon'

-
 'Out of shape' Lukaku named in Belgium World Cup squad
'Out of shape' Lukaku named in Belgium World Cup squad
-
Hearts ready to 'rip up the script' in Celtic title showdown

-
 X pledges crackdown on illegal content in UK
X pledges crackdown on illegal content in UK
-
Possible contenders in UK Labour Party leadership race

-
 Germany's Merz says wouldn't advise young people to move to US
Germany's Merz says wouldn't advise young people to move to US
-
Israel strikes Lebanon as talks in US enter second day

-
 Kyiv in mourning after 24 killed as Ukraine, Russia swap POWs
Kyiv in mourning after 24 killed as Ukraine, Russia swap POWs
-
Beckham becomes first British billionaire sportsman

-
 Aussie star, Danish clubbing ode through to Eurovision final
Aussie star, Danish clubbing ode through to Eurovision final
-
German Oscar winner Huller feels war guilt 'every day'

-
 Thai lawmakers vote to revive clean air bill
Thai lawmakers vote to revive clean air bill
-
Bayern warn that Canada's Davies struggling to be fit for World Cup

-
 Long-serving Coleman to end Everton career at end of season
Long-serving Coleman to end Everton career at end of season
-
Energy-hungry German industries in decline since Ukraine war: data

-
 Gordon may have made last Newcastle appearance: Howe
Gordon may have made last Newcastle appearance: Howe
-
Denmark's Queen Margrethe has angioplasty in hospital: palace


US limits Covid boosters to over-65s or those at high risk
The United States will limit Covid-19 boosters to people over 65 or those at risk of serious illness, while requiring vaccine makers to run fresh clinical trials before offering shots to younger and healthier individuals, officials said Tuesday.
Writing in the New England Journal of Medicine, the Food and Drug Administration's Vinayak Prasad and Commissioner Martin Makary framed the policy shift as "evidence-based" and would align the United States more closely with guidance in Europe.
But it comes as Health Secretary Robert F. Kennedy Jr., a longtime vaccine skeptic, pushes to remake federal public health policy.
Kennedy previously led a nonprofit that was critical of immunization programs, and during the pandemic petitioned the FDA to revoke Covid vaccine authorizations, citing rare side effects including heart inflammation.
Prasad and Makary praised the initial Covid-19 vaccine rollout as "a major scientific, medical, and regulatory accomplishment," but argued that the benefits of repeated boosters for low-risk individuals are uncertain.
They criticized the US approach of recommending boosters for all adults regardless of age or health status, calling it a "one-size-fits-all" model based on the mistaken belief that Americans couldn't handle more nuanced, risk-based advice.
Rather than building public trust, they wrote, it had backfired -- fueling vaccine hesitancy that has spilled over into skepticism toward childhood shots, including those for measles.
The FDA said it would rely on lab test results to approve boosters for people who are over 65, or over six months old with at least one underlying condition.
But for healthy individuals between six months and 64 years, regulators will now require data from randomized trials.
"We simply don't know whether a healthy 52-year-old woman with a normal BMI (body mass index) who has had Covid-19 three times and has received six previous doses of a Covid-19 vaccine will benefit from the seventh dose," they wrote.
Some infectious disease experts welcomed the shift.
Amesh Adalja of Johns Hopkins University said it matched with the approach taken by other countries in a population that already carries significant immunity.
"For lower-risk individuals, the goal has always been less clear, as protection against infection is transient and they don't have a high risk of severe disease," he told AFP.
But others voiced concern about the practical consequences. Paul Offit, a leading vaccine expert at the Children's Hospital of Philadelphia, said it could limit access for people who still want boosters.
"Any use, say in a healthy 35-year-old, would be considered off-label, and you wonder whether an insurance company would pay for it," he told AFP.
- Not like annual flu shot -
Under the revised framework, companies like Pfizer and Moderna will be encouraged to test updated boosters in adults aged 50 to 64.
These studies should measure whether the vaccines reduce symptomatic infections, hospitalizations and deaths.
Rather than comparing new shots to earlier formulations, Prasad and Makary suggested placebo-controlled trials -- with saline as the comparator -- to better evaluate both benefit and potential side effects.
The proposal, first floated by Kennedy earlier this month, has proved divisive. Critics argue that using a placebo -- when authorized vaccines already exist -- could expose participants to unnecessary harm.
"Imagine if there was a death or two in the placebo group," said Offit. "I don't see how you conscience that."
Supporters of continued Covid-19 boosters often draw parallels to annual flu shots.
But Makary and Prasad pushed back on that comparison, arguing the genetic changes in Covid variants haven’t been significant enough to justify automatically updating the vaccine each year.
The FDA officials also sought to reassure Americans concerned they might lose access to boosters under the new framework.
The Centers for Disease Control and Prevention's (CDC) definition of risk factors is "vast, including obesity and even mental health conditions such as depression," they wrote, noting that between 100 million and 200 million Americans would likely still qualify.
L.Mason--AMWN


