
-
 Sabalenka downs Osaka to reach Madrid Open quarter-finals
Sabalenka downs Osaka to reach Madrid Open quarter-finals
-
'Nobody is better than us' says Luis Enrique as PSG prepare for Bayern

-
 Hridoy, Shamim pull off record home chase for Bangladesh against NZ
Hridoy, Shamim pull off record home chase for Bangladesh against NZ
-
Thrilling Kvaratskhelia hoping to drive PSG to another Champions League final

-
 Swiss canton votes with centuries-old show of hands
Swiss canton votes with centuries-old show of hands
-
Mali attacks kill defence minister, deepening security crisis

-
 How remarkable Sawe made marathon history in London
How remarkable Sawe made marathon history in London
-
British Open to be staged at Royal Lytham and St Annes in 2028

-
 Mbappe doubt for Clasico after Real Madrid confirm thigh injury
Mbappe doubt for Clasico after Real Madrid confirm thigh injury
-
Salah will get fitting Liverpool farewell despite injury, says Van Dijk

-
 African players in Europe: Injury may end Salah's Liverpool reign
African players in Europe: Injury may end Salah's Liverpool reign
-
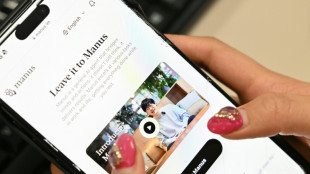
China blocks Meta's acquisition of AI firm Manus
-
 US woman speaks of ordeal in France Al-Fayed trafficking probe
US woman speaks of ordeal in France Al-Fayed trafficking probe
-
French teen faces jail in Singapore for licking vending machine straw

-
 Iran FM blames US for failure of talks after landing in Russia
Iran FM blames US for failure of talks after landing in Russia
-
Steep mountainside offers respite for daring Afghans

-
 Teenage wonder Sooryavanshi says criticism 'affects me a bit'
Teenage wonder Sooryavanshi says criticism 'affects me a bit'
-
Japan startup seeks approval of cat kidney disease treatment

-
 Technician dies installing stage for Shakira concert in Rio
Technician dies installing stage for Shakira concert in Rio
-
Cut off from the West, Muscovites rediscover Russian 'roots'

-
 'Joint venture in reverse': foreign carmakers seek edge with China partners
'Joint venture in reverse': foreign carmakers seek edge with China partners
-
Nations backing fossil fuel exit 'a new power': conference host Colombia

-
 Rockets thrash Lakers, Wembanyama triumphant on Spurs return
Rockets thrash Lakers, Wembanyama triumphant on Spurs return
-
ECB set to hold rates steady with eye on Iran crisis

-
 Team-first Kane propelling Bayern to glory as PSG showdown looms
Team-first Kane propelling Bayern to glory as PSG showdown looms
-
Pogacar vows to keep going until Seixas 'destroys' him

-
 From Adele to Raye, the UK school nurturing future stars
From Adele to Raye, the UK school nurturing future stars
-
Final talks begin on missing piece for pandemic treaty

-
 Oil rises, stocks swing as peace talk hopes wobble
Oil rises, stocks swing as peace talk hopes wobble
-
'Heartbroken' Xavi Simons out of World Cup and Spurs relegation fight

-
 North Korea's Kim reaffirms support for Russia's 'sacred' Ukraine war
North Korea's Kim reaffirms support for Russia's 'sacred' Ukraine war
-
Spurs win in Wembanyama return to take 3-1 lead over Trail Blazers

-
 As some hijabs come off in Iran, restrictions still in place
As some hijabs come off in Iran, restrictions still in place
-
Orangutan uses Indonesia canopy bridge in 'world first': NGO

-
 Dealing with the dead in the ruins of Sudan's war
Dealing with the dead in the ruins of Sudan's war
-
North Korea strengthens nuclear push as US flails in Middle East

-
 Stage set for Elon Musk's court battle with OpenAI
Stage set for Elon Musk's court battle with OpenAI
-
Caught between wars, US Afghan allies trapped in Qatar without safe exit

-
 British royals begin four-day US visit despite shooting
British royals begin four-day US visit despite shooting
-
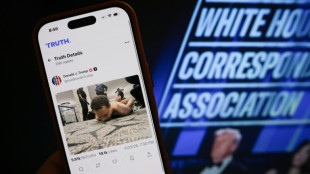
Suspect in shooting at Trump press dinner to appear in court
-
 Apex Auto Solutions Inc. Launches Mobile App to Provide Consumers With Centralized Financial Visibility
Apex Auto Solutions Inc. Launches Mobile App to Provide Consumers With Centralized Financial Visibility
-
Black Book Research Announces Release of State of Digital Healthcare IT: Finland 2026

-
 Truth & Purpose Learning Experience for Healthcare Professionals
Truth & Purpose Learning Experience for Healthcare Professionals
-
Ittihad Delivers record EBITDA with 22% YoY growth

-
 DDC Executive Vice President Named Dayton Business Journal Aerospace & Defense Awards Honoree
DDC Executive Vice President Named Dayton Business Journal Aerospace & Defense Awards Honoree
-
Tenzai Assembles Elite Security Leadership Team as AI Redefines Offensive Security

-
 ProScore Honors Apprenticeship Week and Highlights the Individuals Powering the Next Generations of Skilled Trades
ProScore Honors Apprenticeship Week and Highlights the Individuals Powering the Next Generations of Skilled Trades
-
Rain Enhancement Technologies Introduces Industry-First Guaranteed 10% Precipitation Enhancement

-
 Stagwell (STGW) Builds AI-Powered, Outcome-First TV Advertising Platform with FreeWheel
Stagwell (STGW) Builds AI-Powered, Outcome-First TV Advertising Platform with FreeWheel
-
Vertical Data Opens New Delhi Office to Serve India's AI Infrastructure Market


Adolore BioTherapeutics to Present CA8* Neuromodulating Gene Therapy at the 2025 NANS Annual Meeting
DELRAY BEACH, FL / ACCESS Newswire / February 4, 2025 / Adolore BioTherapeutics ("Adolore" or the "Company"), is developing intracellularly-delivered neuromodulators that naturally reduce neuronal excitability to restore balance to the PNS/CNS to treat the underlying root cause of serious neurological disorders, including chronic pain, today announced that Roy C. Levitt, MD, the Company's founder, Chief Medical Officer, and Executive Chairman, Clinical Professor, University of Miami, and Principal Investigator of a NIH, NINDS, HEAL UH3 Award will present at the 2025 North American Neuromodulation Society, ("NANS"), annual meeting being held January 30 - February 1, 2025 in Orlando, Fl.
"Neuromodulation using gene therapy is an exciting new area that has wide-spread applications in treating not only chronic pain but other serious undertreated neurological disorders. We are delighted to present our cutting-edge technology at NANS," commented Dr. Levitt. There is a paucity of safe, efficacious non-opioid analgesic treatments for chronic pain, creating a large and very urgent unmet medical need given the ongoing opioid crisis. Dr. Levitt will present Adolore's innovative CA8* chronic pain therapeutic (*carbonic anhydrase-like analgesic peptides, CA8 variants) and its cutting-edge technology using disease-free, nontoxic replication-defective Herpes Simplex Virus, ("rdHSV"), vectors. The company currently has two therapeutic programs in development, including peripheral neuropathy caused by erythromelalgia, which is an orphan disease, (ADB-101), and Adolore's lead program (ADB-102) treating patients with chronic pain caused by moderate-to-severe knee osteoarthritis. Based on compelling preclinical data, the Company is progressing these programs toward Investigational New Drug, ("IND"), filings and first-in-human clinical studies. These CA8* pain therapies were licensed in 2023 by Adolore from the University of Miami, Miami, FL. The rdHSV intracellular biotherapeutics delivery technology was licensed by Adolore from the University of Pittsburgh in 2023.
The Company's development program for the treatment of chronic pain due to moderate-to-severe knee osteoarthritis is supported by the NIH/NINDS HEAL UH3 Award to the University of Miami that funds all formal pre-clinical GLP/GMP/GCP development work through a first-in-human clinical study in patients, which is expected to commence in 2026.
For more information about the 2025 NANS Annual Meeting, visit the meeting website at: https://www.neuromodulation.org/annual-meeting-overview.html
About Adolore BioTherapeutics, Inc.
Adolore BioTherapeutics, Inc., is focused on developing novel intracellularly delivered treatments for chronic pain, drug-resistant epilepsy, seizure disorders, neurodegeneration and neurotrauma/neuroprotection, e.g., hearing loss. The Company's chronic pain treatment platform comprises two highly innovative technologies: our CA8* analgesic peptides, and our cutting-edge, disease-free, nontoxic rdHSV vector-based intracellular biotherapeutics delivery technology. Our best-in-class programs are long-acting, localized gene-therapies that are opioid-free Disease Modifying Anti-Pain therapies (DMAPs).
The Company's two current CA8* DMAP programs are in preclinical development for treating patients suffering from erythromelalgia, a life-long heritable chronic pain condition representing an orphan drug disease with no approved therapy, and patients suffering with chronic pain due to moderate-to-severe knee osteoarthritis, which affects a large number of patients that are often treated with opioids due to the lack of good alternatives, thus contributing to the ongoing opioid crisis.
For more information, visit adolore.com.
Forward Looking Statements
To the extent this announcement contains information and statements that are not historical, they are considered forward-looking statements within the meaning of the federal securities laws. You can identify forward-looking statements by the use of the words "believe," "expect," "anticipate," "intend," "estimate," "project," "will," "should," "may," "plan," "intend," "assume" and other expressions which predict or indicate future events and trends and which do not relate to historical matters. You should not rely on forward-looking statements, because they involve known and unknown risks, uncertainties and other factors, some of which are beyond the control of the Company. These risks and uncertainties include, but are not limited to, those associated with drug development. These risks, uncertainties and other factors may cause the actual results, performance or achievements of the Company to be materially different from the anticipated future results, performance or achievements expressed or implied by the forward-looking statements.
Investor Relations Contact
Paul Barone
(215)622-4542
[email protected]
SOURCE: Adolore Biotherapeutics, Inc.
View the original press release on ACCESS Newswire
F.Bennett--AMWN